Classify antitubercular drugs. Explain the mechanisms of action of first-line anti-TB drugs. Discuss the antitubercular therapy for drug-sensitive tuberculosis in adults as per NTEP program uses of antitubercular drugs. 2nd yr mbbs refer kdt
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Searching the Web
NTEP program antitubercular therapy drug-sensitive TB adults 2023 regimen
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Reading Web Page
NTEP drug sensitive TB adult treatment regimen weight band FDC
I now have comprehensive information from textbooks and the official NTEP guidelines. Let me compose the complete answer:
Antitubercular Drugs — Classification, Mechanisms & NTEP Therapy
1. Classification of Antitubercular Drugs
A. Based on Efficacy (KDT/WHO Classification)
| Category | Drugs |
|---|---|
| First-line (Essential) | Isoniazid (H), Rifampicin (R), Pyrazinamide (Z), Ethambutol (E), Streptomycin (S) |
| Second-line | Fluoroquinolones (Levofloxacin, Moxifloxacin), Injectable agents (Amikacin, Kanamycin, Capreomycin), Ethionamide, Cycloserine, PAS (Para-aminosalicylic acid) |
| Third-line / Newer | Bedaquiline, Linezolid, Clofazimine, Delamanid, Pretomanid |
B. Based on Bactericidal vs. Bacteriostatic Action
| Action | Drugs |
|---|---|
| Bactericidal | Isoniazid, Rifampicin, Pyrazinamide, Streptomycin |
| Bacteriostatic | Ethambutol, PAS, Cycloserine, Ethionamide |
C. Based on Activity on Bacterial Populations (Sterilizing Activity)
- Rapidly multiplying extracellular bacilli → Isoniazid (most active), Rifampicin
- Slow/intermittently multiplying bacilli (caseous lesions) → Rifampicin
- Intracellular bacilli (acidic environment of macrophages) → Pyrazinamide (most active), Rifampicin
- Dormant bacilli → Rifampicin (some activity)
The combination of INH + RIF + PZA targets all three bacterial populations, which is the rationale for the 6-month regimen.
2. Mechanisms of Action of First-Line Anti-TB Drugs
A. Isoniazid (INH / H)
- Class: Isonicotinic acid hydrazide (synthetic)
- Mechanism: INH is a prodrug activated by the mycobacterial catalase-peroxidase enzyme KatG. The activated form forms a covalent complex with acyl carrier protein (AcpM) and KasA (β-ketoacyl-ACP synthase), blocking mycolic acid synthesis — an essential component of the mycobacterial cell wall. Inhibition of InhA (NADH-dependent acyl carrier protein reductase) also contributes.
- Bactericidal against rapidly dividing bacilli; bacteriostatic against dormant organisms
- Active against both intracellular and extracellular organisms
- Resistance: Mutation/deletion of katG (most common, high-level resistance); overexpression of inhA; mutations in kasA or ahpC
- Key pharmacology:
- Well absorbed orally (peak in 1–2 h, 3–5 µg/mL)
- Penetrates all body fluids including CSF (20–100% of serum levels)
- Metabolized by liver N-acetyltransferase (NAT2) — genetic polymorphism:
- Slow acetylators: t½ ≈ 3–4 h → higher plasma levels → more neuropathy risk
- Fast acetylators: t½ < 1 h → lower plasma levels
- Adverse effects:
- Hepatotoxicity (most serious — age >35, daily alcohol, co-rifampicin increases risk)
- Peripheral neuropathy (pyridoxine/B₆ deficiency — prevented by pyridoxine 10 mg/day)
- CNS effects (seizures in predisposed patients)
- Drug interaction: Inhibits CYP450 → potentiates phenytoin and carbamazepine toxicity
B. Rifampicin (Rifampin / R)
- Class: Rifamycin antibiotic (semisynthetic macrocyclic)
- Mechanism: Binds the β-subunit of mycobacterial DNA-dependent RNA polymerase, thereby blocking RNA transcription (inhibits initiation, not elongation). Bactericidal for both intracellular and extracellular organisms.
- Active against M. tuberculosis, NTM, M. leprae, and many gram-positive/gram-negative bacteria
- Resistance: Point mutation in rpoB gene (encodes β-subunit of RNA polymerase) → reduced drug binding affinity. rpoB mutation = surrogate marker for MDR-TB.
