Bence jones protein what's it describe with illustrative diagrams for 10 marks question for orthopaedic exam
Reading File
Finding Sources
Searching Images
Bence Jones protein multiple myeloma immunoglobulin light chain
Searching the Web
Bence Jones protein multiple myeloma light chain pathophysiology orthopaedic exam
Reading File
Reading File
Finding Sources
Loading Image
Loading Image
Reading File
Reading File
Loading Image
Searching Images
multiple myeloma lytic bone lesions skull X-ray plasma cell
Loading Image
Reading File
Loading Image
Loading Image
Reading File
Loading Image
I now have all the images and textbook content needed. Here is the complete 10-mark exam answer:
Bence Jones Protein — 10-Mark Orthopaedic Exam Answer
1. Definition
Bence Jones (BJ) protein refers to free monoclonal immunoglobulin light chains (either κ or λ type) produced in excess by a clone of neoplastic plasma cells. They are present in blood and are filtered by the kidney to appear in urine. Under normal circumstances, no Bence Jones protein is detectable in urine.
Named after Henry Bence Jones (1847), who first described the thermally precipitable urinary protein.
2. Cell of Origin & Molecular Structure
Normal plasma cells produce complete immunoglobulins (heavy + light chains). In malignant plasma cell disorders (particularly multiple myeloma), the neoplastic clone secretes:
- Intact monoclonal immunoglobulin → detected as an M-spike on serum protein electrophoresis (SPEP)
- Excess free light chains → BJ proteins, too small (MW ~22,000–44,000 Da) to be retained by glomeruli → filtered freely into urine
Complete Immunoglobulin:
[Heavy chain] — [Heavy chain]
| |
[Light chain] [Light chain]
Bence Jones Protein = Free light chain only
κ (kappa) or λ (lambda) monomer / dimer
Light chains are either κ or λ; in myeloma they are monoclonal (only one type), unlike the normal polyclonal mixture.
3. Disease Context — Multiple Myeloma (Orthopaedic Relevance)
Multiple myeloma is a malignant proliferation of plasma cells in bone marrow causing:
