Prenatal anomaly screening and diagnosis
Reading File
Finding Sources
Finding Sources
Searching PubMed
"prenatal screening" AND "fetal anomaly"
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
Finding Sources
I now have rich content from three major textbooks. Let me compile a comprehensive, well-structured response.
Prenatal Anomaly Screening and Diagnosis
Prenatal anomaly assessment encompasses a spectrum of screening and diagnostic tools, stratified by gestational age, invasiveness, and the type of anomaly being detected. The goal is to identify chromosomal abnormalities, structural malformations, single-gene disorders, and inherited conditions — balancing detection rates against procedure-related risks.
1. Overview and Principles
Screening estimates the probability of a condition in an asymptomatic population; it is not diagnostic. A positive screen triggers an offer of diagnostic testing.
Diagnostic testing (amniocentesis, CVS) provides a definitive result but carries a small procedural risk. ACOG now recommends that all women — regardless of age — be offered both screening and diagnostic options. The decision to proceed is personal, influenced by values, religious beliefs, and the perceived burden of a genetic condition.
Thompson & Thompson Genetics and Genomics in Medicine, 9th ed.
2. First-Trimester Screening (11–13+6 weeks)
2a. Combined First-Trimester Test
The most widely used first-trimester screen integrates three parameters:
| Component | Analyte / Measure | Direction in Trisomy 21 |
|---|---|---|
| Nuchal Translucency (NT) | Ultrasound (mm) | ↑ |
| Free β-hCG | Maternal serum | ↑ |
| PAPP-A | Maternal serum | ↓ |
| Maternal age | Risk modifier | — |
Performance (for 5% screen-positive rate): ~88% detection rate for Down syndrome (trisomy 21). In women >35, detection rises to 90–92% with a 16–22% FPR. For trisomy 18, >90% of affected pregnancies are screen-positive with combined biochemical + NT screening.
| Study | N Screened | Detection Rate |
|---|---|---|
| Wapner et al. 2003 (BUN) | 8,216 | 79% |
| Malone et al. 2005 (FASTER) | 38,033 | 86% |
| Wald et al. 2003 (SURUSS) | 47,053 | 83% |
| Nicolaides et al. 2005 | 75,821 | 93% |
| Total | 167,210 | 88.2% |
Creasy & Resnik's Maternal-Fetal Medicine, Principles and Practice
2b. Nuchal Translucency (NT) Measurement
NT is measured as the subcutaneous fluid-filled space at the back of the fetal neck in a sagittal plane between 11–13+6 weeks. Correct caliper placement (measuring inner-to-inner edge, with the fetus in a neutral position) is critical for accuracy. Quality monitoring programs use epidemiologic review of individual operator performance rather than expert image review.
Elevated NT (>3.5 mm): Even with a normal karyotype and normal chromosomal microarray, an elevated NT warrants:
- Detailed first-trimester anatomy scan + echocardiogram at 13–14 weeks
- Repeat second-trimester scan + echo
- Chromosomal microarray (detects pathogenic CNVs in ~4–7% of isolated elevated NT cases)
- RASopathy/Noonan syndrome panel (pathogenic variants found in ~5.2% when NT >4 mm; incidence rises to ~1:90 vs. population 1:1,000)
- Consider whole-exome sequencing if additional anomalies present
Most common pathogenic CNVs with elevated NT: 22q11.2 deletion, 22q11.2 duplication, 10q26.12q26.3 deletion, 12q21q22 deletion.
2c. First-Trimester Ultrasound Markers
- Nasal bone (NB) absence: Absent in ~25% of Down syndrome fetuses (vs. 0% of euploid controls in some series). Adds independent screening value to NT + biochemistry.
- Ductus venosus flow, tricuspid regurgitation: Additional soft markers used in specialist centers.