- Key pharmacology:
- Well absorbed orally; distributed widely including into CSF (10–20% of serum levels; higher with meningeal inflammation)
- Undergoes enterohepatic recirculation; excreted mainly in bile/feces
- Potent inducer of CYP1A2, 2C9, 2C19, 2D6, 3A4 → major drug interactions (reduces levels of OCP, antiretrovirals, warfarin, cyclosporine, methadone)
- Autoinduction of its own metabolism (t½ shortens over first 1–2 weeks)
- Adverse effects:
- Orange discoloration of urine, sweat, tears, saliva (harmless; may stain soft contact lenses permanently)
- Hepatotoxicity (cholestatic jaundice, hepatitis)
- Flu-like syndrome (if given less than twice weekly — intermittent dosing)
- Thrombocytopenia, nephritis, rashes
C. Pyrazinamide (PZA / Z)
- Class: Synthetic analog of nicotinamide
- Mechanism: A prodrug hydrolyzed by mycobacterial pyrazinamidase to pyrazinoic acid (active form). Exact mechanism remains incompletely understood but it:
- Disrupts membrane energy metabolism and the proton motive force
- Inhibits fatty acid synthesis (FAS I system)
- Active only in acidic pH (pH < 6) — therefore uniquely effective against intracellular bacilli in the acidic environment of macrophage phagolysosomes and caseous lesions
- Sterilizing activity in this population allows shortening of treatment from 9 months to 6 months
- Resistance: Loss of pyrazinamidase enzyme activity (mutations in pncA gene)
- Pharmacology: Good oral absorption; penetrates CSF well; discontinued after 2 months of the 6-month regimen (most benefit early)
- Adverse effects:
- Hepatotoxicity (dose-dependent)
- Hyperuricemia (inhibits uric acid excretion) → arthralgia; rarely precipitates gout
- Nausea, rash, flushing, photosensitivity
D. Ethambutol (EMB / E)
- Class: Synthetic ethylene diamine derivative
- Mechanism: Inhibits arabinosyl transferase (encoded by embB gene) — an enzyme essential for the synthesis of arabinogalactan, a key component of the mycobacterial cell wall. Disrupts cell wall integrity and allows other drugs better penetration.
- Bacteriostatic (at standard doses); bactericidal at higher doses
- Active only against multiplying mycobacteria
- Resistance: Mutations in embB gene
- Pharmacology: Well distributed; variable CSF penetration; excreted mainly in urine (reduce dose in renal failure)
- Adverse effects:
- Retrobulbar (optic) neuritis — most important toxicity
- Manifests as: decreased visual acuity, red-green colour blindness, central scotoma
- Dose-dependent; reversible if stopped early
- Baseline and monthly visual acuity + colour vision testing mandatory
- Hyperuricemia (mild)
- Avoid in children <5 years (cannot report visual symptoms reliably)