| Feature | Details |
|---|---|
| Bone pain | Most common presenting symptom; vertebrae, ribs, skull, pelvis, femur |
| Lytic bone lesions | "Punched-out" defects on X-ray, 1–4 cm, no sclerotic rim |
| Pathological fractures | Most common in vertebrae; cause of acute back pain in elderly |
| Hypercalcaemia | Due to osteoclast activation → confusion, polyuria, constipation |
| Anaemia | Marrow replacement → normocytic normochromic anaemia |
| Renal failure | BJ protein nephrotoxicity → myeloma kidney |
| Infections | Suppression of normal immunoglobulins |
Classic "CRAB" Criteria
- C – hyperCalcaemia
- R – Renal failure
- A – Anaemia
- B – Bone lesions
4. Pathogenesis of Bence Jones Proteinuria
Neoplastic plasma cell clone
↓
Excess free light chains produced
↓
Freely filtered at glomerulus (MW < 45,000 Da)
↓
Proximal tubule reabsorption overwhelmed
↓
BJ protein in urine (Bence Jones proteinuria)
Renal toxicity occurs by two mechanisms (Robbins Pathologic Basis of Disease):
- Direct tubular toxicity — certain light chains (esp. λ6, λ3 families) are directly toxic to proximal tubular epithelial cells
- Cast nephropathy — BJ proteins combine with Tamm-Horsfall glycoprotein under acidic conditions → large obstructive casts in distal/collecting tubules → inflammatory reaction → "myeloma kidney"
5. Radiological Illustration — Skull X-Ray
Classic "Punched-out" / "Pepper-pot" Skull
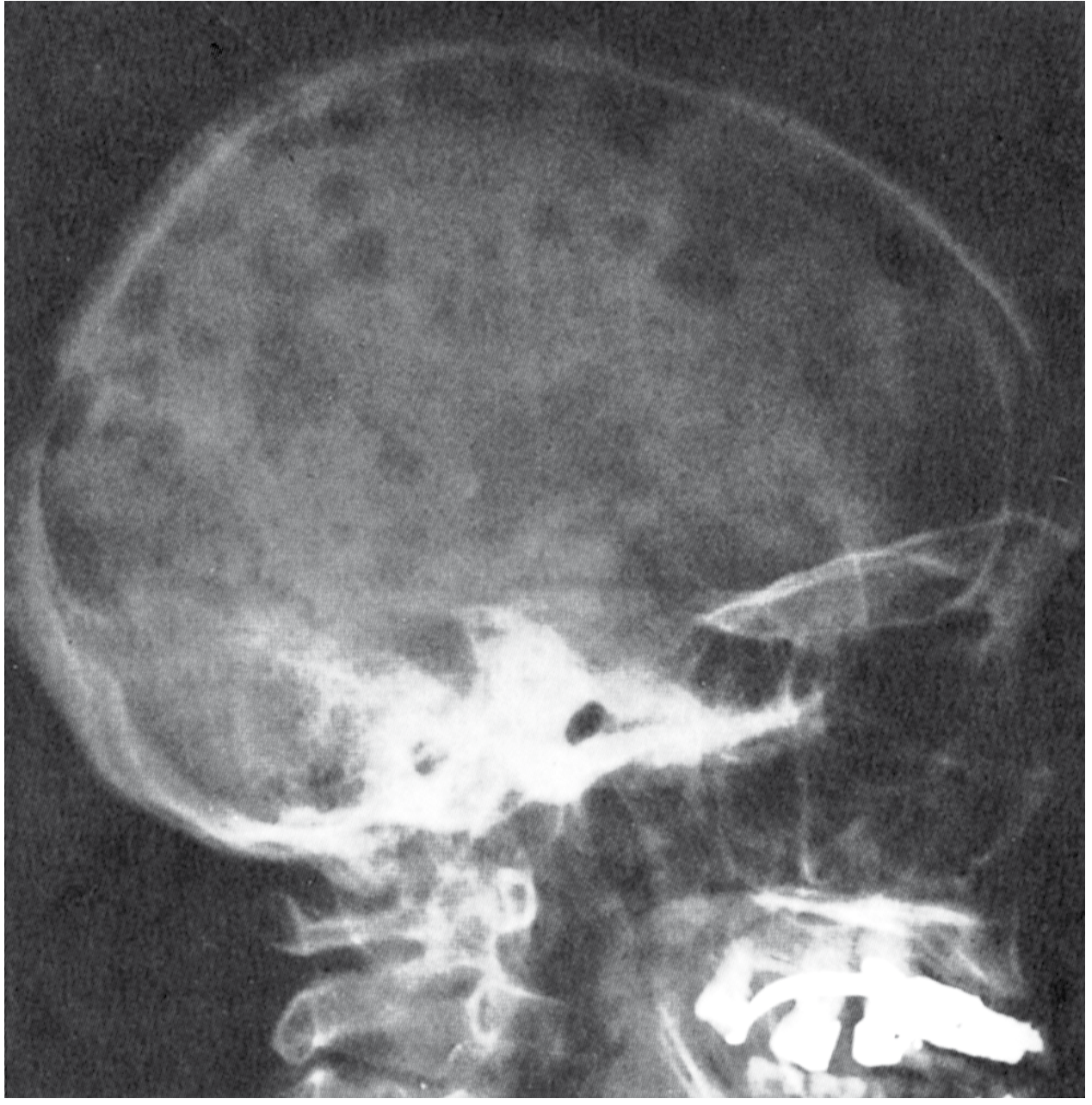
Fig. 1 — Lateral skull radiograph demonstrating multiple sharply demarcated "punched-out" lytic lesions throughout the calvaria. No sclerotic rim. Generalised osteopenia is also present. This is the classical appearance of plasma cell myeloma on plain X-ray.
(Source: Robbins, Cotran & Kumar Pathologic Basis of Disease)
Frontal skull view showing the same appearance:
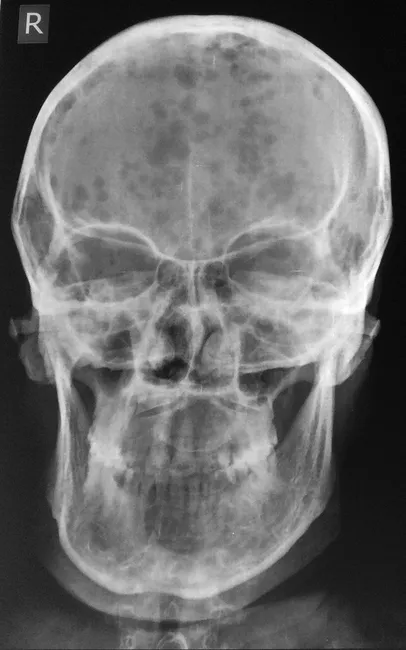
Fig. 2 — AP skull radiograph: multiple discrete, rounded radiolucencies ("punched-out" lesions) scattered across the calvarium without a sclerotic rim. Diffuse osteopenia is also evident.
