Write down the pathogens and pathogenesis over tetanus and write short notes on botulism
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
I now have all the information needed for a comprehensive, well-structured answer. Here it is:
Tetanus
The Pathogen — Clostridium tetani
| Feature | Details |
|---|---|
| Type | Gram-positive, anaerobic, slender, motile bacillus |
| Spore | Terminal, round spore — gives a classic drumstick / tennis-racket appearance |
| Oxygen requirement | Strictly anaerobic; extremely oxygen-sensitive |
| Antigen | Several types distinguished by flagellar (H) antigens; all share a common O (somatic) antigen and produce the same neurotoxin |
| Environment | Spores ubiquitous in soil (especially manure-treated), lower GI tract of animals and humans; viable for years; resistant to boiling and most disinfectants |
The Toxin — Tetanospasmin
- Plasmid-encoded protein neurotoxin (~150 kDa), cleaved by a bacterial protease into two peptides: a heavy chain (100 kDa) and a light chain (50 kDa), linked by a disulfide bond.
- Heat-labile, readily neutralized by antitoxin, destroyed by intestinal proteases.
- One of the most potent known microbial toxins — lethal in humans at ~2.5 ng/kg.
- Formaldehyde treatment converts it to a non-toxic toxoid that retains antigenicity (basis of vaccination).
Pathogenesis
Step 1 — Entry of Spores
Tetanus spores enter through contaminated wounds — puncture wounds, lacerations, burns, surgical wounds, injection sites (IV drug users), or the umbilical stump of neonates. The wound itself is often minor and may go unnoticed.
Step 2 — Germination
Germination requires a low oxidation-reduction (redox) potential, created by:
- Necrotic tissue
- Calcium salts
- Associated pyogenic (facultative or anaerobic) co-infections
- Foreign bodies (splinters, soil)
Spores germinate → vegetative bacteria multiply locally in devitalized tissue → do not invade adjacent tissue (non-invasive organism). The disease is essentially a toxemia.
Step 3 — Toxin Production and Transport
- Tetanospasmin is produced at the local wound site.
- The heavy chain binds to receptors on presynaptic membranes of lower motor neurons at the wound periphery.
- Toxin is taken up and transported retrogradely via axonal transport (at ~75–250 mm/day) in membrane-bound vesicles → travels up the motor neuron to its cell body in the anterior horn of the spinal cord and brainstem.
Step 4 — Mechanism of Action
At the spinal cord/brainstem level, the toxin diffuses to the inhibitory interneurons (glycinergic and GABAergic neurons):
- The light chain acts as a metalloproteinase that cleaves synaptobrevin (VAMP2) — a protein essential for docking of neurotransmitter vesicles on the presynaptic membrane.
- This blocks release of the inhibitory neurotransmitters glycine and GABA.
- Without inhibition → motor neurons fire unopposed → sustained, simultaneous contraction of agonist and antagonist muscles → spastic/spasmodic paralysis.
This is the opposite of botulism: tetanus causes spastic (rigid) paralysis by blocking inhibitory neurons; botulism causes flaccid paralysis by blocking excitatory (acetylcholine) release.
Toxin binding is irreversible — recovery requires growth of new axon terminals.
Step 5 — Autonomic Involvement
As the disease progresses, toxin affects the autonomic nervous system, causing labile blood pressure, cardiac arrhythmias, profuse diaphoresis, and hyperthermia.
Clinical Features (Brief)
- Incubation: 4 days–3 weeks (mean 8 days); shorter incubation = more severe disease
