Epidemiology and prevention and control of malaria
Reading File
Finding Sources
Searching PubMed
"malaria"[MeSH Terms] AND "prevention AND control"[MeSH Terms]
Searching the Web
malaria epidemiology prevention control 2025 WHO
Finding Sources
Searching PubMed
"malaria"[MeSH Terms] AND "epidemiology"[MeSH Terms]
Reading Web Page
malaria epidemiology global burden prevention control 2025
Reading File
Reading File
Finding Sources
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Reading File
Finding Sources
Loading Image
Reading File
Reading File
I now have comprehensive, authoritative content. Let me compose the full response.
Malaria: Epidemiology, Prevention, and Control
1. Global Burden and Epidemiology
Malaria remains one of the most serious infectious diseases in the world. WHO estimates >200 million cases and >600,000 deaths annually, with the vast majority in sub-Saharan Africa. Nigeria (31%) and Democratic Republic of Congo (12%) alone account for nearly half of all global deaths. Children under 5 and pregnant women bear the greatest burden.
Key epidemiological facts (WHO World Malaria Report 2025):
- 2024 global malaria funding of US$3.9 billion fell far short of the Global Technical Strategy target of US$9.3 billion
- Progress in malaria mortality reduction between 2000–2015 has reversed since then; estimated case numbers are rising
- An increasing number of low-transmission countries are now targeting elimination — but this is threatened by drug and insecticide resistance
- ~54 million children received seasonal malaria chemoprevention (SMC) across 19 countries in 2024
"Malaria remains today, as it has been for centuries, a heavy burden on tropical communities, a threat to nonendemic countries, and a danger to travelers." — Harrison's Principles of Internal Medicine 22E (2025)
2. Causative Agents and Transmission
Five Plasmodium species infect humans:
| Species | Geographic Distribution | Clinical Notes |
|---|---|---|
| P. falciparum | Sub-Saharan Africa, Hispaniola, PNG | Most lethal; cerebral malaria; drug resistance |
| P. vivax | Indian subcontinent, Central America, S/SE Asia | Relapses (hypnozoites); chloroquine-resistant forms emerging |
| P. ovale | Predominantly West Africa | Relapses (hypnozoites) |
| P. malariae | Wide but less common distribution | Recrudescence; quartan fever |
| P. knowlesi | SE Asia (Borneo, Malaysia, Philippines) | Zoonotic (simian); misidentified as P. malariae; can be fatal |
Transmission: Bite of the female Anopheles mosquito (primarily dusk to dawn). Humans are the only natural reservoir. Uncommon routes: congenital transmission, blood transfusion, shared needles.
Half the world's population lives in areas where transmission occurs. — Red Book 2021, Report of the Committee on Infectious Diseases
3. Life Cycle
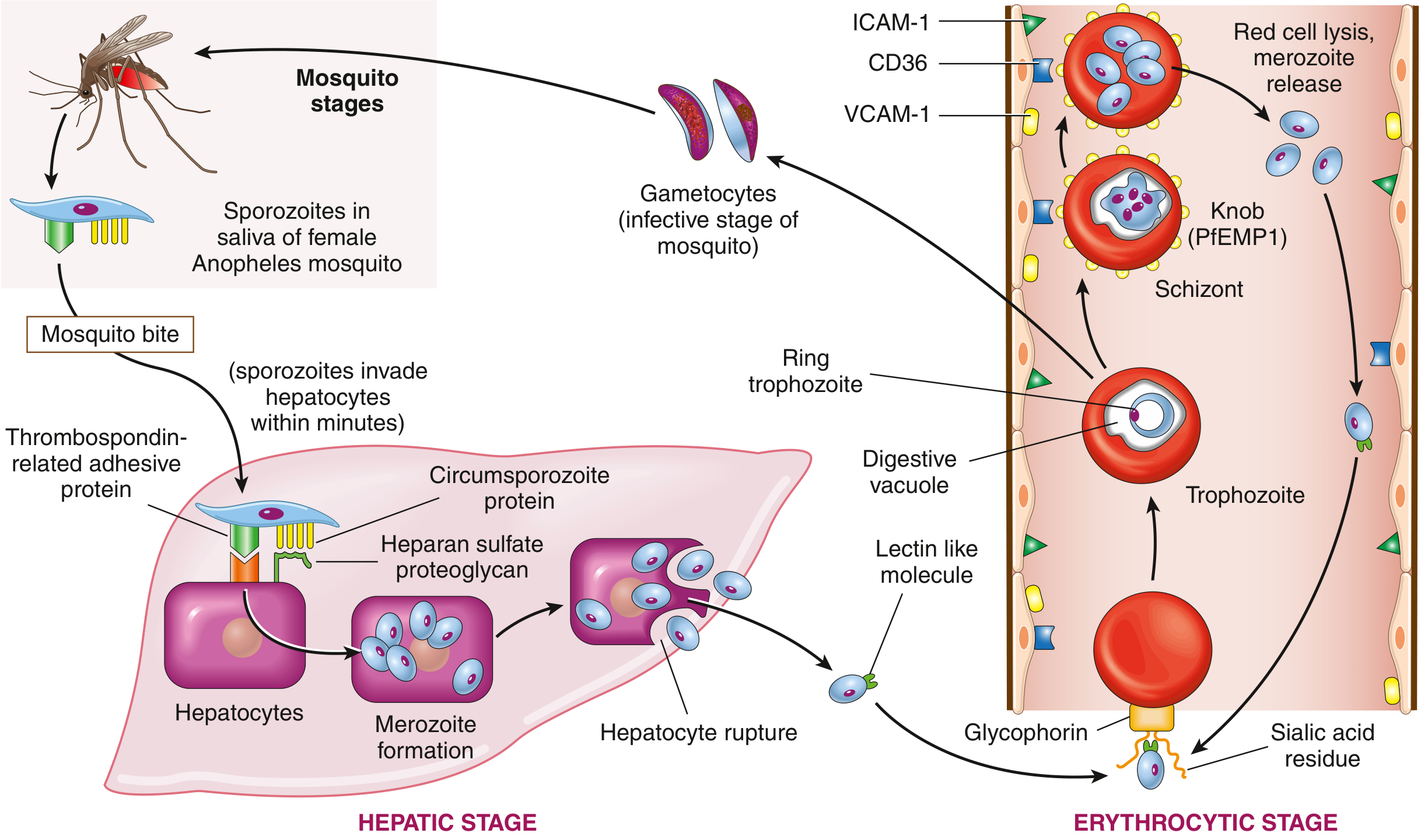
FIG: Life cycle of P. falciparum — Robbins & Kumar Basic Pathology
Key stages:
- Sporozoites injected into bloodstream during mosquito bite → travel to liver
- Hepatic (pre-erythrocytic) stage: Sporozoites bind to hepatocyte proteoglycans via thrombospondin-related adhesive protein and circumsporozoite protein → differentiate into merozoites (clinically silent; 1–4 weeks)
- Erythrocytic stage: Merozoites bind glycophorin (via lectin-like molecule) → enter RBCs → become ring trophozoites → schizonts → merozoites → lysis of RBC → clinical disease
