Classify the anti-retroviral drugs therapy and discuss their mechanisms of action in details 2nd yr mbbs refer kdt
Reading File
Finding Sources
Finding Sources
Reading File
Finding Sources
Reading File
Reading File
Finding Sources
Loading Image
Reading File
Reading File
Reading File
I have comprehensive content from both Katzung and Lippincott. Now I'll compile the detailed answer.
Antiretroviral (ARV) Drug Therapy — Classification & Mechanisms of Action
(Based on Katzung's Basic & Clinical Pharmacology 16th ed. and Lippincott Illustrated Reviews: Pharmacology)
HIV Life Cycle — The Basis of Drug Targets
Before classifying drugs, understanding the HIV replication cycle is essential, as each drug class targets a specific step:
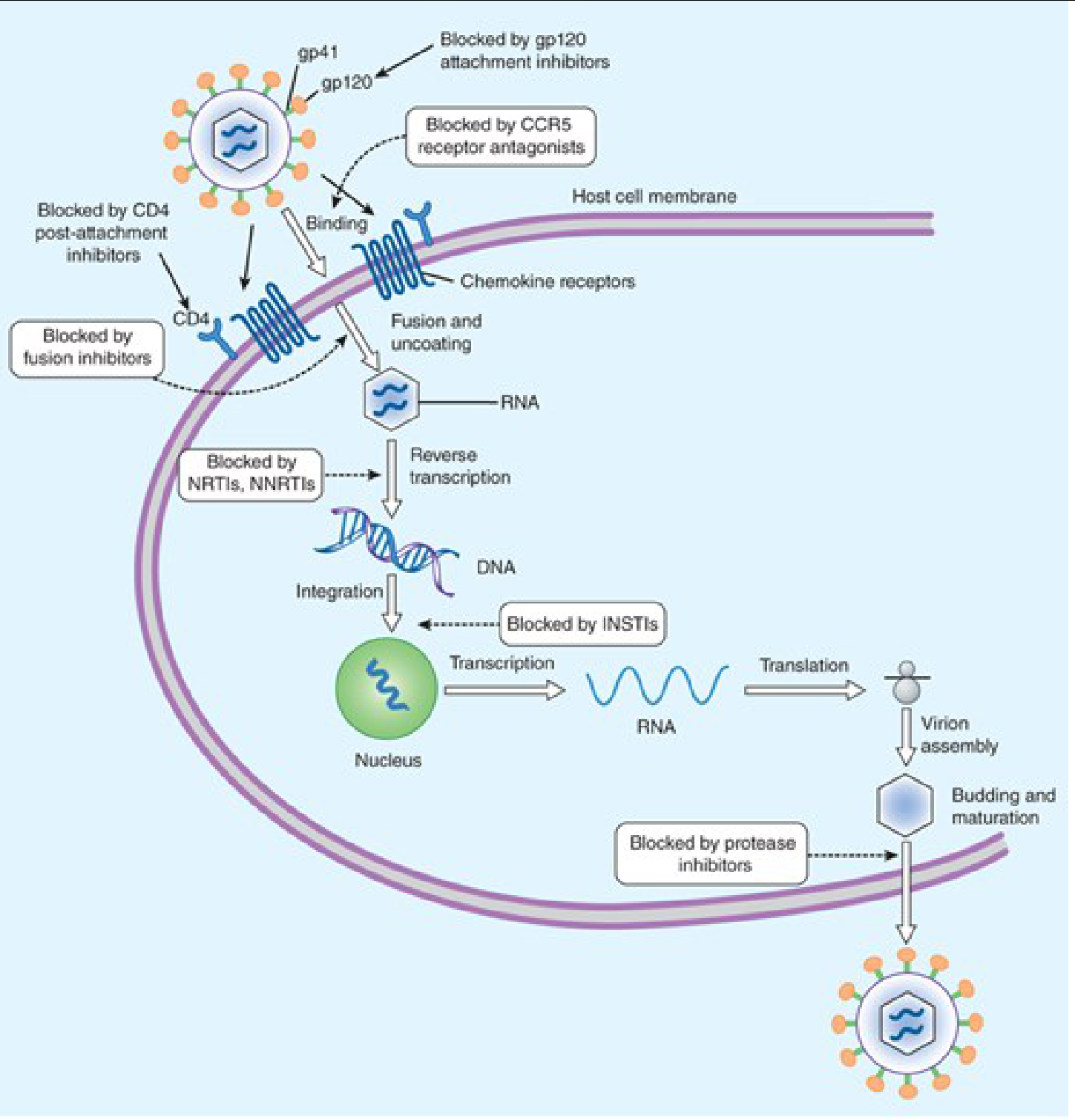
Steps of HIV replication:
- Binding/Attachment — gp120 binds CD4 receptor → conformational change → gp120 binds CCR5/CXCR4 co-receptor
- Fusion — gp41 mediates membrane fusion; viral RNA enters cell
- Reverse Transcription — viral RNA → double-stranded DNA (via reverse transcriptase, RT)
- Integration — viral DNA integrates into host genome (via integrase)
- Transcription & Translation — host machinery produces viral proteins
- Assembly, Budding & Maturation — protease cleaves polyproteins → infectious virions
Classification of Antiretroviral Drugs
| Class | Mechanism | Key Drugs |
|---|---|---|
| NRTIs (Nucleoside/Nucleotide Reverse Transcriptase Inhibitors) | Chain termination of viral DNA | Zidovudine, Lamivudine, Tenofovir, Abacavir, Emtricitabine |
| NNRTIs (Non-Nucleoside Reverse Transcriptase Inhibitors) | Allosteric inhibition of RT | Nevirapine, Efavirenz, Rilpivirine, Doravirine |
| PIs (Protease Inhibitors) | Block viral polyprotein cleavage | Atazanavir, Darunavir, Lopinavir, Ritonavir |
| INSTIs (Integrase Strand Transfer Inhibitors) | Block HIV DNA integration | Raltegravir, Dolutegravir, Bictegravir, Elvitegravir |
| Fusion Inhibitors | Block gp41-mediated membrane fusion | Enfuvirtide (T-20) |
| CCR5 Co-receptor Antagonists | Block CCR5 binding | Maraviroc |
| CD4 Post-attachment Inhibitors | Block post-CD4 binding | Ibalizumab |
| gp120 Attachment Inhibitors | Block gp120–CD4 interaction | Fostemsavir |
| Capsid Inhibitors | Block capsid-mediated nuclear import | Lenacapavir |