- Trismus (lockjaw) — masseter spasm, the earliest sign in ~75% of cases
- Opisthotonos — arching spasm of paraspinal muscles
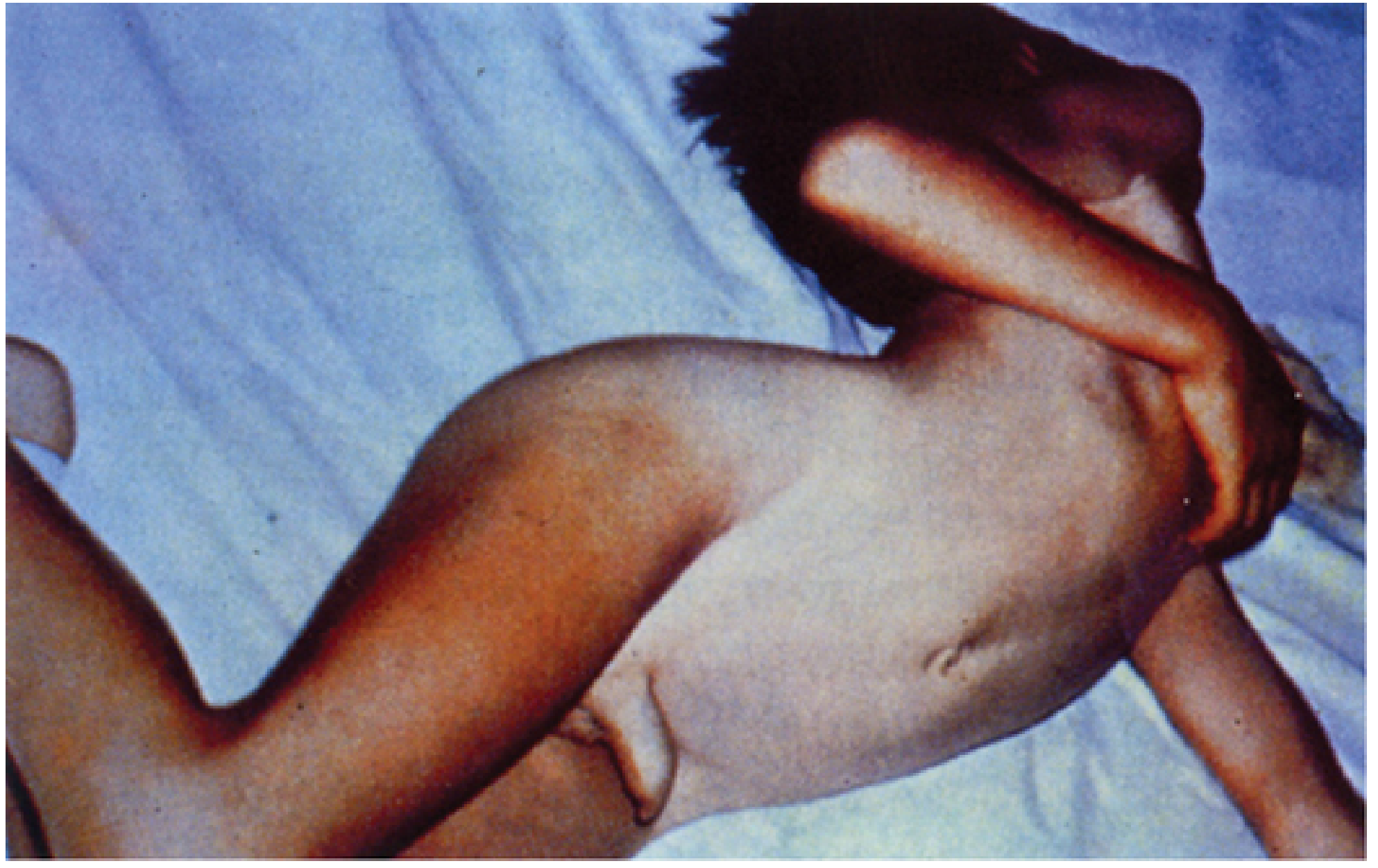
- Generalized convulsions triggered by minor stimuli (sound, touch, light)
- Patient remains fully conscious throughout
- Death from respiratory failure — untreated mortality 15–60%
Short Notes on Botulism
Pathogen — Clostridium botulinum
A Gram-positive, anaerobic, spore-forming bacillus. Spores are found worldwide in soil, pond, and lake sediments. Seven antigenically distinct toxin types (A–G) exist; types A, B, E, and F cause human disease.
The Toxin — Botulinum Toxin
- A heat-labile neurotoxin; the most potent biological toxin known.
- Like tetanospasmin, it is a metalloproteinase that cleaves synaptobrevin (VAMP2), blocking neurotransmitter vesicle docking.
- However, its target is peripheral cholinergic synapses (neuromuscular junctions) → blocks acetylcholine release → flaccid (descending) paralysis.
- Alkaline foods (green beans, mushrooms, fish) particularly support toxin production in improperly canned/heated foods.
Forms of Botulism
| Form | Mechanism |
|---|---|
| Foodborne | Ingestion of preformed toxin in improperly preserved food (intoxication, not infection) |
| Infant botulism | Ingestion of spores (e.g., honey) → germinate in infant colon → in vivo toxin production; most common form in the USA |
| Wound botulism | Spores contaminate wounds (e.g., IV/intranasal drug users) → in vivo toxin production |
| Inhalation botulism | Bioterrorism context — aerosolized toxin |
Pathogenesis
- Preformed toxin is absorbed in the GI tract → enters bloodstream.
- Toxin binds irreversibly to presynaptic terminals of peripheral motor neurons.
- Blocks acetylcholine (ACh) release at the neuromuscular junction → flaccid paralysis.
- Autonomic involvement → cardiac arrhythmias, blood pressure instability, dry mouth, constipation.
- Recovery requires growth of new presynaptic axons and formation of new synapses (weeks–months).
Clinical Features
- Onset: 12–36 hours after ingestion
- Early: nausea, dry mouth, diarrhea
- Cranial nerve signs: blurred vision, diplopia, pupillary dilation, dysarthria, dysphagia
- Symmetric descending flaccid paralysis → respiratory muscles → complete respiratory paralysis
- Infant botulism: constipation, poor tone ("floppy baby"), lethargy, poor feeding — may mimic SIDS
- Patient remains afebrile and conscious
Diagnosis
- Toxin detection in serum, stool, or food by immunoassay (EIA) or nucleic acid amplification (NAA) — available only at reference laboratories.
- Culture of C. botulinum from stool or implicated food.
Treatment & Prevention
- Supportive care (mechanical ventilation) is the most critical intervention — with proper support, mortality <10%
- Equine botulinum antitoxin to neutralize free toxin (not for infants — hypersensitivity risk; use human-derived BabyBIG for infant botulism)
- Antibiotics only for wound botulism
- Prevention: adequate pressure cooking/autoclaving kills spores; heating food at 100°C for 10 min destroys preformed toxin; avoid damaged or bulging cans
- No vaccine available
Therapeutic use: Botulinum toxin (Botox) — injected locally for spasmodic neuromuscular conditions and cosmetic applications (wrinkle reduction).
Sources: Sherris & Ryan's Medical Microbiology, 8e | Jawetz, Melnick & Adelberg's Medical Microbiology, 28e | Goldman-Cecil Medicine, International Edition | Medical Microbiology, 9e
This is a shared conversation. Sign in to Orris to start your own chat.