- Retrobulbar (optic) neuritis — most important toxicity
E. Streptomycin (S) — Historical First-Line; Still Used in Special Cases
- Class: Aminoglycoside
- Mechanism: Binds 30S ribosomal subunit (16S rRNA) → misreading of mRNA → faulty protein synthesis → bactericidal. Active only against extracellular bacilli (cannot penetrate macrophages; inactive in acidic pH).
- Route: IM only (not orally absorbed)
- Adverse effects: Ototoxicity (vestibular > cochlear), nephrotoxicity; contraindicated in pregnancy
Summary Diagram of Mechanisms
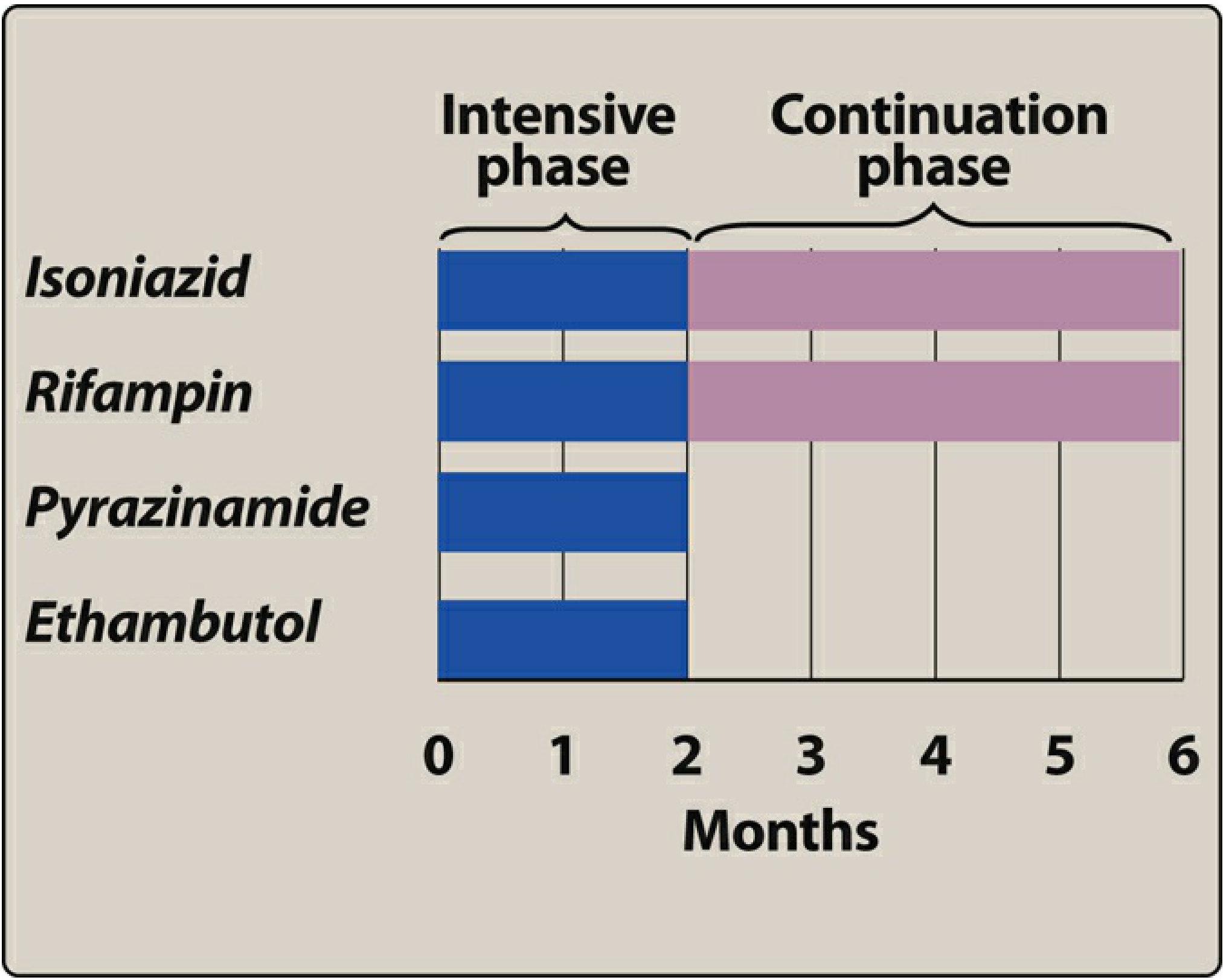
3. NTEP Antitubercular Therapy for Drug-Sensitive TB in Adults
Background & Principles
The National Tuberculosis Elimination Programme (NTEP) — formerly RNTCP — governs TB care in India under the Ministry of Health & Family Welfare. Key principles:
- Daily directly observed treatment (DOT) — mandatory
- Fixed-Dose Combinations (FDCs) — prescribed as per weight bands to ensure correct dosing
- Universal Drug Susceptibility Testing (UDST) at diagnosis
- Treatment in public or private sector follows the same regimen; FDCs supplied by NTEP
Standard Regimen for Drug-Sensitive TB (DS-TB)
Regimen: 2HRZE / 4HRE
| Phase | Duration | Drugs | Doses |
|---|---|---|---|
| Intensive Phase (IP) | 2 months (56 doses) | H + R + Z + E (4-FDC) | Daily |
| Continuation Phase (CP) | 4 months (112 doses) | H + R + E (3-FDC) | Daily |
Total duration: 6 months | Total doses: 168
FDC Composition
| Phase | FDC | Composition per tablet |
|---|---|---|
| Intensive Phase | 4-FDC | Isoniazid 75 mg + Rifampicin 150 mg + Pyrazinamide 400 mg + Ethambutol 275 mg |
| Continuation Phase | 3-FDC | Isoniazid 75 mg + Rifampicin 150 mg + Ethambutol 275 mg |
Weight-Band Dosing (Adults)
| Weight Category | IP Tablets (4-FDC) | CP Tablets (3-FDC) |
|---|---|---|
| 25–34 kg | 2 tablets | 2 tablets |
| 35–49 kg | 3 tablets | 3 tablets |
| 50–64 kg | 4 tablets | 4 tablets |
| 65–75 kg | 5 tablets | 5 tablets |
| > 75 kg | 6 tablets | 6 tablets |
If patient's weight increases by >5 kg crossing a weight band during treatment → upgrade to next higher band
Standard Drug Doses (Reference)
| Drug | Dose |
|---|---|
| Isoniazid (H) | 5 mg/kg/day (4–6 mg/kg) |
| Rifampicin (R) | 10 mg/kg/day (8–12 mg/kg) |
| Pyrazinamide (Z) | 25 mg/kg/day (20–30 mg/kg) |
| Ethambutol (E) | 15 mg/kg/day (12–18 mg/kg) |
Special Considerations Under NTEP
| Situation | Modified Regimen |
|---|---|
| TB Meningitis | IP: 2 months RHZE (or RHZS) → CP: RHE for 10 months (total 12 months) + corticosteroids (Dexamethasone 0.4 mg/kg/day IV, then oral taper over 8–12 weeks) |
| Osteoarticular TB | 2HRZE + 10HRE (total 12 months) |
