Residual consequence of flaccid poliomyelitis. Cerebral palsy.
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Finding Sources
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
I now have comprehensive information to provide a thorough response on both topics. Let me compile the answer.
Residual Consequences of Flaccid Poliomyelitis & Cerebral Palsy
Part 1: Residual Consequences of Flaccid (Paralytic) Poliomyelitis
Pathophysiology of Acute Paralytic Polio
Poliovirus is a neurotropic enterovirus that selectively destroys anterior horn cells (lower motor neurons) of the spinal cord, brainstem motor nuclei, and the reticular formation. The virus triggers a cycle of motor neuron loss → muscle denervation → reinnervation by surviving motor neurons → loss of muscle function. At a critical concentration, the virus also spreads to vestibular and brainstem motor nuclei, hypothalamus, thalamus, cerebellum, and the precentral motor cortex.
- Only 1–2% of poliovirus infections result in paralytic disease
- ~2/3 of paralytic cases have residual paresis
- Spinal polio causes asymmetric proximal limb weakness (especially lower extremities), with flaccid muscles, absent tendon reflexes, and fasciculations
- Sensory deficits are characteristically absent on examination, despite pain and transient paresthesias
Immediate Residuals After Acute Infection
| Feature | Detail |
|---|---|
| Flaccid paralysis | Asymmetric, proximal-predominant, lower > upper limbs |
| Muscle wasting | Occurs over weeks after maximal paralysis |
| Areflexia | Deep tendon reflexes absent in affected limbs |
| Muscle atrophy | Permanent in proportion to motor neuron loss |
| Skeletal deformities | Limb shortening, joint contractures, scoliosis (from asymmetric paralysis) |
| Autonomic dysfunction | Sweating disturbances, urinary retention, delayed gastric emptying, constipation |
| Bulbar polio (20%) | Speech, swallowing, facial, and extraocular muscle dysfunction |
| Respiratory | Ventilatory insufficiency if respiratory muscles affected |
Patients deprived of >50% of lower motor neurons in a given muscle group may have no clinically detectable weakness initially — the remaining enlarged motor units compensate. This reserve is exhausted over decades.
Most spinal polio patients show improved motor function within the first year after acute infection, but recovery is incomplete in those with extensive neuron loss.
Post-Polio Syndrome (PPS) — The Long-Term Residual
Post-polio syndrome is the major delayed consequence, expected to affect up to 100,000 of the 250,000 U.S. adults with a history of polio.
Definition: Recurrence of motor symptoms following a latent period of 20–35 years after resolution of the acute illness.
Diagnostic criteria (all required):
- History of acute paralytic poliomyelitis with documented stable motor recovery
- Residual muscle atrophy, weakness, and areflexia with normal sensation in ≥1 limb
- New muscle symptoms/weakness NOT attributable to acute injury, neuropathy, radiculopathy, or other systemic/neurologic/psychiatric illness
Risk factors for earlier onset:
- Advanced age at time of initial infection
- Greater residual motor disability after acute illness
- Residual bulbar or respiratory signs
- Recent injuries requiring limb immobilization
Clinical features:
- New muscle fatigue, weakness, and atrophy — in both previously affected and previously unaffected muscles
- Joint pain; worsening skeletal deformities
- Fasciculations and paresthesias in affected groups
- New bulbar symptoms: laryngeal weakness → progressive dyspnea, dysphagia, hoarseness
- New respiratory difficulties and sleep-disordered breathing (central hypoventilation, sleep apnea)
- Restless leg syndrome; abnormal sleep movements
Mechanism: Dysfunction of individual nerve axons in surviving (but overworked) motor neurons — chronic metabolic stress on reinnervated motor units enlarges over decades until they fail.
Treatment:
- Primarily symptomatic: analgesics, anti-inflammatory medications
- Muscle training and daily exercise benefit most patients
- Lamotrigine — used adjunctively with exercise to reduce pain and fatigue and improve quality of life
- Sleep apnea/hypoventilation: non-invasive ventilatory support (BiPAP/NIV)
- Post-polio syndrome is a diagnosis of exclusion
Part 2: Cerebral Palsy (CP)
Definition
Cerebral palsy is a nonprogressive neuromuscular disorder caused by injury to the immature brain before age 2 years (or before/during birth in the congenital form). Although the underlying brain injury is static, its functional manifestations evolve over time as the nervous system matures.