6. Bone Marrow Histology
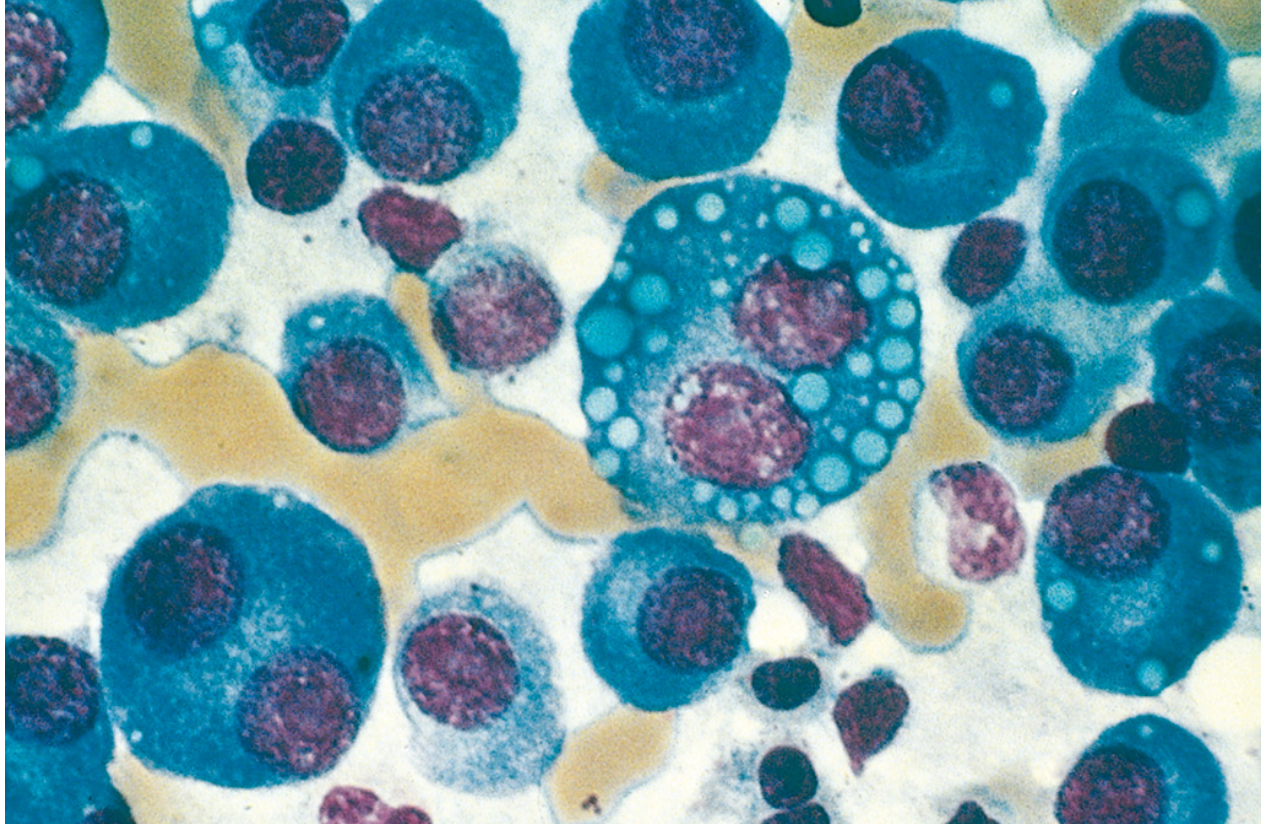
Fig. 3 — Bone marrow aspirate (Wright–Giemsa stain). Normal marrow elements replaced by neoplastic plasma cells showing:
- Eccentric nucleus with clock-face/cartwheel chromatin
- Perinuclear hof (clearing due to prominent Golgi)
- Multinucleated forms (plasmablasts)