3. Second-Trimester Screening (14–20 weeks)
When first-trimester screening has not been performed, the quadruple screen (14–20 weeks) is offered:
| Analyte | Trisomy 21 | Open NTD |
|---|---|---|
| AFP (α-fetoprotein) | ↓ | ↑↑↑ |
| hCG | ↑ | — |
| Unconjugated estriol (uE3) | ↓ | — |
| Inhibin A | ↑ | — |
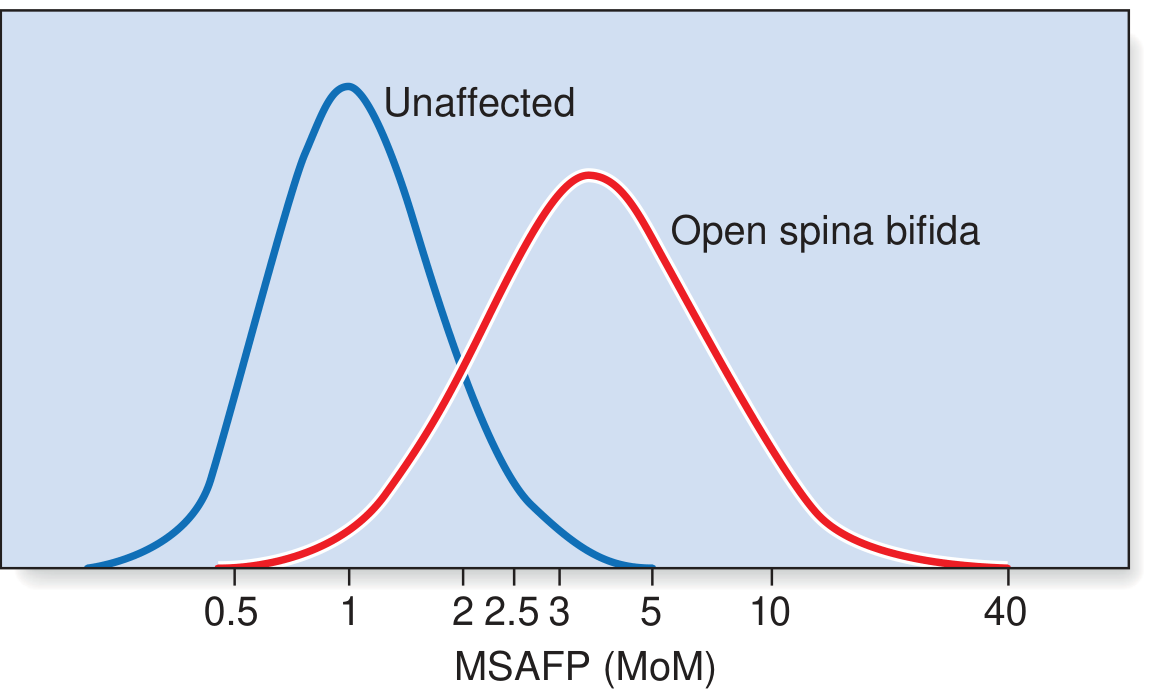
MSAFP for neural tube defects (NTDs): A cutoff of ≥2.5 MoM at 16 weeks identifies the majority of open NTDs, which was first recognised in 1972. The distributions of MSAFP in unaffected vs. open spina bifida pregnancies overlap significantly, necessitating confirmatory ultrasound:
The quadruple screen detects ~70–75% of Down syndrome cases — less accurate than the combined first-trimester test.
4. Non-Invasive Prenatal Testing (NIPT) / Cell-Free Fetal DNA (cffDNA)
Principle: Fetal cell-free DNA fragments circulate in maternal plasma from early pregnancy (~10 weeks). If the fetus has trisomy 21, ~1.05× more chromosome 21 fragments appear relative to disomic controls. By massively parallel sequencing of millions of fragments and quantifying their chromosomal origin, aneuploidies are detected with very high sensitivity.
Two technical approaches:
- Massively parallel shotgun sequencing (MPSS) — sequences all genomic fragments, identifies chromosomal origin of each
- Targeted (chromosome-selective) sequencing — focuses on chromosomes 13, 18, 21, X, Y; reduces cost ~10-fold and enables benchtop sequencers
Performance (meta-analysis, singleton pregnancies):
| Condition | Detection Rate | False-Positive Rate | +LR |
|---|---|---|---|
| Trisomy 21 | 99.7% (99.1–99.9%) | 0.04% | 2,506 |
| Trisomy 18 | 97.9% (94.9–99.1%) | ~0% | 2,122 |
| Trisomy 13 | 99.0% (65.8–100%) | 0.04% | 2,819 |
| Monosomy X | 95.8% (70.3–99.5%) | 0.14% | 694 |
| 47,XXX / 47,XYY / 47,XXY | 100% (83.6–100%) | 0.004% | NA |
Creasy & Resnik's Maternal-Fetal Medicine
Key limitations:
- NIPT is a screening test, not diagnostic — positive results require confirmatory invasive testing
- Relies on adequate fetal fraction (typically ≥4%); low fetal fraction (obesity, early GA) can cause no-call results
- Cannot reliably detect all structural anomalies or single-gene disorders
- 22q11.2 deletion testing by cffDNA is available but has lower sensitivity/specificity than microarray
A 2024 Cochrane meta-analysis (PMID: 38721874) confirmed ultrasound screening performance for structural abnormalities across first and second trimesters in low-risk/unselected populations — a recent evidence update on detection rates.