Epidemiology
- Most common neuromotor disorder of childhood: ~2 per 1000 live births
- Prevalence is much higher in very premature infants: 40–60 per 1000 in those <32 weeks' gestation
- Adults with CP outnumber children with CP (3:1 ratio in some countries)
- Life expectancy correlates with severity: immobile, tube-fed 15-year-olds have a mean additional life expectancy of 13 years; ambulatory, self-feeding 15-year-olds — 55 years
Etiology
Congenital CP (85–90%) — injury before or during birth:
- Prematurity (most common) — periventricular leukomalacia on MRI
- Periventricular/intraventricular hemorrhage
- Intrauterine hypoxic-ischemic encephalopathy
- TORCH infections (toxoplasmosis, rubella, CMV, herpes)
- Anoxic brain injury
- Multiple births
Acquired CP (~10–15%) — injury after 1 month:
- Perinatal stroke (ischemic, hemorrhagic, or thromboembolic) — most common acquired cause
- Meningitis or encephalitis in infancy
- Abusive head trauma / motor vehicle accidents
Classification
By Motor Type:
| Type | Proportion | Features |
|---|---|---|
| Spastic | ~80% | Upper motor neuron injury; hyperreflexia, spasticity, weakness |
| Dyskinetic | ~15% | Basal ganglia/extrapyramidal; dystonia and/or choreoathetosis |
| Ataxic | ~5% | Cerebellar injury; ataxia, balance impairment |
Spastic CP — Topographic Subtypes:
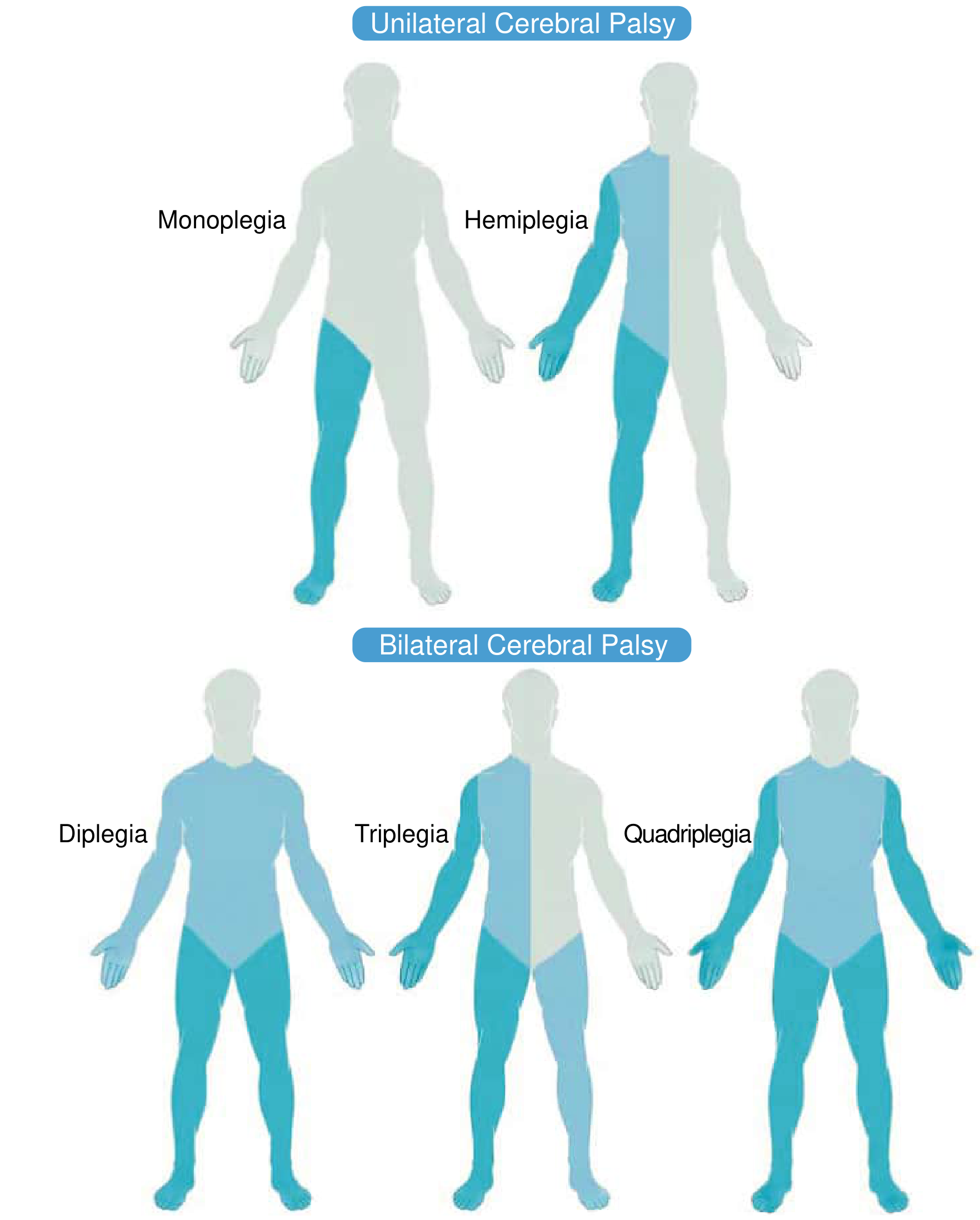
- Hemiplegia — one-sided; arm and leg on same side
- Diplegia — bilateral; predominantly lower extremities
- Triplegia — both lower extremities + one arm
- Quadriplegia — all four extremities involved
By Severity (Gross Motor Function Classification System — GMFCS):
Levels I–V, based on usual motor function across settings (home, school, community), ranging from independent ambulation (I) to complete dependence (V).