- P. falciparum schizonts express PfEMP1 on RBC surface (knobs) → bind ICAM-1, VCAM-1, CD36 on microvascular endothelium → sequestration in capillary beds → cerebral and organ-specific pathology
- Some trophozoites → gametocytes → taken up by mosquito → sexual reproduction → new sporozoites
Relapses occur in P. vivax and P. ovale due to dormant hypnozoites in the liver. P. falciparum and P. malariae show recrudescence (persistent low-density parasitemia).
4. High-Risk Groups
- Children <5 years — most deaths
- Pregnant women (especially primigravidae) — risk of severe disease, spontaneous abortion, stillbirth, low birth weight
- Non-immune travelers to endemic areas — most fatalities in developed world are from delayed diagnosis
- Immunocompromised persons (including HIV+)
- Blood transfusion recipients / IV drug users (uncommon)
5. Clinical Features (Brief)
- Fever — classically periodic: daily (P. knowlesi), every 48 h (P. falciparum, vivax, ovale), every 72 h (P. malariae) — though unreliable in practice
- Severe malaria (mainly P. falciparum): cerebral malaria (coma), severe anemia, respiratory distress/ARDS, renal failure, thrombocytopenia, hypoglycemia, shock, >5% parasitemia
- Malaria has driven profound selective pressure on human genetics: sickle cell trait, G6PD deficiency, α-thalassemia, and pyruvate kinase deficiency are protective against severe malaria
6. Drug Resistance — Major Challenge
- Chloroquine resistance in P. falciparum spread globally from the 1960s; chloroquine-resistant P. vivax now reported in Indonesia, PNG, Myanmar, India, Guyana
- Artemisinin resistance (P. falciparum) emerged in SE Asia (late 2000s), now prevalent throughout the Greater Mekong Subregion, and has emerged and spread in East Africa — a critical threat
- Key resistance markers: Pfkelch13 mutations (artemisinin), Pfcrt (chloroquine), Pfmdr1 amplification (mefloquine)
- Falsified/substandard antimalarials in Asian and African markets are a major contributor to treatment failures
7. Diagnosis
- Gold standard: Thick and thin blood film microscopy — thick film for sensitivity (detects low parasitemia); thin film for species identification and parasite density
- If negative but malaria suspected: repeat every 12–24 hours for 72 hours (minimum 3 smears)
- Rapid Diagnostic Tests (RDTs): Detect specific malaria antigens; FDA-approved tests available; must be confirmed by microscopy
- PCR: Reference laboratory test; most useful for species confirmation; too slow for initial treatment decisions
- Serology: Generally not useful clinically; reserved for epidemiologic surveys
8. Treatment
WHO recommends Artemisinin-Based Combination Therapy (ACT) as first-line for uncomplicated P. falciparum:
- ACT = artemisinin derivative (artesunate, artemether, or dihydroartemisinin) + a partner drug in fixed-dose coformulation
- For P. knowlesi: ACT recommended
- For P. vivax, ovale, malariae: chloroquine (where sensitive) or ACT; add primaquine (after ruling out G6PD deficiency) to eliminate hypnozoites and prevent relapse
Severe malaria: IV artesunate is the drug of choice — reduces mortality by 35% vs. quinine in Asian adults and by 22.5% in African children (large RCTs). Patients require ICU care.