5. Invasive Diagnostic Procedures
5a. Indications (Box 18.1 — Thompson & Thompson)
- Patient preference / wishes for diagnostic certainty
- Positive maternal screening result (serum, NT, NIPT)
- Advanced maternal age (now offered to all, not age-restricted)
- Family history of chromosomal abnormality
- Previous child with trisomy or unbalanced chromosomal rearrangement
- Parent known to carry a chromosomal rearrangement (inversion, translocation)
- Elevated NT + normal NIPT (microarray + karyotype indicated)
- Risk for a specific single-gene disorder (CF, thalassemia, Huntington's, etc.)
- Fetal structural anomaly on ultrasound
5b. Chorionic Villus Sampling (CVS)
- Timing: 10–13 weeks
- Route: Transcervical or transabdominal
- Advantage: Earlier result than amniocentesis (first trimester)
- Cells analysed: Cytotrophoblast (rapid, lower resolution) vs. mesenchymal core (long culture, higher resolution, more representative of fetal lineage)
- Result time: Short-term culture result in days; long-term culture 10–14 days
- Complications: Procedure-related fetal loss ~1 in 450 (≈ background 2–5%); limb reduction defects reported historically but not confirmed in large series performed >10 weeks GA by experienced physicians
- Mosaicism: ~1% of CVS yield ambiguous mosaicism — follow-up amniocentesis may be needed
5c. Amniocentesis
- Timing: Typically 15–18 weeks (can be earlier or later)
- Cells analysed: Amniocytes (fetal cells in fluid); cultured for 10–14 days for karyotype
- Lower mosaicism rate than CVS
- Also used for: AFP in amniotic fluid (NTD diagnosis), viral studies, enzyme assays
- Procedure-related fetal loss: ~1 in 450 (comparable to CVS in experienced centres)
ACOG recommendation: If invasive testing proceeds, perform both karyotype AND chromosomal microarray (CMA) on the extracted fetal DNA, as CMA detects clinically significant copy number variants (submicroscopic) not visible on standard karyotype.
6. Laboratory Studies on Fetal Samples
| Test | Resolution | Detects |
|---|---|---|
| Karyotype | ~5–10 Mb | Trisomies, monosomies, large structural rearrangements |
| Chromosomal Microarray (CMA) | 50–100 kb | Submicroscopic deletions/duplications (CNVs) |
| FISH | Targeted | Rapid aneuploidy for chr 13, 18, 21, X, Y |
| Single-gene sequencing / panels | Point mutations | CF, SMA, monogenic disorders |
| Whole Exome/Genome Sequencing | Base-pair | Novel/rare pathogenic variants, especially with anomalies |
CMA yield: In fetuses with structural anomalies and a normal karyotype, CMA identifies an additional pathogenic CNV in ~6% of cases. For elevated NT + normal karyotype, yield is 4–7%.
7. Carrier Screening
Carrier screening identifies heterozygous carriers of autosomal recessive or X-linked recessive conditions. Modern practice has shifted from ancestry-based to pan-ethnic expanded panels:
- Previously: Targeted by ancestry — thalassaemia (Mediterranean/Asian), sickle cell (African), cystic fibrosis (Northern European), Ashkenazi Jewish panel (Tay-Sachs, CF, familial dysautonomia, Canavan, Gaucher, etc.)
- Now (ACOG recommended): Broad pan-ethnic carrier screening panels covering >200 conditions, regardless of stated ancestry — because precise ancestry is increasingly difficult to assign
Thompson & Thompson Genetics and Genomics in Medicine, 9th ed.
8. Second-Trimester Anatomy Scan (18–22 weeks)
The fetal anomaly scan (also called the "20-week scan") is a systematic real-time ultrasound evaluation of:
| System | Structures assessed |
|---|---|
| CNS | Head shape, BPD, cerebellum, cisterna magna, choroid plexus, spine |
| Face | Orbits, lips, palate (limited) |
| Cardiac | 4-chamber view, outflow tracts |
| Thorax | Lung echogenicity, diaphragm integrity |
| Abdomen | Stomach, abdominal wall, cord insertion |
| Renal | Kidneys, bladder |
| Limbs | Long bone measurements, hands/feet |
| Placenta/fluid | Placental location, liquor volume, umbilical cord |
Detection rates vary by organ system, operator experience, maternal BMI, and fetal position. Major structural anomalies (anencephaly, major cardiac defects, exomphalos, gastroschisis, major renal anomalies) are generally detected at higher rates than minor or subtle ones.