- Russell bodies (cytoplasmic globular inclusions of immunoglobulin)
- Mott cells (grape-like cytoplasmic droplets)
(Source: Robbins, Cotran & Kumar Pathologic Basis of Disease)
7. Protein Electrophoresis — Detection of BJ Protein
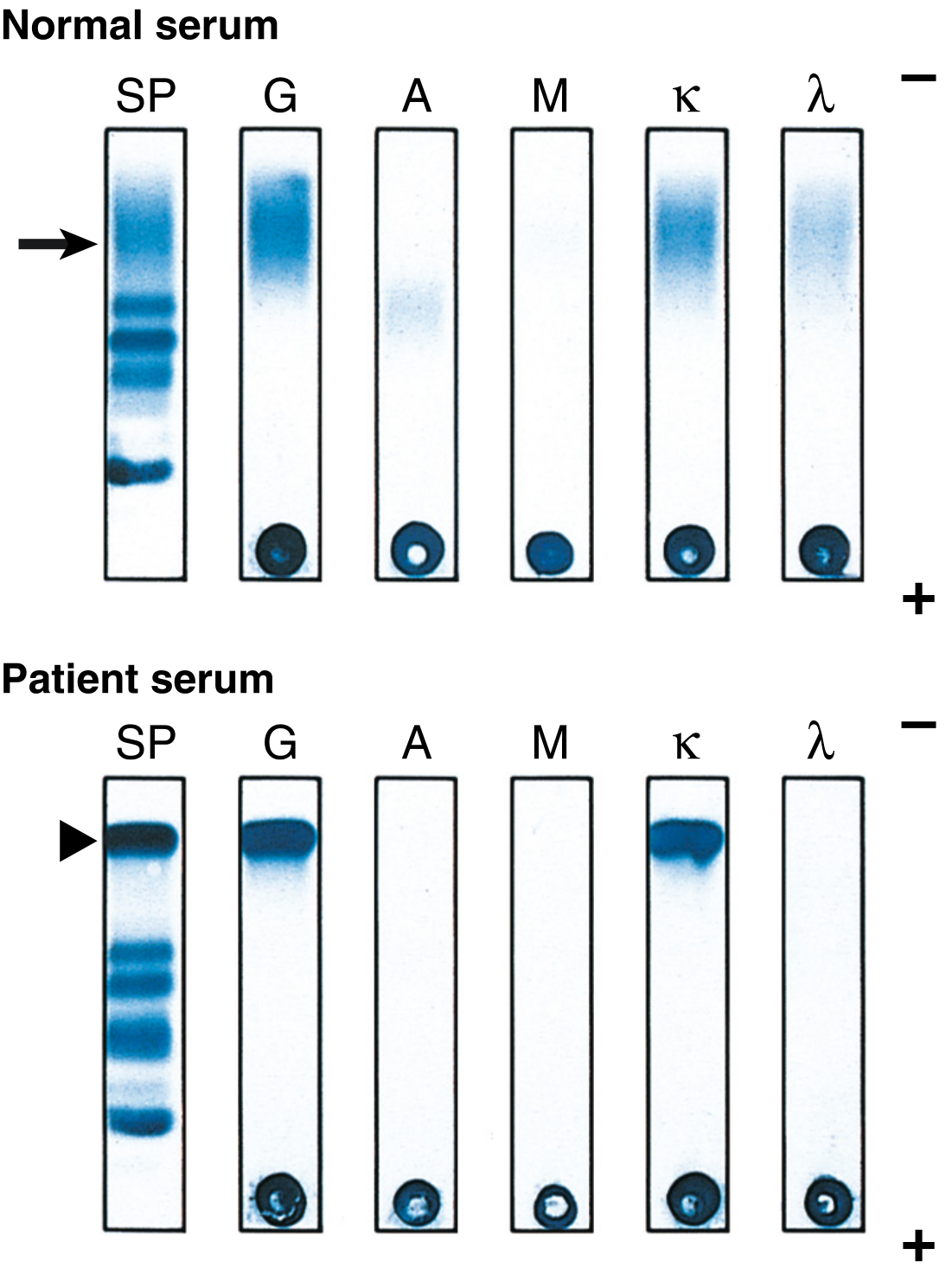
Fig. 4 — Serum protein electrophoresis (SP column) and immunofixation electrophoresis (IFE).
