Anatomy histology and physiology of PARATHYROID GLAND
Reading File
Finding Sources
Finding Sources
Finding Sources
Searching Images
parathyroid gland histology anatomy
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Loading Image
Loading Image
Finding Sources
Finding Sources
Reading File
Finding Sources
Reading File
Loading Image
Loading Image
Loading Image
I now have comprehensive material from multiple authoritative textbooks. Here is a thorough, well-illustrated account:
Parathyroid Gland — Anatomy, Histology & Physiology
1. ANATOMY
Gross Features
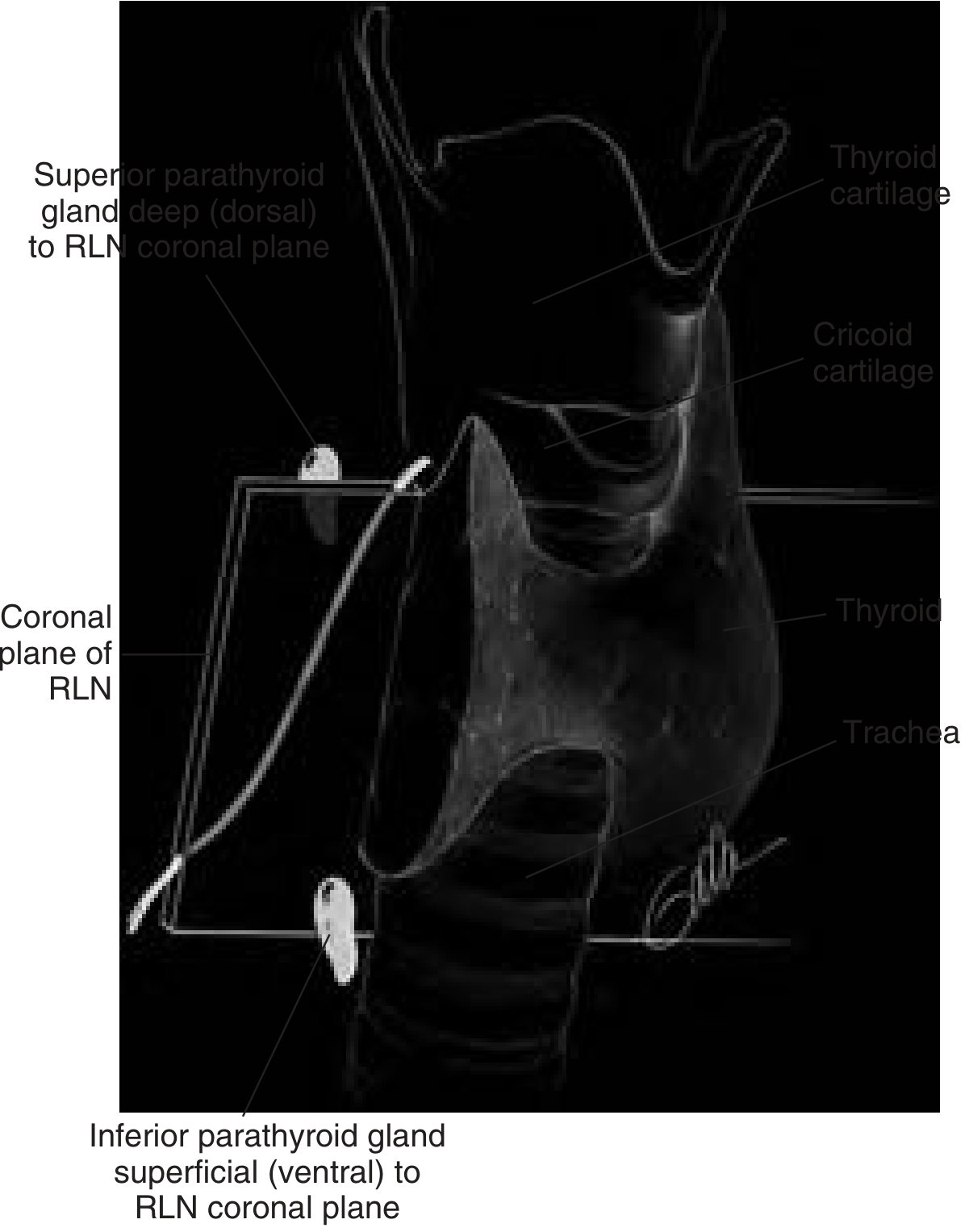
The parathyroid glands are four small, yellowish-brown, ovoid structures averaging 5 × 3 × 1 mm and weighing 30–40 mg each. They are located on the posterior surface of the thyroid gland, each enclosed in its own thin fibrous capsule. Despite their proximity to the thyroid, they are entirely distinct glands with separate blood supplies.
