Explain 1) bronchiectasis
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Searching Images
bronchiectasis CT scan signet ring tram track
Reading File
Loading Image
Loading Image
I now have comprehensive content from Harrison's, Robbins, Murray & Nadel, and Grainger & Allison. Let me compile a thorough answer.
Bronchiectasis
Definition
Bronchiectasis is the permanent, irreversible dilation of bronchi and bronchioles caused by destruction of smooth muscle, elastic tissue, and cartilage in the airway walls. It is never a primary disease — it always arises secondary to persistent infection, obstruction, or other predisposing conditions.
- Robbins & Kumar Basic Pathology, p. 455
- Harrison's Principles of Internal Medicine 22E, p. 2286
Morphological (Radiographic) Classification
Three subtypes reflect increasing severity:
| Type | Description |
|---|---|
| Cylindrical | Uniform, regular dilation; airways fail to taper distally |
| Varicoid | Irregular, non-uniform, somewhat serpiginous dilation (like varicose veins) |
| Saccular / Cystic | Focal cystic or sac-like distortion of distal airways; most severe form |
- Murray & Nadel's Respiratory Medicine, p. 1575
Pathogenesis
The Vicious Cycle Hypothesis
The central mechanism of infectious bronchiectasis:
- Impaired mucociliary clearance (from CF, ciliary dyskinesia, a single severe infection, etc.) → microbial colonization
- Chronic infection (especially Pseudomonas aeruginosa, which colonizes damaged airways and evades host defenses) → persistent inflammation
- Neutrophil-driven damage: neutrophils release elastase, reactive oxygen species, and proteolytic enzymes → airway wall destruction (loss of elastin, smooth muscle, cartilage)
- Destruction → further impaired clearance → more infection → worsening dilation
Three Mechanical Pathways to Dilation
- Bronchial obstruction → mucus stasis → superimposed infection → wall damage
- Direct bronchial wall damage from necrotizing infection/inflammation
- Traction bronchiectasis: parenchymal fibrosis (e.g., IPF, post-radiation) retracts and pulls open adjacent airways
"Two intertwined processes contribute to bronchiectasis: obstruction and chronic infection. Either may be the initiator." — Robbins & Kumar Basic Pathology, p. 455
Etiology & Predisposing Conditions
Postinfectious
- Childhood lower respiratory tract infections (pertussis, measles, adenovirus)
- Granulomatous infections: tuberculosis (a major cause in endemic regions); post-TB bronchiectasis remains an important cause of morbidity worldwide
- Necrotizing pneumonias (S. aureus, Klebsiella spp.)
- Post-SARS-CoV-2 pneumonia (advanced bronchiectasis reported)
Congenital / Hereditary
- Cystic fibrosis: most common cause in developed nations; viscid mucus → obstruction → severe bilateral bronchiectasis (upper lobe predominance)
- Primary ciliary dyskinesia (immobile cilia syndrome): autosomal recessive; inherited ciliary abnormalities → impaired mucociliary clearance → recurrent infections → bronchiectasis; associated with male sterility (Kartagener syndrome = PCD + situs inversus)
- Immunodeficiency states: hypogammaglobulinemia, IgG subclass deficiencies → recurrent bacterial infections
- α₁-antitrypsin deficiency: may be associated with bronchiectasis (especially ZZ genotype)
Immunological / Systemic
- ABPA (allergic bronchopulmonary aspergillosis): immune-mediated; characteristically causes central airway bronchiectasis
- Autoimmune diseases: rheumatoid arthritis, Sjögren's syndrome (immune-mediated wall damage)
Obstructive
- Tumors, foreign bodies, mucus impaction → localized bronchiectasis in the obstructed segment
- COPD and asthma (mild bronchiectasis is common; moderate-to-severe should trigger workup for underlying cause)
Anatomical Distribution Clues to Etiology
| Distribution | Likely Etiology |
|---|---|
| Upper lobe predominance | Cystic fibrosis, post-radiation fibrosis |
| Lower lobe predominance | Recurrent aspiration, immunodeficiency, traction (IPF) |
| Mid-lung fields | NTM infection (MAC), primary ciliary dyskinesia |
| Central airways | ABPA, tracheobronchomegaly (Mounier-Kuhn), Williams-Campbell syndrome |
25–50% of referred cases remain idiopathic after workup. — Harrison's, p. 2286
Epidemiology
- Prevalence in the US is increasing, partly due to wider use of HRCT
- More common in women than men
- Incidence increases with age; NTM-associated bronchiectasis classically affects non-smoking women >50 years
- CF-related bronchiectasis presents in late adolescence/early adulthood
- Higher burden in developing nations (TB, malnutrition, immune dysfunction)
Pathology (Gross & Microscopic)
-
Gross: Usually affects lower lobes bilaterally (most vertical airways); airways dilated up to 4× normal diameter, visible to within 1 cm of pleural surface (normally bronchioles cannot be followed beyond 2–3 cm from pleura); filled with purulent mucus
-
Microscopy: Acute and chronic inflammatory infiltrate within bronchial walls; desquamation of lining epithelium; ulceration; mixed bacterial flora in sputum (Staphylococci, Streptococci, Pseudomonas aeruginosa, H. influenzae). In chronic disease: peribronchial fibrosis, loss of elastin and cartilage. Necrosis may produce abscess cavities.