- Normal serum (top): Polyclonal IgG appears as a broad diffuse band in the gamma region (arrow)
- Patient serum (bottom): Single sharp monoclonal spike (arrowhead) in gamma region = M protein; confirmed by IFE as IgG κ — note sharp band crosses with anti-G and anti-κ lanes; polyclonal IgA and λ are suppressed
(Source: Robbins, Cotran & Kumar Pathologic Basis of Disease)
Urine Immunofixation for BJ protein:
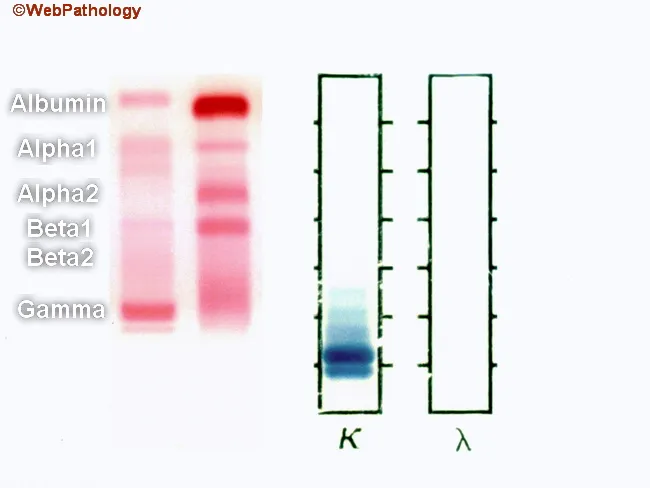
Fig. 5 — Urine protein electrophoresis (UPEP) showing free κ light chains (Bence Jones proteins) in the urine with a positive band in the kappa (κ) column and absent in the lambda (λ) column, confirming monoclonal κ free light chains.
8. Renal Pathology (Myeloma Kidney)
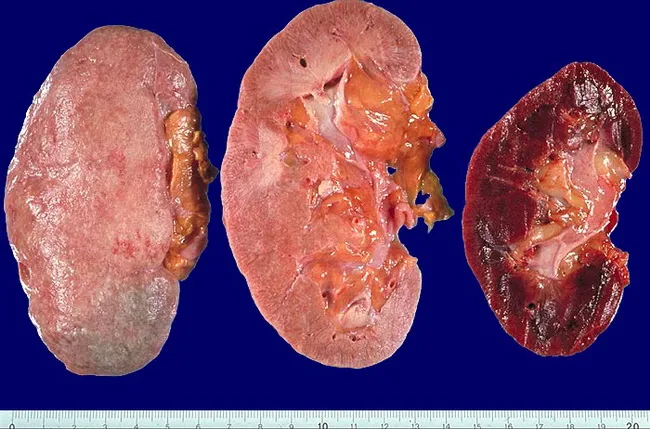
Fig. 6 — Gross photograph: three renal specimens. Left and centre kidneys show pale, scarred cortex with cortical destruction from chronic BJ protein deposition and cast nephropathy. Right kidney is normal for comparison.
Pathological sequence in Myeloma Kidney:
BJ protein filtered → tubular cast formation
↓
Obstruction of distal & collecting tubules
↓
Tubular cell toxicity & necrosis
↓
Inflammatory reaction (giant cell response)
↓
Tubular atrophy & interstitial fibrosis
↓
Progressive renal failure ("Myeloma Kidney")
9. Laboratory Diagnosis — Summary
| Test | Finding |
|---|---|
| Urine BJ protein (heat test) | Precipitates at 45–60°C, redissolves at 100°C |
| Urine protein electrophoresis (UPEP) | Monoclonal light chain band |
| Urine immunofixation electrophoresis (IFE) | Gold standard — identifies κ or λ type |
| Serum free light chain (FLC) assay | Quantitative; κ:λ ratio abnormal |
| Serum protein electrophoresis (SPEP) | M-spike in gamma region |
| 24-hour urine BJ protein | > 6 mg/dL significant; several grams/day in severe cases |
| Bone marrow biopsy | Plasma cells > 10–30% of cellularity |
BJ proteinuria is present in 50–80% of multiple myeloma cases. It may be missed on routine dipstick (which detects albumin, not light chains) — electrophoresis and IFE are essential.