Positional Relationships to the Recurrent Laryngeal Nerve (RLN)
A clinically critical anatomical landmark is the coronal plane defined by the RLN:
- Superior parathyroid glands lie deep (dorsal) to the RLN coronal plane, typically near the posterior aspect of the middle third of the thyroid lobe, close to the cricothyroid junction.
- Inferior parathyroid glands lie superficial (ventral) to the RLN coronal plane, typically near the inferior pole of the thyroid.
Blood Supply
Both superior and inferior glands receive their dominant arterial supply from the inferior thyroid artery, a branch of the thyrocervical trunk. However, abundant anastomoses exist with:
- Superior thyroid artery (dominant supply in ~10% of inferior glands)
- Arteries of the larynx, pharynx, esophagus, and trachea
Venous drainage is into the thyroid venous plexus. This rich anastomotic network is why ligation of the inferior thyroid artery during thyroidectomy does not always cause permanent hypoparathyroidism. Still, transient hypoparathyroidism from ischemia occurs in up to 20% of patients after total thyroidectomy.
— Cummings Otolaryngology Head and Neck Surgery
2. EMBRYOLOGY
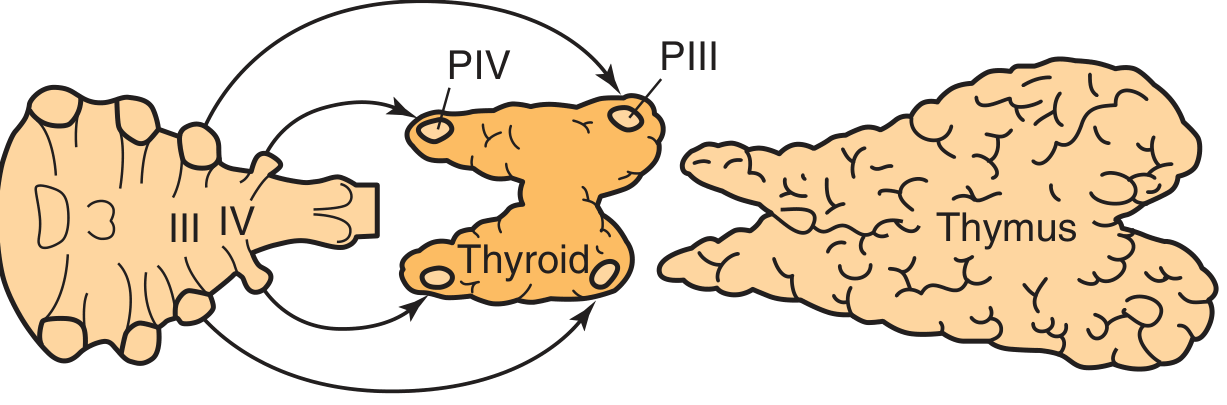
| Gland | Pharyngeal Pouch of Origin | Descent Pattern |
|---|---|---|
| Superior parathyroid (P-IV) | 4th pharyngeal pouch | Short, predictable migration → posterior mid-thyroid |
| Inferior parathyroid (P-III) | 3rd pharyngeal pouch (with thymus) | Long caudal migration with the thymus |
Because the inferior glands travel the longer embryologic path with the thymus, they are more frequently ectopic. Ectopic locations include:
- Anterior superior mediastinum (most common ectopic site; ~1/3 of missed parathyroid tumors)
- Carotid sheath
- Retroesophageal space
- Submandibular region
- Intrathyroidal
The superior glands have a more predictable position, but may descend into the tracheoesophageal groove.