The 2024 Cochrane review (PMID 38721874) evaluated detection accuracy across first and second trimester scans in low-risk populations, providing the most current meta-analytic evidence for counselling.
9. Prenatal Diagnosis of Specific Conditions
Neural Tube Defects
- MSAFP ≥2.5 MoM at 16 weeks → detailed ultrasound / fetal anatomy scan
- "Lemon sign" (frontal bossing) and "banana sign" (cerebellum) on ultrasound are hallmarks of open spina bifida
- Amniocentesis for amniotic fluid AFP + acetylcholinesterase can confirm open NTD
Haemoglobin Disorders
- Sickle cell disease, thalassaemia: DNA-based diagnosis on CVS or amniocyte DNA
- Carrier couples identified on preconception / first antenatal visit screening → offered invasive testing
Single-Gene Disorders
Numerous autosomal dominant, recessive, and X-linked disorders can be diagnosed prenatally once the family's specific mutation is known:
- CF (CFTR), SMA (SMN1 deletion), Huntington's (CAG repeat), Duchenne MD (dystrophin)
- Preimplantation genetic testing (PGT) is an alternative avoiding the need for pregnancy termination
10. Genetic Counselling
Genetic counselling is integral throughout. The prenatal diagnosis team (physician, genetic counsellor, nurse) must:
- Obtain accurate three-generation pedigree
- Offer pre-test and post-test counselling for all screens and diagnostic tests
- Clarify the distinction between screening and diagnosis
- Present options non-directively, respecting autonomy, cultural and religious values
- Discuss the limitations of each test — no test excludes all abnormalities
Post-result counselling should address:
- Implications of positive/negative/uncertain results (variants of uncertain significance — VUS)
- Reproductive options: continuation of pregnancy, termination, adoption, fetal therapy
- Family implications (carrier status of parents and siblings)
11. Fetal Therapy — When Diagnosis Leads to Intervention
Prenatal diagnosis increasingly opens the door to in utero therapy:
| Condition | Intervention |
|---|---|
| Twin-twin transfusion syndrome | Laser ablation of communicating placental vessels (standard of care) |
| Fetal anaemia (Rh alloimmunisation) | Intrauterine transfusion |
| Myelomeningocele (spina bifida) | Open or laparoscopic in utero repair (MOMS trial) |
| Congenital diaphragmatic hernia | Tracheal balloon occlusion (FETO) in selected centres |
| Bladder outlet obstruction | Vesicoamniotic shunt |
| Fetal arrhythmias | Maternal digoxin / sotalol |
| Fetal rhabdomyomas | Sirolimus (established efficacy) |
| Hypoplastic left heart / critical AS | Aortic valvuloplasty / balloon septostomy |
| Hydrops | Drain pleural effusion (shunt) |
A fetal karyotype at minimum (and usually CMA) is recommended before any fetal procedure, with multidisciplinary prenatal genetic counselling integral to decision-making.
Thompson & Thompson Genetics and Genomics in Medicine, 9th ed.
Summary: Screening vs. Diagnostic Pathway
All pregnant women
│
├──► CARRIER SCREENING (preconception / early 1st trimester)
│ Pan-ethnic expanded panel
│
├──► 1ST TRIMESTER COMBINED TEST (11–13+6 weeks)
│ NT + free β-hCG + PAPP-A + maternal age
│ Detection ~88% for T21 (5% FPR)
│
├──► NIPT / cffDNA (from ~10 weeks)
│ T21 DR 99.7%, T18 DR 97.9%
│ Positive → confirm with invasive testing
│
├──► ANATOMY SCAN (18–22 weeks)
│ Structural anomaly detection
│ AFP for NTD (if 2nd trimester serum screen)
│
↓ High-risk screen result, anomaly, family history
INVASIVE DIAGNOSTIC TESTING
CVS (10–13 wks) or Amniocentesis (≥15 wks)
│
├─ Karyotype (5–10 Mb resolution)
├─ CMA / microarray (50–100 kb, recommended routinely)
├─ FISH (rapid, targeted)
├─ Single-gene / panel testing
└─ WES/WGS (structural anomalies + normal karyotype/CMA)
Key references:
- Creasy & Resnik's Maternal-Fetal Medicine, Chs. 30–31
- Thompson & Thompson Genetics and Genomics in Medicine, 9th ed., Ch. 18
- Emery's Elements of Medical Genetics and Genomics, Ch. 20
- Buijtendijk et al. (2024). Diagnostic accuracy of ultrasound screening for fetal structural abnormalities — Cochrane Database Syst Rev. PMID: 38721874
This is a shared conversation. Sign in to Orris to start your own chat.