| Pharmacokinetic Enhancers (Boosters) | CYP3A4 inhibition → increase PI/INSTI levels | Ritonavir (low dose), Cobicistat |
Detailed Mechanisms of Action
1. Nucleoside/Nucleotide Reverse Transcriptase Inhibitors (NRTIs)
Step targeted: Reverse Transcription (RNA → DNA)
Mechanism:
NRTIs are structural analogs of natural nucleosides but critically lack a 3'-hydroxyl group on the ribose sugar.
- They enter the cell and are phosphorylated by host cellular kinases to their active triphosphate form
- The triphosphate form is competitively incorporated into the growing viral DNA chain by RT (in preference to natural nucleotides)
- Because the 3'-OH group is absent, the next nucleotide cannot form a 3',5'-phosphodiester bond → obligate chain termination
- Viral DNA synthesis is halted
Exception — Tenofovir (Nucleotide analog): Requires only two phosphorylation steps (already carries one phosphate) instead of three. Available as prodrugs TDF (tenofovir disoproxil fumarate) and TAF (tenofovir alafenamide). TAF achieves higher intracellular drug concentrations with lower plasma levels → fewer systemic side effects (less renal toxicity, less bone mineral density loss).
Key drugs & notes:
- Zidovudine (ZDV/AZT): Thymidine analog; prototype NRTI; used in pregnancy (prevention of vertical transmission)
- Lamivudine (3TC): Cytosine analog; also active against HBV; very well tolerated; selects M184V resistance mutation
- Emtricitabine (FTC): Fluorinated analog of 3TC; longer intracellular half-life (≥24h) → once-daily dosing
- Abacavir (ABC): Guanosine analog; 5% risk of hypersensitivity (HLA-B*5701 screening mandatory); never rechallenge
- Tenofovir: Adenosine nucleotide analog; active against HIV + HBV; backbone of most first-line regimens
Class toxicity (NRTI): Mitochondrial toxicity (inhibit mitochondrial DNA polymerase γ) → lactic acidosis, hepatomegaly with steatosis (rare but potentially fatal), lipodystrophy, peripheral neuropathy, pancreatitis
2. Non-Nucleoside Reverse Transcriptase Inhibitors (NNRTIs)
Step targeted: Reverse Transcription (RNA → DNA)
Mechanism:
NNRTIs are not incorporated into viral DNA. Instead, they bind directly and non-competitively to a hydrophobic allosteric pocket on the p66 subunit of HIV-1 RT (distinct from the active/catalytic site).
