Cell injury explanation notes
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
I now have all the content needed. Here are comprehensive, well-structured notes on cell injury:
Cell Injury — Study Notes
Source: Robbins & Kumar Basic Pathology; Robbins, Cotran & Kumar Pathologic Basis of Disease
Overview
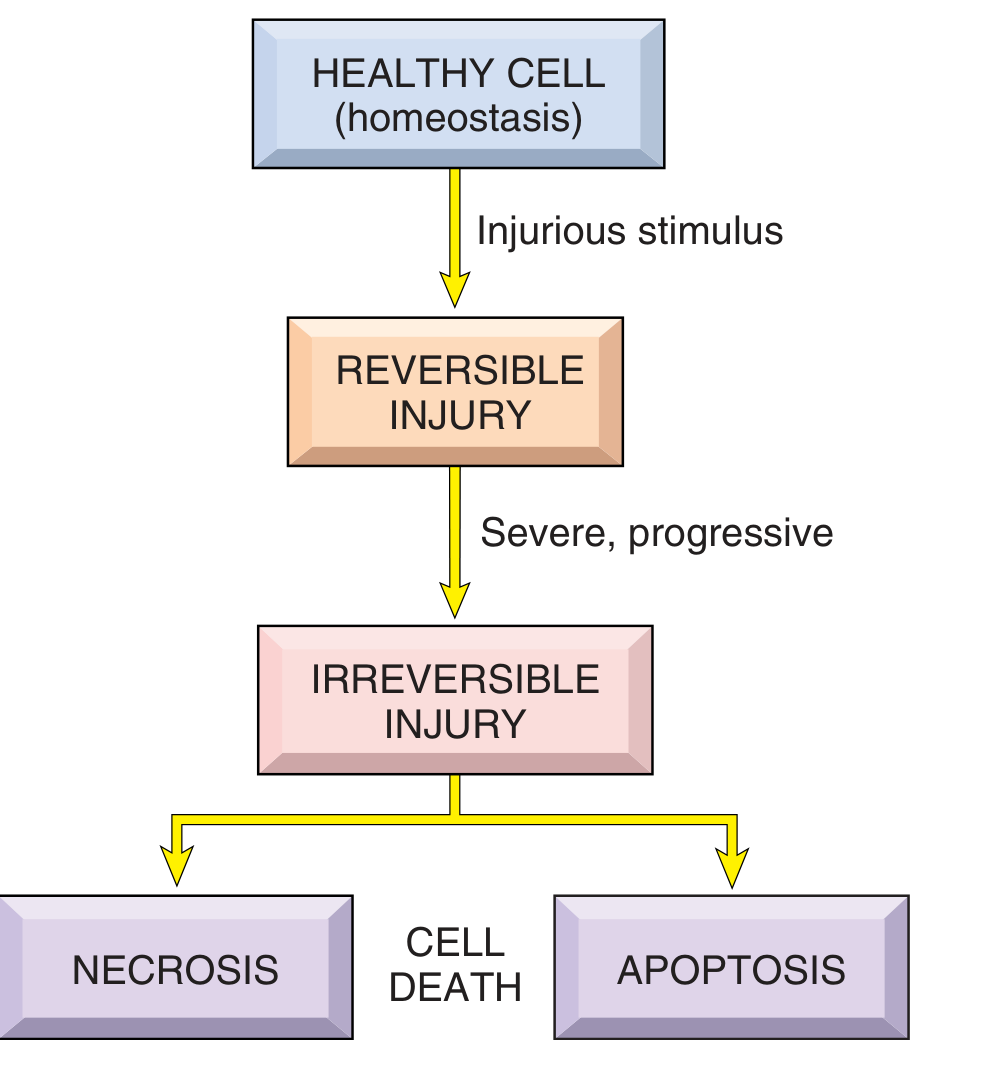
Cell injury is the basis of all disease. When a cell is stressed by an injurious stimulus, it can either adapt, sustain reversible damage, or die. The outcome depends on the type, duration, and severity of the stimulus, as well as the cell type and its metabolic state.