10. Associated Conditions
BJ proteinuria is also found in:
- Macroglobulinaemia (Waldenström)
- Primary (AL) amyloidosis — λ light chains (λ6, λ3 families) deposited as amyloid in tissues
- Malignant lymphomas
- MGUS (Monoclonal Gammopathy of Undetermined Significance) — precursor state
Summary Table for Quick Revision
| Key Point | Detail |
|---|---|
| What is it? | Free monoclonal Ig light chain (κ or λ) in urine |
| Source | Neoplastic plasma cells in bone marrow |
| Key disease | Multiple myeloma (also MGUS, AL amyloidosis, lymphoma) |
| MW | ~22,000–44,000 Da → freely glomerular filtered |
| Orthopaedic significance | Bone pain, pathological fractures, lytic lesions, hypercalcaemia |
| Classic X-ray | Punched-out lesions in skull/vertebrae — no sclerotic rim |
| Renal complication | Myeloma kidney (cast nephropathy) → renal failure |
| Best detection test | Urine immunofixation electrophoresis (IFE) |
| Heat test | Precipitates at 45–60°C, redissolves at 100°C (classic but insensitive) |
| Normal level in urine | None (absence is normal) |
Sources: Robbins, Cotran & Kumar — Pathologic Basis of Disease (10th ed.); Henry's Clinical Diagnosis and Management by Laboratory Methods; Tietz Textbook of Laboratory Medicine (7th ed.)
There's no treatment in this add that too
Finding Sources
Searching PubMed
"multiple myeloma" treatment bortezomib lenalidomide
Searching the Web
multiple myeloma treatment guidelines 2024 bortezomib lenalidomide daratumumab bone disease bisphosphonates
Reading File
Finding Sources
Reading File
Loading Image
Here is the Treatment section to add to your answer:
11. Treatment of Multiple Myeloma (and BJ Proteinuria)
Treatment is divided into: (A) Anti-myeloma systemic therapy, (B) Bone disease management (orthopaedic relevance), and (C) Supportive/renal care.
A. Systemic Anti-Myeloma Therapy
Step 1 — Assess Transplant Eligibility
Newly Diagnosed Multiple Myeloma
↓