— Cummings Otolaryngology Head and Neck Surgery
3. HISTOLOGY
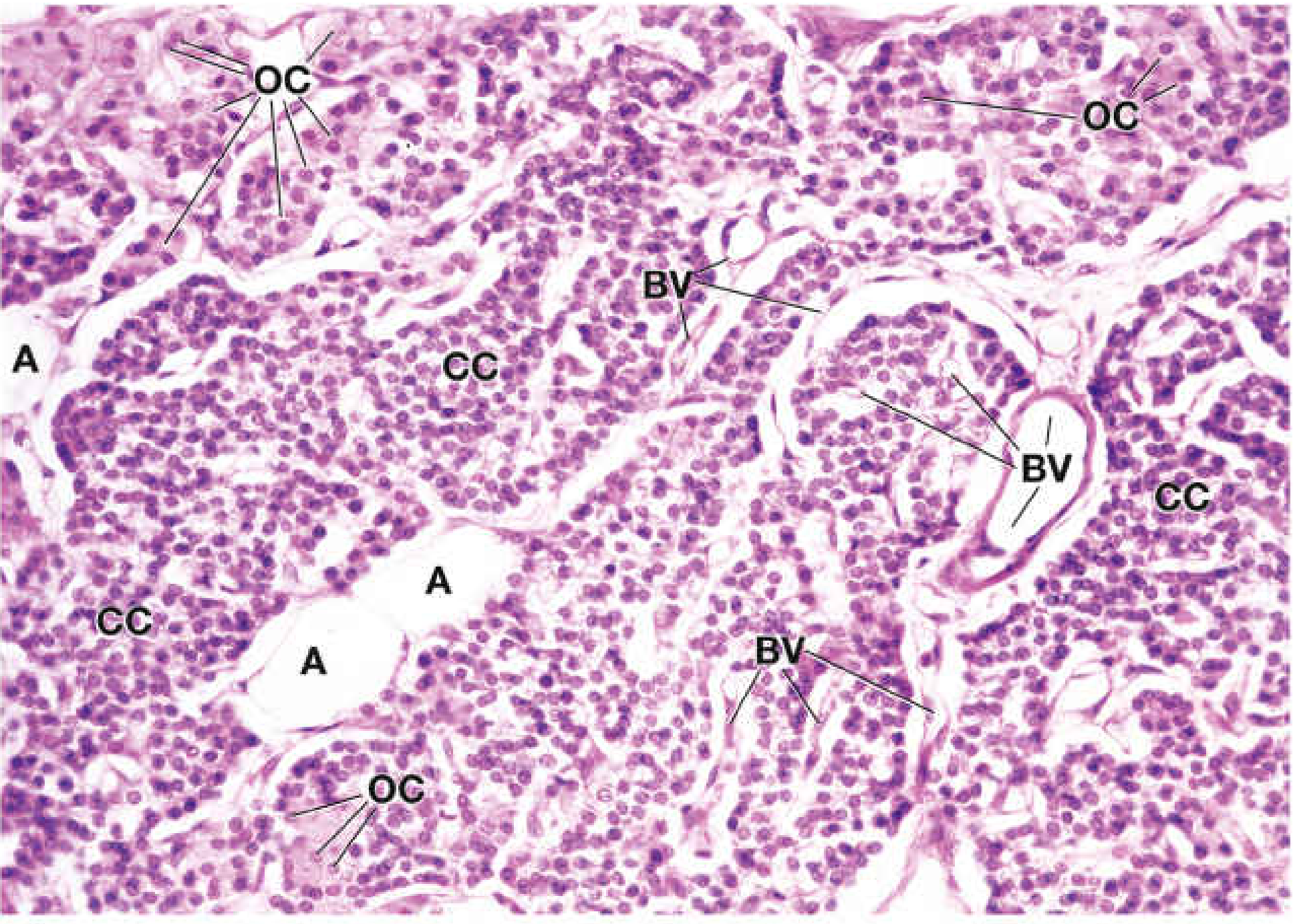
Capsule & Stroma
The gland is enclosed by a thin fibrous capsule from which septa extend inward, carrying blood vessels. The parenchyma is arranged in cords or sheets separated by capillaries and delicate connective tissue septa. Adipose tissue increases progressively from puberty onward, eventually comprising up to 50% of the gland mass in adults (useful to assess "normal" gland weight during surgery).