1. Causes of Cell Injury
| Category | Examples |
|---|---|
| Hypoxia / Ischemia | Arterial blockage, anemia, cardiorespiratory failure, CO poisoning |
| Toxins | Air pollutants, insecticides, carbon monoxide, asbestos, ethanol, drugs |
| Infectious agents | Viruses, bacteria, fungi, parasites (via toxins & immune responses) |
| Immunologic reactions | Autoimmunity, hypersensitivity, chronic inflammation |
| Genetic abnormalities | Enzyme defects (inborn errors of metabolism), DNA damage, misfolded proteins, chromosomal aberrations |
| Nutritional imbalances | Protein-calorie insufficiency, vitamin deficiencies, obesity |
| Physical agents | Trauma, extremes of temperature, radiation, electric shock |
Key principle: Low doses / brief duration → reversible injury; large doses / prolonged duration → irreversible injury and necrosis.
2. Reversible Cell Injury
Reversible injury is a derangement of function and morphology from which the cell can recover if the stimulus is removed.
Morphologic Features
- Cellular swelling (hydropic change / vacuolar degeneration) — earliest sign; small clear vacuoles in cytoplasm (distended ER segments); organ shows pallor, increased turgor and weight
- Fatty change — lipid vacuoles accumulate in organs active in lipid metabolism (e.g., liver)
- Cytoplasmic eosinophilia — loss of RNA (which binds the blue hematoxylin dye) makes cytoplasm appear redder
Ultrastructural Features (EM)
- Plasma membrane blebbing, blunting/loss of microvilli
- Mitochondrial swelling + small amorphous densities
- ER dilation with detachment of ribosomes and dissociation of polysomes
- Nuclear chromatin clumping
- Myelin figures — collections of phospholipids from damaged membranes
Mechanism
Reduced ATP → failure of Na⁺/K⁺-ATPase → intracellular Na⁺↑, K⁺↓ → osmotic water gain → cell swelling. Compensatory anaerobic glycolysis → lactic acid → ↓ intracellular pH → enzyme dysfunction.
3. Irreversible Injury ("Point of No Return")
Three hallmarks signal irreversibility:
- Inability to restore mitochondrial function (oxidative phosphorylation and ATP generation)
- Membrane damage — loss of structural integrity of plasma and intracellular membranes
- Loss of DNA/chromatin structural integrity
4. Cell Death
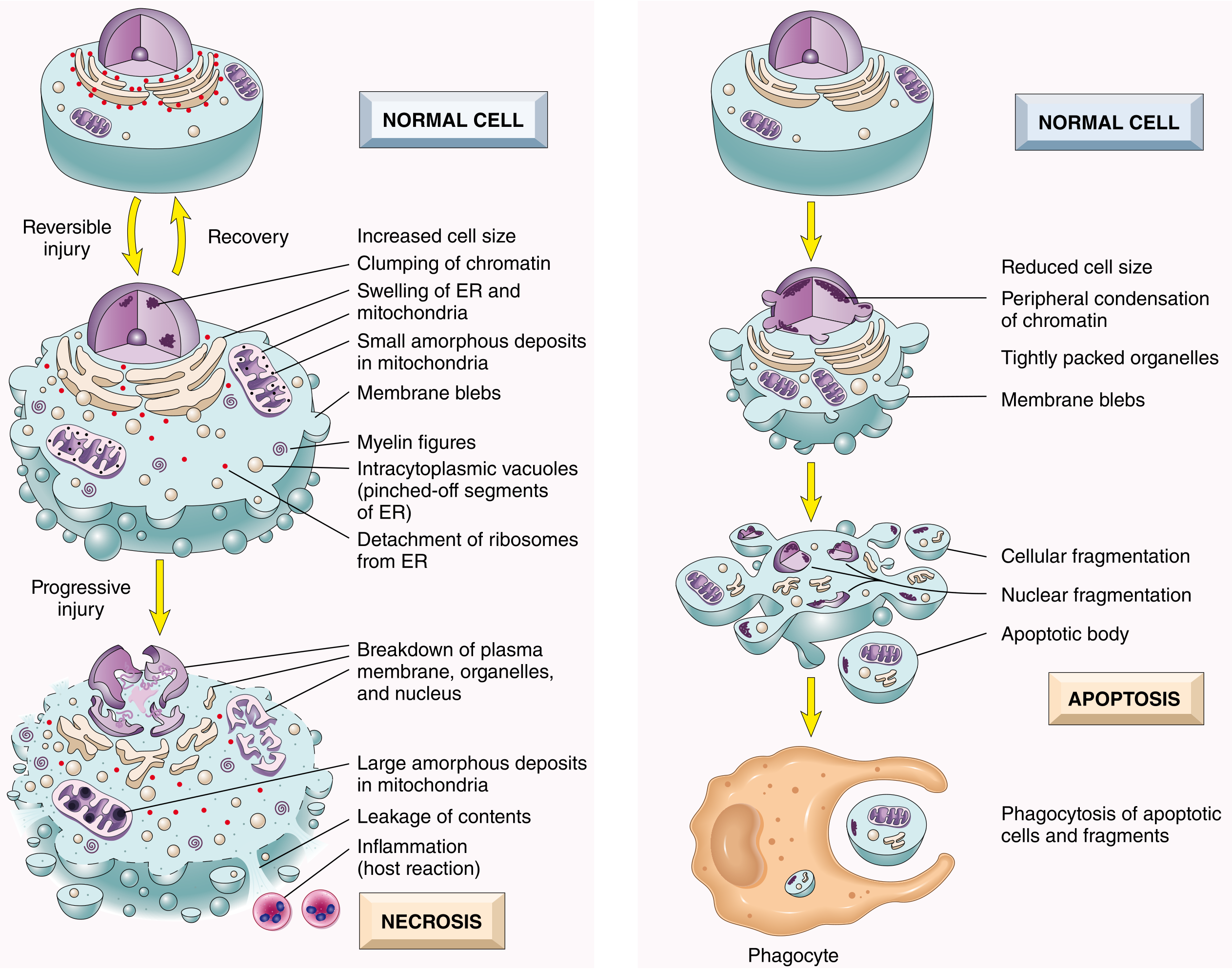
A. Necrosis