Clinical Manifestations
The fundamental lesion is an upper motor neuron disorder producing a mixture of:
- Muscle weakness and spasticity → dynamic joint deformities initially
- Persistent spasticity → contractures, bony deformity, joint subluxation/dislocation
- MRI: commonly periventricular leukomalacia (PVL)
Associated impairments (comorbidities):
- Epilepsy (seizure management is lifelong; often less severe in adulthood)
- Cognitive impairment
- Speech/language disorders
- Dysphagia and feeding difficulties
- Visual and hearing impairments
- Bladder dysfunction
Management
Tone management (spastic CP):
- Botulinum toxin A (BoNT-A) — intramuscular chemodenervation; first-line focal treatment
- Oral baclofen — muscle relaxant for spasticity; also used in dyskinesia
- Benzodiazepines (diazepam) — spasticity and dystonia
- Intrathecal baclofen (ITB) — continuous infusion; especially for severe spasticity ± combined dystonia; higher catheter placement improves effect
- Selective dorsal rhizotomy (SDR) — surgical reduction of spasticity
Dyskinetic CP (dystonia):
- Oral: baclofen, benzodiazepines, trihexyphenidyl, clonidine, gabapentin
- BoNT-A for targeted chemodenervation
- ITB (higher catheter placement)
- Deep brain stimulation (DBS) — globus pallidus or subthalamic nucleus; for medically refractory dystonia; effect takes months; consider over DBS if severe combined spasticity + dystonia
Orthopedic interventions:
- Muscle-tendon lengthening for contractures (rarely before age 6)
- Derotational osteotomies for femoral/tibial torsion (ideally 6–12 years)
- Hip surveillance — risk of displacement increases with GMFCS level
- Spinal monitoring for neuromuscular scoliosis
- Bracing, stretching, and exercise for contracture prevention
Diagnostic Approach
An MRI finding that explains the presentation (e.g., periventricular leukomalacia in diplegia, perinatal stroke in hemiplegia) with a consistent history and clinical picture may not require further workup. Red flags prompting genetic evaluation include: developmental regression, progressive worsening, normal MRI, prominent extrapyramidal features without metabolic explanation, or family history of CP/consanguinity.
Key Contrasts: Polio Residual vs. Cerebral Palsy
| Feature | Polio Residual / PPS | Cerebral Palsy |
|---|---|---|
| Neuron type affected | Lower motor neuron (anterior horn) | Upper motor neuron (developing brain) |
| Tone | Flaccid, hypotonic | Spastic (80%), dystonic (15%), ataxic (5%) |
| Reflexes | Absent (areflexia) | Hyperreflexic (spastic type) |
| Onset | Acute illness then stable → late decline (PPS) | Perinatal/neonatal brain injury |
| Sensory loss | Absent | Absent in motor forms |
| Progression | Static residual → decades later PPS | Injury static; manifestations evolve |
| Key late complication | Post-polio syndrome | Contractures, scoliosis, hip dislocation |
Sources: Tintinalli's Emergency Medicine, 10e; Bradley and Daroff's Neurology in Clinical Practice; Miller's Review of Orthopaedics, 9e; Fishman's Pulmonary Diseases and Disorders.
This is a shared conversation. Sign in to Orris to start your own chat.