- Binding induces a conformational change in the RT enzyme
- This restricts flexibility of the catalytic site, reducing RT polymerase activity
- Result: decreased rate and processivity of reverse transcription
NNRTIs have no activity against HIV-2 (which has a different RT pocket conformation) and they do not require intracellular phosphorylation.
Key drugs:
- Nevirapine: First NNRTI; induces its own metabolism (CYP3A4 inducer); rash and hepatotoxicity are main concerns
- Efavirenz: Most widely used historically; once-daily; excellent CNS penetration but causes neuropsychiatric effects (vivid dreams, dizziness, depression); teratogenic (Category D)
- Rilpivirine: Second-generation; better tolerated than efavirenz; requires food for absorption; acid-reducing agents reduce levels
- Doravirine: Newest NNRTI; fewer drug interactions; no lipid effects
Resistance: A single point mutation (e.g., K103N) confers high-level resistance to all first-generation NNRTIs — low genetic barrier to resistance.
3. Protease Inhibitors (PIs)
Step targeted: Virion Maturation (post-budding)
Mechanism:
- After HIV buds from the host cell as an immature, non-infectious virion, HIV protease cleaves Gag and Gag-Pol polyproteins into functional structural proteins (p24 capsid, p17 matrix) and viral enzymes (RT, integrase)
- PIs are peptidomimetic compounds that act as competitive inhibitors of HIV aspartyl protease — they bind to the protease's catalytic site
- Without proteolytic cleavage, virions remain immature and non-infectious
Key drugs:
- Atazanavir: Once daily; inhibits UGT1A1 → indirect hyperbilirubinemia (jaundice); prolongs PR interval
- Darunavir: Most potent PI; high genetic barrier to resistance; used with ritonavir or cobicistat booster
- Lopinavir/ritonavir (Kaletra): Fixed-dose co-formulation; ritonavir here acts as a pharmacokinetic booster (see below)
- Ritonavir: At full doses — PI; at low doses (100–200 mg) — pharmacokinetic booster only (potent CYP3A4 inhibitor)
Class toxicity: GI intolerance (nausea, diarrhea), metabolic syndrome (dyslipidemia, insulin resistance, fat redistribution/lipodystrophy), nephrolithiasis (especially indinavir/atazanavir — crystalluria), indirect hyperbilirubinemia (atazanavir), drug interactions (CYP3A4 inhibitors)
4. Integrase Strand Transfer Inhibitors (INSTIs)
Step targeted: Integration of viral DNA into host genome
Mechanism:
- After reverse transcription, viral double-stranded DNA forms the pre-integration complex (PIC) with integrase
- Integrase performs two reactions:
- 3'-processing: Cleaves 3' ends of viral DNA
- Strand transfer: Inserts ("integrates") viral DNA into host chromosomal DNA
- INSTIs bind the integrase–viral DNA complex via chelation of two Mg²⁺ ions in the integrase active site
- This blocks strand transfer (the 3rd and final step of integration), preventing provirus formation
Without integration, no permanent infection can be established.