A pathologic process characterized by membrane destruction, enzyme leakage, and local inflammation.
Nuclear changes:
- Pyknosis — nuclear shrinkage and condensation
- Karyorrhexis — nuclear fragmentation
- Karyolysis — nuclear dissolution
Patterns of Tissue Necrosis:
| Pattern | Mechanism / Features | Examples |
|---|---|---|
| Coagulative | Protein denaturation preserves cell outlines; "ghost cells" | Infarcts of heart, kidney, spleen |
| Liquefactive | Enzymatic digestion dissolves tissue; liquefied mass | Brain infarct, bacterial abscesses |
| Caseous | Cheese-like whitish debris; structureless; granuloma | Tuberculosis |
| Fat | Lipases digest adipose tissue; calcium soap formation (saponification) | Acute pancreatitis, breast trauma |
| Fibrinoid | Immune complexes + fibrin deposits in vessel walls; bright pink | Vasculitis, malignant hypertension |
| Gangrenous | Coagulative necrosis + superimposed infection (wet) | Ischemic limb |
Clinical use: Tissue-specific proteins leak from necrotic cells into blood — troponin (cardiac), transaminases (liver), alkaline phosphatase (bile duct).
B. Apoptosis
A programmed, regulated cell death that eliminates unwanted cells with minimal inflammation.
Features:
- Cell shrinkage, chromatin condensation at periphery
- Cellular fragmentation into apoptotic bodies (membrane-bound)
- Plasma membrane remains intact — no leakage of contents
- Phosphatidylserine flips to outer leaflet → "eat me" signal for phagocytes → efferocytosis
- No inflammation (contrast with necrosis)
Physiologic apoptosis: embryogenesis, hormone-dependent involution (endometrium), lymphocyte selection, cell turnover in gut epithelium
Pathologic apoptosis: DNA damage, viral infections (e.g., hepatocyte death in viral hepatitis), self-reactive lymphocyte elimination
Two pathways of apoptosis:
- Intrinsic (mitochondrial) pathway — triggered by DNA damage, loss of growth factors, ER stress → BCL-2 family proteins regulate mitochondrial membrane permeability → cytochrome c release → caspase activation → cell death. Anti-apoptotic BCL-2 and BCL-XL keep this in check; pro-apoptotic BAX/BAK promote it.
- Extrinsic (death receptor) pathway — ligands (e.g., FasL, TNF) bind death receptors (Fas/CD95, TNFR1) → FADD recruitment → caspase-8 activation → execution caspases. Used by cytotoxic T lymphocytes; eliminates self-reactive lymphocytes.