┌─────────────────────┐
↓ ↓
Transplant candidate Not transplant candidate
(age <65–70, fit) (elderly, comorbid)
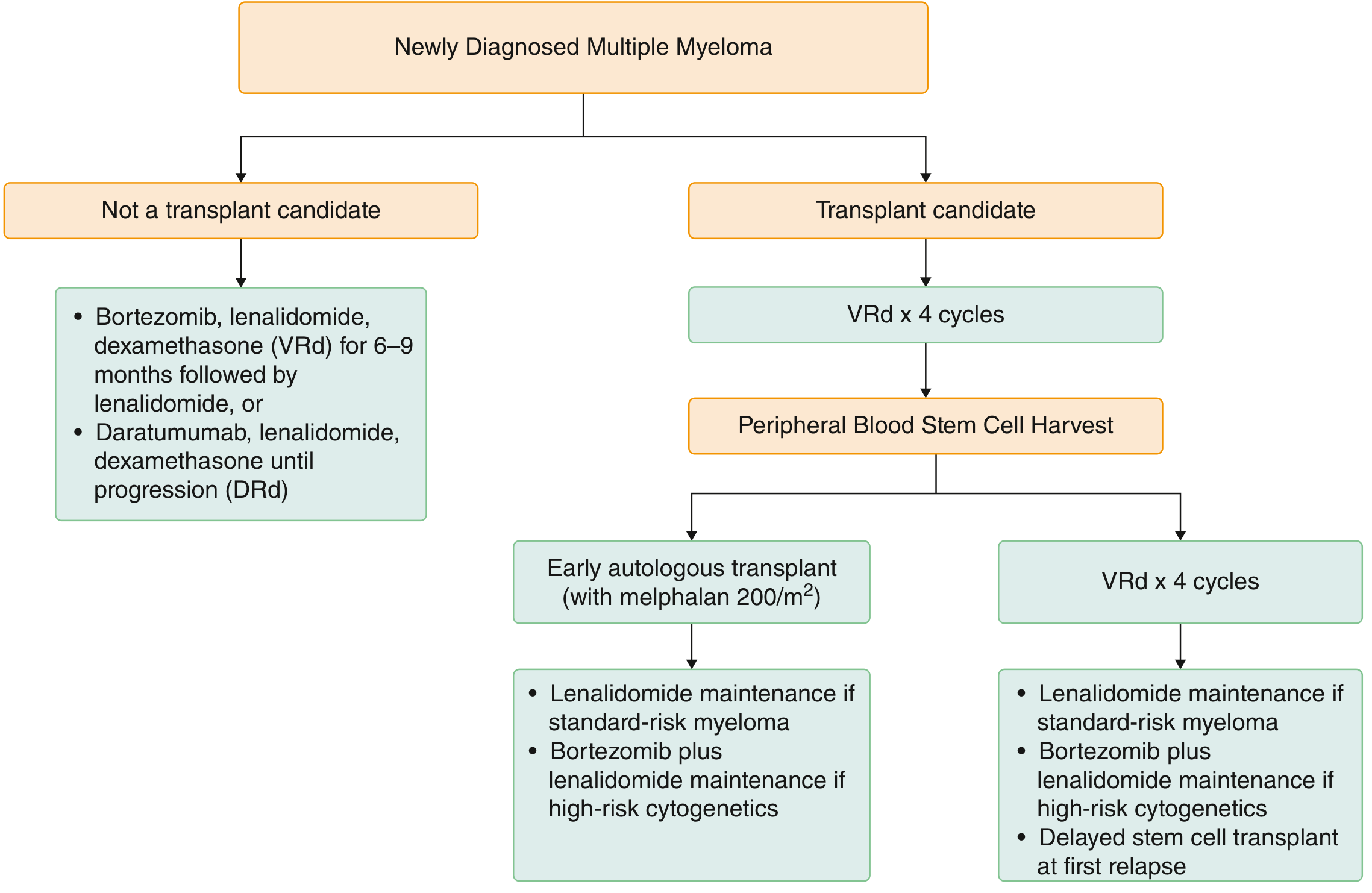
Fig. 7 — Treatment algorithm for newly diagnosed multiple myeloma. Transplant-eligible patients receive VRd induction → stem cell harvest → autologous transplant → maintenance. Non-eligible patients receive VRd or DRd until progression. (Source: Goldman-Cecil Medicine)
Drug Classes Used
| Class | Agents | Mechanism |
|---|---|---|
| Proteasome inhibitors (PI) | Bortezomib (V), Carfilzomib (K) | Block ubiquitin-proteasome pathway → plasma cell apoptosis |
| Immunomodulatory drugs (IMiD) | Lenalidomide (R), Thalidomide | Anti-angiogenic, immune stimulation, plasma cell apoptosis |
| Corticosteroids | Dexamethasone (d) | Anti-inflammatory, direct cytotoxicity to plasma cells |
| Anti-CD38 monoclonal Ab | Daratumumab (D), Isatuximab | Target CD38 on plasma cells → complement/ADCC killing |
| Alkylating agents | Melphalan, Cyclophosphamide (Cy) | DNA crosslinking |
| CAR-T / BCMA-targeted | Idecabtagene vicleucel | Targets BCMA on plasma cells (relapsed/refractory) |
Key Induction Regimens (Table 173-5, Goldman-Cecil Medicine)
| Setting | Regimen | Details |
|---|---|---|
| Transplant eligible | VRd (standard) | Bortezomib 1.3 mg/m² IV + Lenalidomide 25 mg PO + Dexamethasone 40 mg × 3–4 cycles |
| Transplant eligible | DVRd (high-risk) | + Daratumumab 16 mg/kg IV — ASCO 2026 guideline preferred |
| Non-transplant eligible | VRd × 6–9 months then lenalidomide maintenance | Or DRd (Daratumumab + Rd) until progression |
| Conditioning (ASCT) | Melphalan 200 mg/m² | Followed by autologous stem cell infusion |
Maintenance (post-transplant)
- Standard risk: Lenalidomide alone
- High-risk cytogenetics (del17p, t[4;14]): Bortezomib + Lenalidomide
B. Bone Disease Management — Orthopaedic Focus
This is the most directly orthopaedic component of myeloma treatment.