Clinical Features
- Chronic productive cough — the dominant symptom; copious, thick, mucopurulent, sometimes foul-smelling sputum (can be massive in "wet" bronchiectasis)
- Dyspnea
- Hemoptysis — from mucosal vessel injury; can be massive and life-threatening
- Rhinosinusitis
- Crackles and wheezing on auscultation
- Digital clubbing in some patients
- Acute exacerbations: increased sputum volume and purulence; fever and new infiltrates may be absent
Severe/longstanding disease → obstructive ventilatory defects → hypoxemia → pulmonary hypertension → cor pulmonale
Diagnosis
Chest X-Ray
- Poor sensitivity; may be normal
- "Tram tracks" — parallel opaque lines representing thickened dilated bronchial walls seen longitudinally
- Tubular or ring shadows
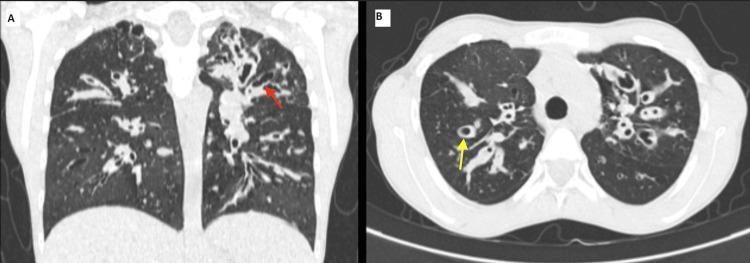
HRCT (Imaging of Choice)
- Signet-ring sign: cross-section of dilated bronchus with diameter ≥1.5× that of the adjacent pulmonary artery (the bronchus is the "ring," the artery is the "signet")
- Tram-track sign: parallel lines representing longitudinal sections of dilated, thickened, non-tapering bronchi
- Bronchial wall thickening, failure of airways to taper, lack of bronchial disappearance at periphery
- Cystic lucencies (in saccular type); air-fluid levels; mucus plugging; tree-in-bud opacities
Pulmonary Function Tests
- Mild-to-moderate airflow obstruction (overlaps with COPD pattern)
- In severe disease: significant obstructive defect
Microbiological
- Sputum cultures: commonly yield H. influenzae, P. aeruginosa, S. aureus, NTM
- BAL if needed (especially for NTM diagnosis)
Workup for Underlying Etiology
- Sweat chloride / CFTR mutation testing (CF)
- Serum immunoglobulins and IgG subclasses (immunodeficiency)
- Ciliary electron microscopy / nasal NO / genetic testing (PCD)
- Serum IgE, Aspergillus-specific IgE/IgG, skin test (ABPA)
- ANA, RF, anti-CCP (autoimmune)
- HIV testing
Treatment
Airway Clearance (Cornerstone)
- Chest physiotherapy: postural drainage, percussion, oscillatory PEP devices, high-frequency chest wall oscillation vest
- Pulmonary rehabilitation and regular exercise
- Bronchodilators and hyperosmolar agents (e.g., hypertonic saline) by aerosol
- DNase (dornase alfa): recommended in CF-related bronchiectasis; not recommended in non-CF bronchiectasis (lacks efficacy, potential harm)
Antibiotics
- Acute exacerbations: targeted antibiotics (commonly H. influenzae and P. aeruginosa coverage) for 7–14 days
- Chronic macrolide therapy (e.g., azithromycin) has been shown to reduce exacerbation frequency in non-CF bronchiectasis
- NTM (MAC): macrolide + rifampin + ethambutol (only after meeting diagnostic criteria — two positive sputum cultures, or one positive BAL, or histopathology + one positive sputum)
Anti-Inflammatory Therapy
- Inhaled glucocorticoids: may reduce dyspnea and sputum; no proven effect on lung function or exacerbation rates; risks of immunosuppression
- Systemic glucocorticoids: important in ABPA and autoimmune-driven bronchiectasis
- Itraconazole: prolonged course for ABPA
- Neutrophil elastase inhibitors: emerging therapy (phase 2 data showing improved outcomes)
Surgery
- Resection of a focal area of disease in refractory cases
- Lung transplantation in advanced disease
Complications
| Complication | Notes |
|---|---|
| Massive hemoptysis | Bronchial artery embolization ± surgery; intubation to protect non-bleeding lung |
| Antibiotic resistance | From repeated courses; may require combination antibiotics |
| Cor pulmonale | From chronic hypoxemia in severe disease |
| Brain abscess | Rare with modern treatment |
| Secondary amyloidosis | Now rare |
Prognosis
Outcomes vary widely with underlying etiology, comorbidities, and exacerbation frequency. The frequent exacerbator phenotype is the strongest predictor of future exacerbations. P. aeruginosa colonization and associated exacerbations are linked to worse outcomes. Modern treatment has substantially improved survival and quality of life.
Sources: Robbins & Kumar Basic Pathology (p. 455–456); Harrison's Principles of Internal Medicine 22E (pp. 2285–2288); Murray & Nadel's Textbook of Respiratory Medicine (Chapters 69, 15); Grainger & Allison's Diagnostic Radiology (p. 154–155)
This is a shared conversation. Sign in to Orris to start your own chat.