In areas with artemisinin resistance: Triple ACTs (two slowly eliminated partner drugs) have been proposed to provide mutual protection.
9. Prevention and Control
A. Vector Control
| Measure | Details |
|---|---|
| Insecticide-Treated Nets (ITNs/LLINs) | Long-lasting insecticidal nets with pyrethroid — reduce incidence where vectors bite indoors at night; cornerstone of prevention |
| Indoor Residual Spraying (IRS) | Spraying insecticide on interior walls; key component of integrated vector control programs |
| Combination insecticides | Neonicotinoid–pyrethroid combination developed to delay emergence of insecticide resistance |
| Larval source management | Environmental modification to reduce mosquito breeding sites |
B. Personal Protection (Travelers and Endemic Populations)
- Avoid peak mosquito biting hours (dusk to dawn)
- Insect repellents: DEET 20–50% (preferred); 7% picaridin if DEET unacceptable; apply to all exposed skin
- Permethrin-treated clothing for added protection
- ITNs: Sleep under insecticide-impregnated bed nets if not in well-screened accommodation
- Protective clothing: Long-sleeved shirts and long trousers
C. Chemoprophylaxis (Travelers to Endemic Areas)
Available drugs (per Harrison's 22E and Red Book):
| Drug | Indication | Timing |
|---|---|---|
| Atovaquone-proguanil (Malarone) | Chloroquine- and mefloquine-resistant areas | Start 1–2 days before; take daily; stop 7 days after return |
| Doxycycline | Chloroquine- and mefloquine-resistant areas | Start 1–2 days before; daily; stop 4 weeks after return |
| Mefloquine | Mefloquine-sensitive areas; preferred in pregnancy (2nd/3rd trimester) | Start 2 weeks before; weekly; stop 4 weeks after |
| Chloroquine/Hydroxychloroquine | Only chloroquine-sensitive areas (few remain) | Start 1–2 weeks before; weekly; stop 4 weeks after |
| Primaquine | Terminal prophylaxis for P. vivax/ovale; requires G6PD testing | Start 1 day before; daily; stop 7 days after |
| Tafenoquine | P. vivax/ovale prophylaxis; single-dose anti-relapse; requires G6PD testing | — |
- Regimen choice depends on: travel itinerary (region, drug resistance pattern), medical history, pregnancy status, potential drug interactions
- Chemoprophylaxis is never 100% reliable — always consider malaria in febrile returned travelers even if taking prophylaxis
D. Preventive Treatment in Special Populations
Intermittent Preventive Treatment in Pregnancy (IPTp):
- Sulfadoxine-pyrimethamine (SP) at each antenatal visit (maximum monthly) in 2nd and 3rd trimesters in high-transmission areas
- Coverage target: 80%; actual 2024 coverage: only 45% of eligible women received ≥3 doses
- Women on TMP-SMX for HIV prophylaxis should not receive SP concurrently
Seasonal Malaria Chemoprevention (SMC):
- Amodiaquine + SP given to children <5 years during high-transmission season in the Sahel
- ~54 million children reached across 19 countries in 2024
Intermittent Preventive Treatment in Infants (IPTi) — SP given with routine vaccinations
E. Vaccines
| Vaccine | Status |
|---|---|
| RTS,S/AS01 (Mosquirix) | WHO-recommended for children in sub-Saharan Africa; first malaria vaccine licensed; moderate (≈30–40%) efficacy |
| R21/Matrix-M | Second vaccine approved (2023); higher efficacy (~75% in phase III trials); WHO-recommended 2023 |
| Irradiated sporozoite vaccine (PfSPZ) | Late-stage development; sterile immunity in trials |
As of 2025, no malaria vaccine is approved for use in travelers; vaccines are intended for endemic country deployment.
10. Elimination and Eradication Strategy (WHO GTS)
The WHO Global Technical Strategy for Malaria 2016–2030 targets:
- 90% reduction in malaria case incidence and mortality rates vs. 2015
- Elimination in ≥35 countries by 2030
- Universal access to malaria prevention, diagnosis, and treatment
Key threats to elimination:
- Antimalarial drug resistance (especially artemisinin resistance spreading to Africa)
- Insecticide resistance in Anopheles vectors
- Funding gaps (2024: $3.9B actual vs. $9.3B needed)
- Climate change — may expand the geographic range of Anopheles mosquitoes
- Inadequate health system infrastructure in high-burden countries
Sources
- Harrison's Principles of Internal Medicine, 22nd Edition (2025, McGraw Hill) — Chapters 130, 231
- Red Book 2021: Report of the Committee on Infectious Diseases (AAP)
- Robbins & Kumar Basic Pathology
- Murray & Nadel's Textbook of Respiratory Medicine
- WHO World Malaria Report 2025
- Medicines for Malaria Venture — Malaria Facts & Statistics 2025
This is a shared conversation. Sign in to Orris to start your own chat.