Necrosis vs. Apoptosis — Quick Comparison
| Feature | Necrosis | Apoptosis |
|---|---|---|
| Cell size | Enlarged (swelling) | Reduced (shrinkage) |
| Nucleus | Pyknosis → karyorrhexis → karyolysis | Fragmentation into nucleosome-sized pieces |
| Plasma membrane | Disrupted | Intact until late |
| Cellular contents | Leaks into extracellular space | Packaged in apoptotic bodies |
| Inflammation | Yes — prominent | No |
| Cause | Pathologic (toxins, ischemia) | Physiologic or pathologic |
| Mechanism | Passive/unregulated | Programmed, caspase-mediated |
5. Other Forms of Cell Death
| Type | Key Features |
|---|---|
| Necroptosis | "Programmed necrosis" — morphology of necrosis but triggered by signaling pathways (RIPK3/MLKL); generates ROS; ruptures plasma membrane; causes inflammation |
| Pyroptosis | Caspase-1/11 activation (inflammasome); releases IL-1β and IL-18; proinflammatory; important in infection defense |
| Ferroptosis | Iron-dependent; driven by lipid peroxide accumulation; no caspase involvement |
| Autophagy | Cells digest own organelles in autophagosomes that fuse with lysosomes; survival mechanism under nutrient deprivation; can trigger apoptosis |
6. Mechanisms of Cell Injury
Mitochondrial Damage
- ATP depletion → Na⁺/K⁺-ATPase failure → cell swelling
- Anaerobic glycolysis → lactic acidosis
- Ribosome detachment → reduced protein synthesis
- Release of pro-apoptotic proteins (cytochrome c) → apoptosis
- Ultimately → irreversible membrane damage → necrosis
Oxidative Stress (ROS)
Reactive oxygen species (superoxide, hydrogen peroxide, hydroxyl radicals) damage:
- Membrane lipids (lipid peroxidation)
- Proteins (cross-linking, fragmentation)
- DNA (strand breaks → apoptosis)
ROS are generated by: mitochondrial leak, ionizing radiation, metabolism of drugs by cytochrome P-450, activated neutrophils/macrophages.
Removed by: superoxide dismutase (SOD), glutathione peroxidase, catalase, antioxidants (vitamins E, A, C).
Membrane Damage
- Loss of selective permeability → cellular swelling
- Lysosomal membrane rupture → enzymatic digestion of cell contents
- Plasma membrane disruption → protein leakage into blood (biomarkers)
DNA Damage
- Radiation or chemical damage → if unrepaired → p53 activation → apoptosis
Calcium Homeostasis Disturbance
- Ischemia/toxins → intracellular Ca²⁺↑ → activates phospholipases (membrane damage), proteases (protein breakdown), ATPases (↓ ATP), endonucleases (DNA fragmentation) → cell death
ER Stress
- Misfolded proteins accumulate in ER → unfolded protein response (UPR) → if unresolved → apoptosis
7. Ischemia–Reperfusion Injury
Paradox: restoring blood flow to ischemic tissue worsens damage because:
- Sudden oxygen reintroduction generates a burst of ROS
- Influx of Ca²⁺
- Neutrophil activation and inflammation
- Mitochondrial permeability transition pore (MPTP) opening → cell death
8. Cellular Adaptations to Sublethal Stress
| Adaptation | Definition | Example |
|---|---|---|
| Hypertrophy | Increased cell/organ size (no cell division) | Cardiac hypertrophy in hypertension |
| Hyperplasia | Increased cell number | Endometrial hyperplasia, liver regeneration |
| Atrophy | Decreased cell size/number | Muscle wasting, disuse |
| Metaplasia | Change of one differentiated cell type to another | Squamous metaplasia of bronchial epithelium in smokers (→ Barrett esophagus) |
9. Summary Flowchart
INJURIOUS STIMULUS
↓
REVERSIBLE INJURY
• Cell swelling (hydropic change)
• Fatty change
• Cytoplasmic eosinophilia
• Mitochondrial swelling
↓ (stimulus persists/worsens)
IRREVERSIBLE INJURY
↓ ↓
NECROSIS APOPTOSIS
(pathologic) (programmed)
• Inflammation • No inflammation
• Membrane • Apoptotic bodies
rupture • Phagocytosis
Robbins & Kumar Basic Pathology (9th ed.), Chapter 1 — Robbins, Cotran & Kumar Pathologic Basis of Disease (10th ed.), Chapter 2
This is a shared conversation. Sign in to Orris to start your own chat.