Cell Types
A. Chief Cells (Principal Cells) — The dominant PTH-secreting cell
- Most numerous cell type
- Small (7–10 µm), polygonal
- Round, centrally placed nucleus with moderate chromatin
- Sparse, pale (slightly amphophilic) cytoplasm with small amounts of glycogen and lipid droplets
- Arranged in cords and nests around capillaries
- Contain secretory granules with PTH in varying stages of synthesis and storage
- In active states: fewer granules, more rER; in resting states: more granules, lipid vacuoles
B. Oxyphil Cells
- Less numerous, appear at end of first decade, increase around puberty, further increase in older individuals
- Larger than chief cells (10–15 µm)
- Smaller, more densely staining nucleus
- Abundant eosinophilic (acidophilic) cytoplasm — due to packed mitochondria
- Arranged in discrete clusters scattered among chief cells; clusters are recognizable even at low magnification due to lower nuclear density per unit area
- Concentrated technetium-99m sestamibi (used in parathyroid scintigraphy)
- Function remains debated; may be transitional forms or inactive chief cells
C. Water-Clear Cells (Wasserhelle cells)
- Rare in normal glands
- Very large cells with clear, glycogen-rich cytoplasm
- Can proliferate in primary hyperplasia ("water-clear cell hyperplasia")
Histology Diagram
— Histology: A Text and Atlas (Ross & Pawlina, 9e); Cummings Otolaryngology