Mechanism of Bone Destruction:
Neoplastic plasma cells
↓ secrete MIP-1α (CCL3), DKK-1, RANKL
↓
↑ Osteoclast activity + ↓ Osteoblast activity
↓
Lytic lesions → Pathological fractures + Hypercalcaemia
1. Bisphosphonates (First-line bone protection)
| Drug | Dose | Route | Frequency |
|---|---|---|---|
| Zoledronic acid (Zometa) | 4 mg | IV infusion over 15 min | Monthly × 2 years, then q3 months |
| Pamidronate | 90 mg | IV infusion over 2 hours | Monthly |
Mechanism: Inhibit farnesyl pyrophosphate synthase → osteoclast apoptosis → prevent bone resorption
Benefits:
- Reduce skeletal-related events (SREs) — fractures, hypercalcaemia, cord compression
- May have direct anti-myeloma activity
- Zoledronic acid preferred (superior to pamidronate in trials)
⚠️ Complication: Osteonecrosis of the jaw (ONJ) — dental review mandatory before starting; avoid invasive dental procedures
2. RANKL inhibitor
- Denosumab (anti-RANKL monoclonal antibody) — alternative to bisphosphonates, especially in renal impairment (does not require dose adjustment)
3. Radiation Therapy
- Local palliative radiotherapy (20–30 Gy) for:
- Painful localised bone lesions unresponsive to systemic therapy
- Impending or actual pathological fracture (post-fixation)
- Spinal cord compression (emergency treatment)
4. Orthopaedic Surgery
- Prophylactic intramedullary nailing — for impending pathological fractures (femur: Mirels score ≥ 9)
- Vertebroplasty / Kyphoplasty — for painful vertebral compression fractures
- Spinal decompression + stabilisation — for cord compression with instability
- Arthroplasty — for destroyed femoral head/acetabulum
5. Hypercalcaemia Management
- IV hydration (normal saline)
- IV bisphosphonates / calcitonin (acute)
- Corticosteroids (address underlying myeloma)
C. Renal (BJ Protein) Specific Management
Since BJ proteinuria is directly nephrotoxic, reducing the plasma cell burden is the primary goal:
| Intervention | Rationale |
|---|---|
| Rapid anti-myeloma therapy (VRd/Bortezomib-based) | Reduces light chain production |
| Vigorous IV hydration | Prevents cast formation, dilutes tubular light chains |
| Urine alkalinisation | BJ protein + Tamm-Horsfall protein bind under acidic conditions; alkaline urine reduces cast formation |
| Avoid NSAIDs, contrast dye, nephrotoxins | Prevent AKI in at-risk kidney |
| Haemodialysis | For established myeloma kidney / ESRD |
| Plasmapheresis | Removes circulating free light chains acutely (in severe light chain crisis) |
D. Supportive Care Summary
| Problem | Management |
|---|---|
| Anaemia | Erythropoiesis-stimulating agents (EPO), transfusions |
| Infections | IVIG replacement; pneumococcal/flu vaccination; prophylactic aciclovir (with bortezomib) |
| DVT (with IMiDs) | Aspirin or LMWH prophylaxis |
| Peripheral neuropathy (bortezomib) | Dose reduction; switch to subcutaneous route |
| Bone pain | Analgesia, bisphosphonates, local radiotherapy |
Treatment Response Monitoring
- Serum M-protein (SPEP) — falls with effective treatment
- 24-hr urine BJ protein — decreases as plasma cell burden reduces
- Serum free light chains — most sensitive marker
- Bone marrow biopsy — assess minimal residual disease (MRD)
- PET-CT / whole-body MRI — assess bone disease and extra-medullary disease
Complete Treatment Summary Diagram
MULTIPLE MYELOMA — TREATMENT OVERVIEW
━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━━
SYSTEMIC THERAPY BONE DISEASE
────────────── ────────────
Induction: Bisphosphonates:
VRd or DVRd Zoledronic acid IV monthly
(Bortezomib + Denosumab (renal impairment)
Lenalidomide +
Dexamethasone Radiotherapy:
± Daratumumab) Local palliative RT
↓
ASCT (if eligible) Surgery:
Melphalan 200 mg/m² IMN for pathological #
↓ Kyphoplasty / vertebroplasty
Maintenance: Spinal stabilisation
Lenalidomide
(± Bortezomib if high-risk)
RENAL PROTECTION SUPPORTIVE
──────────────── ──────────
IV hydration EPO for anaemia
Urine alkalinisation IVIG / antibiotics
Avoid nephrotoxins DVT prophylaxis
Haemodialysis if ESRD Pain management
Sources: Goldman-Cecil Medicine (International Edition); Harrison's Principles of Internal Medicine 22E (2025); Robbins, Cotran & Kumar — Pathologic Basis of Disease; ASCO-Ontario Health Living Guideline 2026 [PMID: 41494138]
This is a shared conversation. Sign in to Orris to start your own chat.