| INH-mono/poly resistant TB | Replace INH with Levofloxacin → 6–9 month regimen |
| HIV co-infection | Same regimen; use Rifabutin instead of Rifampicin if on protease inhibitors |
| Pregnancy | Avoid Streptomycin (teratogenic/ototoxic to fetus); standard HRZE safe |
| Hepatic disease | Reduce hepatotoxic drug burden |
| Renal failure | Reduce Ethambutol dose; Streptomycin avoided |
Rationale for 4-Drug Regimen
The probability of spontaneous resistance to any single drug is ~1 in 10⁵ bacilli. Since a tuberculous lesion may contain >10⁸ organisms, two drugs alone still allow selection of doubly-resistant mutants (~1/10¹⁰). Using four drugs in the intensive phase reduces this probability to ~1 in 10²⁰ — effectively preventing resistance emergence. — Katzung, 16th ed.
Pyridoxine (Vitamin B₆) Supplementation
Pyridoxine 10 mg/day is co-administered with INH to prevent peripheral neuropathy, especially in:
- Malnourished patients
- Pregnant women
- Elderly
- Diabetics
- Alcoholics
- HIV-positive patients
Monitoring During NTEP Therapy
| Parameter | Timing |
|---|---|
| Sputum smear/culture | End of IP (month 2), month 5, end of treatment |
| Liver function tests | Baseline; repeat if symptomatic |
| Visual acuity + colour vision | Baseline; monthly (for Ethambutol) |
| Weight | Monthly (for weight-band adjustment) |
| Uric acid | Baseline (Pyrazinamide) |
Sources: Katzung's Basic & Clinical Pharmacology 16e, p. 1312–1315 | Lippincott Illustrated Reviews: Pharmacology, p. 1055–1064 | NTEP Drug-Sensitive TB Treatment Guidelines (ICMR/MoHFW)
Uses
Asking for Preferences
Reading File
Finding Sources
Finding Sources
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Finding Sources
Finding Sources
I have all the data needed. Here is the comprehensive answer:
Uses of First-Line Antitubercular Drugs
1. Isoniazid (INH / H)
TB-Related Uses
| Use | Details |
|---|---|
| Active TB (drug-sensitive) | Core drug in all first-line regimens. Given throughout both intensive and continuation phases (2HRZE/4HRE) |
| Latent TB infection (LTBI) prophylaxis | INH 300 mg/day × 6 months (or 9 months in HIV+) — most widely used preventive therapy |
| TB contacts | Chemoprophylaxis in household contacts of smear-positive TB patients |
| Immunosuppressed patients | Before starting steroids, TNF-α inhibitors, anti-cancer therapy — prevents reactivation |
Non-TB Uses
- M. kansasii infection (only NTM susceptible to INH at standard doses)
INH is not used as monotherapy for active TB — only for LTBI prophylaxis as a single agent.
2. Rifampicin (Rifampin / R)
Rifampicin has the broadest clinical uses among all antitubercular drugs.