Key drugs:
- Raltegravir: First NNRTI; twice daily; well tolerated; low genetic barrier
- Dolutegravir: Once daily; high genetic barrier to resistance; preferred in first-line regimens; slight weight gain; neural tube defects concern in early pregnancy (requires counseling)
- Bictegravir: Highest genetic barrier; co-formulated with TAF + FTC as a single-pill once-daily regimen (Biktarvy) — preferred first-line regimen for most patients
- Elvitegravir: Requires cobicistat booster; co-formulated in Stribild/Genvoya
5. Fusion Inhibitors
Step targeted: Viral Fusion / Entry
Mechanism:
- Enfuvirtide (T-20): A 36-amino-acid synthetic peptide that mimics the HR2 region of gp41
- When gp120 binds CD4 and co-receptor, gp41 undergoes a conformational change — the HR1 and HR2 heptad repeat regions form a six-helix bundle that pulls viral and host membranes together
- Enfuvirtide binds HR1 of gp41 → prevents formation of the six-helix bundle → membrane fusion is blocked → viral RNA cannot enter the cell
- Must be given subcutaneously twice daily (peptide, not orally bioavailable)
- Reserved for treatment-experienced patients with resistant virus
- Main adverse effect: injection site reactions (pain, nodules, erythema — in nearly all patients)
6. CCR5 Co-receptor Antagonists
Step targeted: Viral Attachment / Entry
Mechanism:
- Maraviroc: Binds the human CCR5 chemokine receptor (a co-receptor used by R5-tropic HIV strains)
- Binding induces allosteric changes in CCR5 → prevents gp120 from binding
- Viral fusion and entry are blocked
- Requires tropism testing before use — only effective against CCR5-tropic (R5) HIV; X4-tropic or dual-mixed virus will not respond
- Well tolerated; hepatotoxicity risk; possible increased risk of cardiovascular events
7. CD4 Post-attachment Inhibitor
Step targeted: Post-CD4 attachment (before CCR5/CXCR4 binding)
Mechanism:
- Ibalizumab: A monoclonal antibody that binds domain 2 of the CD4 receptor on T cells
- After gp120 binds CD4 (domain 1), ibalizumab blocks the conformational change required for gp120 to then engage the co-receptor
- Entry is blocked without impairing CD4's normal immune function (MHC class II interactions use a different CD4 domain)
- Given IV every 2 weeks
- For multidrug-resistant HIV
8. gp120 Attachment Inhibitor
Mechanism:
- Fostemsavir (prodrug → temsavir): Binds directly to HIV gp120, preventing gp120 from attaching to the CD4 receptor
- Blocks the very first step of viral entry — upstream of CCR5 antagonists and fusion inhibitors
- For heavily treatment-experienced adults
9. Capsid Inhibitor
Mechanism:
- Lenacapavir: Binds the HIV-1 capsid protein (CA) — a multi-stage mechanism:
- Interferes with capsid assembly (late stage) and disassembly (early stage after cell entry)
- Blocks nuclear import of the viral capsid complex
- Active against virus resistant to all other ARV classes
- Administered subcutaneously every 6 months — longest-acting ARV available
10. Pharmacokinetic Enhancers (Boosters)
Not antivirals themselves, but critical for optimizing ARV levels:
| Booster | Mechanism | Used With |
|---|---|---|
| Ritonavir (low dose 100–200 mg) | Potent CYP3A4 inhibitor → ↑ co-PI plasma levels | PIs (atazanavir, darunavir, lopinavir) |
| Cobicistat | CYP3A4 + CYP2D6 + P-gp inhibitor → ↑ co-drug levels | Atazanavir, darunavir, elvitegravir |
Both prevent rapid first-pass metabolism of the "boosted" drug → higher Cmin levels → better viral suppression and lower resistance risk.
HAART / cART — Combined Antiretroviral Therapy
Monotherapy and dual-NRTI therapy are obsolete due to rapid resistance. The current standard is:
cART (combination ART): 2 NRTIs + 1 INSTI (preferred) OR 2 NRTIs + 1 NNRTI OR 2 NRTIs + 1 boosted PI
Preferred first-line regimen (most patients):
- Bictegravir/TAF/FTC (single pill once daily) — highest genetic barrier, best tolerability
Goals of therapy:
- Suppress viral load to undetectable (< 50 copies/mL)
- Preserve/restore CD4+ T-cell count
- Prevent opportunistic infections and AIDS-defining illness
- Prevent HIV transmission
High-Yield Summary for 2nd Year MBBS
| Drug Class | Target Step | Key Example | Mnemo |
|---|---|---|---|
| NRTI | Reverse transcription (chain termination) | Zidovudine, Tenofovir | No 3'-OH → chain stops |
| NNRTI | Reverse transcription (allosteric) | Efavirenz, Nevirapine | Bind RT pocket, no phosphorylation needed |
| PI | Maturation (polyprotein cleavage) | Darunavir, Atazanavir | Immature virions → non-infectious |
| INSTI | Integration (strand transfer) | Dolutegravir, Bictegravir | Chelate Mg²⁺ in integrase |
| Fusion inhibitor | Fusion (gp41) | Enfuvirtide | Prevents 6-helix bundle |
| CCR5 antagonist | Entry (co-receptor) | Maraviroc | Blocks CCR5 |
| Attachment inhibitor | Attachment (gp120) | Fostemsavir | Blocks gp120–CD4 binding |
Sources: Katzung's Basic & Clinical Pharmacology, 16th ed., Ch. 49; Lippincott Illustrated Reviews: Pharmacology, Ch. 34
Make a visual flowchart for first-line vs second-line ARV selection based on viral genotype 2nd yr mbbs refer kdt
This is a shared conversation. Sign in to Orris to start your own chat.