4. PHYSIOLOGY
Parathyroid Hormone (PTH)
PTH is an 84-amino acid polypeptide (MW ~9,500 Da) synthesized and secreted by the chief cells. It is the primary regulator of calcium homeostasis, maintaining serum calcium within the normal range of 8.5–10.5 mg/dL (2.1–2.6 mM).
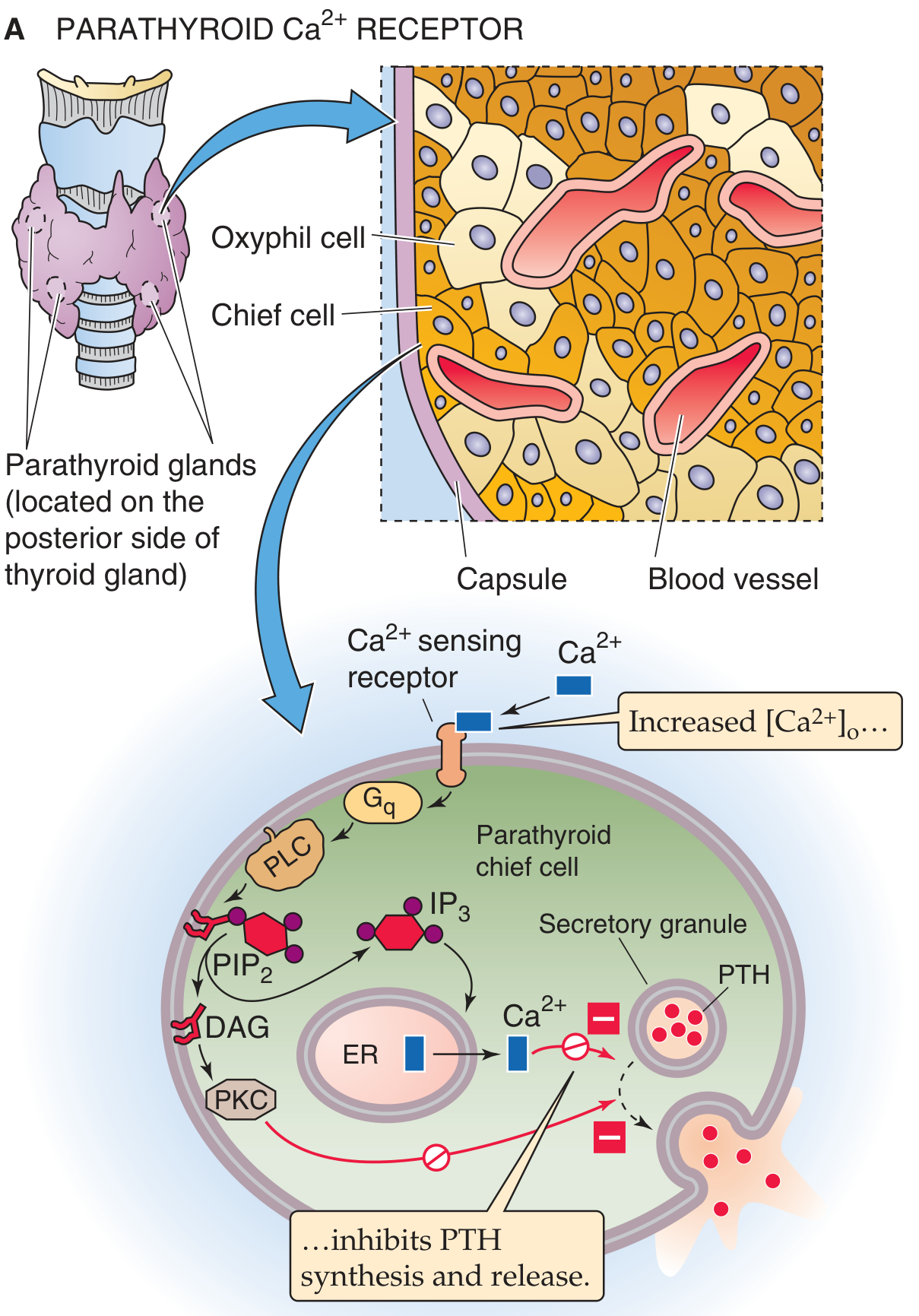
Regulation of PTH Secretion — The Calcium-Sensing Receptor (CaSR)
The key regulator of PTH secretion is the extracellular Ca²⁺ concentration, sensed by the calcium-sensing receptor (CaSR), a class C GPCR on chief cells.
Signaling when [Ca²⁺] is HIGH (inhibits PTH):
Ca²⁺ binds CaSR → couples to Gαq → activates phospholipase C (PLC) → cleaves PIP₂ → produces IP₃ (releases Ca²⁺ from ER) + DAG (activates PKC) → elevated intracellular Ca²⁺ and PKC activity inhibit PTH granule release and synthesis
This is the "paradox": unlike most secretory cells, rising intracellular Ca²⁺ suppresses PTH secretion rather than stimulating it.
When [Ca²⁺] is LOW → CaSR inactive → PTH secreted
The secretory curve is steep — small decreases in free plasma Ca²⁺ produce large increases in PTH output. About 50% of total plasma calcium is free (ionized); this is the fraction sensed by the CaSR.
In Familial Hypocalciuric Hypercalcemia (FHH), inactivating mutations of CaSR shift the set-point rightward — plasma Ca²⁺ must rise higher before PTH is suppressed, resulting in mild hypercalcemia with normal PTH.