TB-Related Uses
| Use | Details |
|---|---|
| Active drug-sensitive TB | Backbone of all TB regimens; used throughout 6-month treatment |
| LTBI prophylaxis | Rifampicin 600 mg daily × 4 months as monotherapy — effective alternative to INH |
| MDR/pre-XDR-TB | Used only if confirmed susceptibility |
Non-TB Mycobacterial Uses
| Infection | Use |
|---|---|
| Leprosy | Part of WHO multidrug therapy (MDT) — paucibacillary and multibacillary leprosy |
| MAC (M. avium complex) | Combined regimen in AIDS patients |
| M. kansasii | Combination therapy |
Non-Mycobacterial Uses
| Infection | Regimen |
|---|---|
| Meningococcal prophylaxis | 600 mg twice daily × 2 days (eliminates nasopharyngeal carriage) |
| H. influenzae type b prophylaxis | 20 mg/kg/day × 4 days (contacts of children with Hib meningitis) |
| Staphylococcal infections | Prosthetic valve endocarditis, prosthetic joint infections, osteomyelitis — always in combination (never monotherapy) |
| Brucellosis | Rifampicin + doxycycline (WHO preferred regimen) |
Rifampicin is never used as monotherapy for active TB — rapid resistance develops.
3. Pyrazinamide (PZA / Z)
Uses
| Use | Details |
|---|---|
| Active TB — Intensive phase only | Used only for the first 2 months of the 6-month regimen; its sterilizing activity in acidic macrophage environment allows shortening treatment from 9 to 6 months |
| TB meningitis | Included in both phases due to excellent CSF penetration |
| TB in HIV | Included in all short-course regimens |
Non-TB Uses
- No significant non-TB uses — PZA is highly specific for Mycobacterium tuberculosis
- No activity against M. bovis (intrinsically resistant — lacks pyrazinamidase)
Rationale for limiting PZA to 2 months: most clinical benefit is early; continued use increases hepatotoxicity risk without adding sterilizing benefit.
4. Ethambutol (EMB / E)
TB-Related Uses
| Use | Details |
|---|---|
| Active TB | Used in intensive phase (2 months); protects against resistance to INH/RIF; can be discontinued once susceptibility is confirmed |
| TB meningitis | Included in extended regimens |
| TB in HIV | Part of standard regimen |
Non-TB Mycobacterial Uses
| Infection | Role |
|---|---|
| MAC (M. avium complex) | Combination therapy — standard MAC regimen: clarithromycin + ethambutol ± rifabutin |
| M. kansasii | Combination therapy |
| M. gordonae, M. marinum, M. szulgai | Activity demonstrated |
Ethambutol is not used as monotherapy — it is bacteriostatic and serves primarily as a companion drug to prevent emergence of resistance.
5. Streptomycin (S)
TB-Related Uses
| Use | Details |
|---|---|
| Active TB | Alternative to Ethambutol in the intensive phase (when EMB cannot be used e.g., in young children, ocular disease); notation: 2HRZE or 2HRZS |
| TB meningitis | NTEP allows 2 RHZS as an alternative IP in meningitis |
| MDR-TB regimens | Reserve injectable agent |
Non-TB Uses
| Infection | Details |
|---|---|
| Plague (Yersinia pestis) | Drug of choice (IM) |
| Tularemia (Francisella tularensis) | Drug of choice |
| Brucellosis | Streptomycin + doxycycline (WHO alternative to rifampicin regimen) |
| Enterococcal endocarditis | Combined with ampicillin/penicillin (synergistic bactericidal effect) |
| Granuloma inguinale | Alternative agent |
Streptomycin is IM-only (not orally absorbed) and acts only against extracellular bacilli. Contraindicated in pregnancy (8th nerve damage in fetus) and avoided in renal failure and elderly.
Quick Reference Summary
| Drug | Primary TB Use | Key Non-TB Uses |
|---|---|---|
| Isoniazid | Active TB + LTBI prophylaxis | M. kansasii |
| Rifampicin | Active TB + LTBI (4 months mono) | Leprosy, meningococcal/Hib prophylaxis, Brucellosis, Staph infections |
| Pyrazinamide | Active TB — IP only (2 months) | None (TB-specific) |
| Ethambutol | Active TB — IP; resistance prevention | MAC, M. kansasii |
| Streptomycin | Active TB (alt. to EMB); TB meningitis | Plague, Tularemia, Brucellosis, Enterococcal endocarditis |
Sources: Goodman & Gilman's Pharmacological Basis of Therapeutics, p. 1302 | Katzung's Basic & Clinical Pharmacology 16e | Lippincott Illustrated Reviews: Pharmacology, p. 1055–1064 | NTEP Guidelines (MoHFW India)
Classify the anti-retroviral drugs therapy and discuss their mechanisms of action in details 2nd yr mbbs refer kdt
Reading File
Your credits are exhausted. Your daily credits reset tomorrow, or you can purchase a top-up for more.
This is a shared conversation. Sign in to Orris to start your own chat.