Target Organs & Actions of PTH
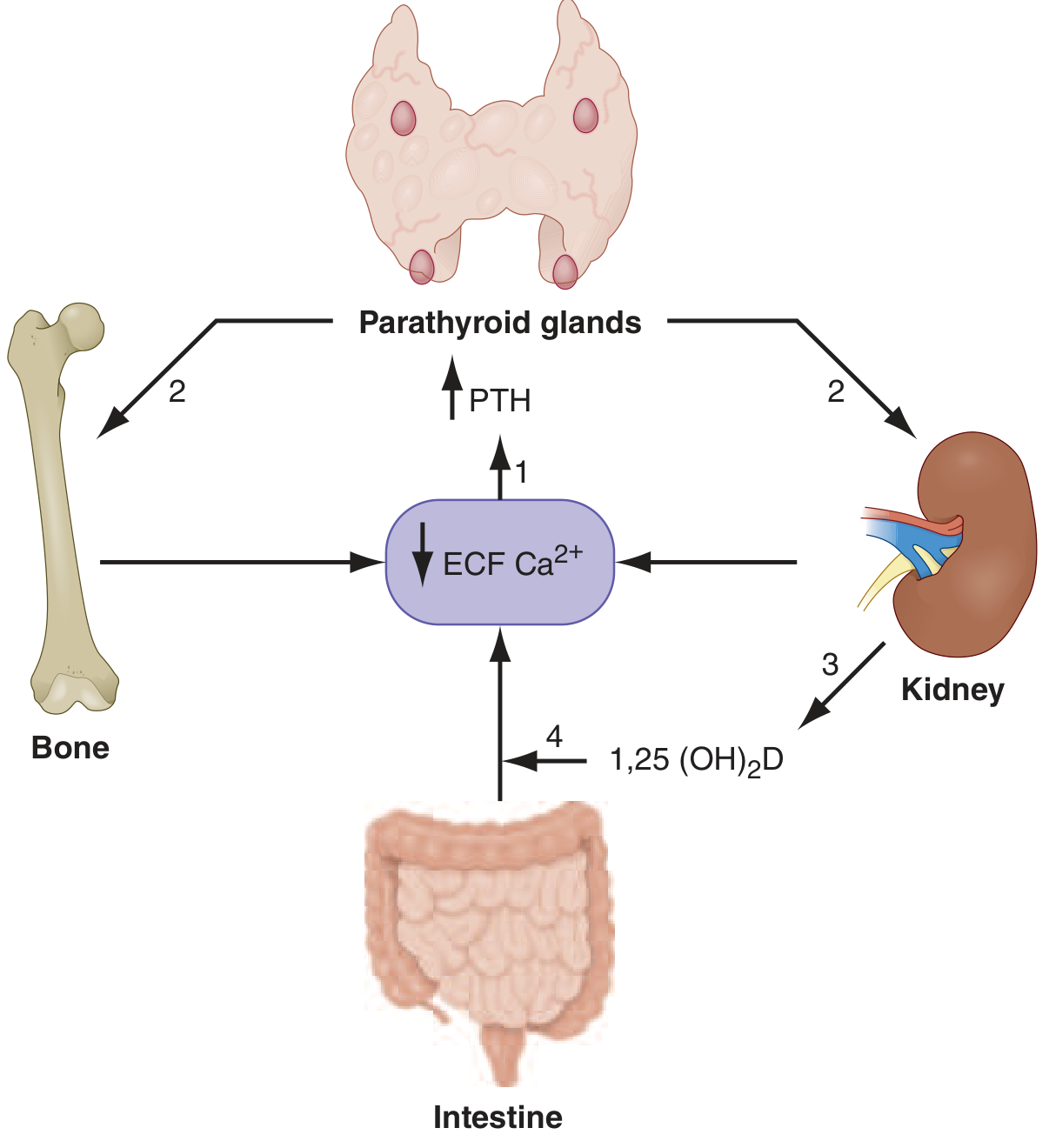
| Organ | PTH Effect | Mechanism |
|---|---|---|
| Bone | ↑ Calcium mobilization (resorption) | ↑ RANKL from osteocytes → osteoclast activation → hydroxyapatite dissolution |
| Kidney — Distal tubule | ↑ Ca²⁺ reabsorption | Activates apical Ca²⁺ channels (TRPV5), basolateral NCX |
| Kidney — Proximal tubule | ↓ Phosphate reabsorption (phosphaturia) | Inhibits NaPi-IIa cotransporter → reduces serum phosphate |
| Kidney — Proximal tubule | ↑ 1,25(OH)₂D production | Stimulates 1α-hydroxylase → converts 25(OH)D → active calcitriol |
| Intestine (indirect) | ↑ Ca²⁺ and PO₄³⁻ absorption | Via 1,25(OH)₂D acting on VDR in enterocytes |
Net result of PTH elevation: ↑ serum Ca²⁺, ↓ serum phosphate.
PTH Receptor Signaling
PTH binds PTH1R (a class B GPCR), coupling to:
- Gs → adenylyl cyclase → cAMP → PKA — major pathway in kidney and bone
- Gq → PLC → IP₃/DAG → PKC — secondary pathway
PTH Synthesis
PTH is synthesized as pre-pro-PTH (115 aa) → cleaved to pro-PTH (90 aa) in the ER → cleaved to mature PTH (1–84) in the Golgi. Half-life of intact PTH in plasma is only 2–4 minutes (rapidly cleaved in liver and kidney to N-terminal and C-terminal fragments). N-terminal fragments (1–34) retain full biological activity.
Other Regulators of PTH Secretion
| Stimulus | Effect on PTH |
|---|---|
| ↓ Serum Ca²⁺ | ↑↑ PTH (primary stimulus) |
| ↑ Serum Ca²⁺ | ↓↓ PTH |
| ↓ Serum Mg²⁺ (mild) | ↑ PTH |
| ↓ Serum Mg²⁺ (severe, chronic) | ↓ PTH (impairs secretory machinery) |
| ↑ Serum PO₄³⁻ | ↑ PTH (indirect — by lowering Ca²⁺; also direct effect) |
| 1,25(OH)₂D (calcitriol) | ↓ PTH (direct negative feedback on parathyroid gene expression) |
| FGF-23 | ↓ PTH (via Klotho/FGFR signaling) |
Relationship with Vitamin D
PTH and vitamin D form a tightly coupled axis:
- Low Ca²⁺ → ↑ PTH → ↑ renal 1α-hydroxylase → ↑ 1,25(OH)₂D → ↑ intestinal Ca²⁺ absorption
- High 1,25(OH)₂D → directly suppresses PTH gene transcription (via VDR in parathyroid chief cells) and promotes Ca²⁺ absorption → lowers PTH
— Medical Physiology (Boron & Boulpaep); Ganong's Review of Medical Physiology 26e; Harrison's Principles of Internal Medicine 22e
5. Summary Table
| Feature | Detail |
|---|---|
| Number of glands | 4 (occasionally 3–6) |
| Weight per gland | ~30–40 mg |
| Embryologic origin — superior | 4th pharyngeal pouch |
| Embryologic origin — inferior | 3rd pharyngeal pouch (with thymus) |
| Blood supply | Inferior thyroid artery (thyrocervical trunk) |
| Major secretory cell | Chief cell (principal cell) |
| Hormone secreted | PTH (parathyroid hormone), 84 aa |
| Primary stimulus for PTH | Hypocalcemia (via CaSR) |
| Primary inhibitor of PTH | Hypercalcemia + 1,25(OH)₂D |
| PTH receptor | PTH1R (Gs/Gq-coupled GPCR) |
| Bone action | ↑ RANKL → osteoclast resorption → ↑ Ca²⁺ |
| Renal action | ↑ Ca²⁺ reabsorption, ↓ phosphate reabsorption, ↑ 1α-hydroxylase |
| Intestinal action | Indirect via 1,25(OH)₂D → ↑ Ca²⁺ absorption |
Key Sources:
- Histology: A Text and Atlas with Correlated Cell and Molecular Biology (Ross & Pawlina, 9e)
- Cummings Otolaryngology Head and Neck Surgery
- Medical Physiology (Boron & Boulpaep)
- Ganong's Review of Medical Physiology, 26e
- Harrison's Principles of Internal Medicine, 22e
This is a shared conversation. Sign in to Orris to start your own chat.