hey tell me about lung cancer?
Reading File
Finding Sources
Searching PubMed
"lung cancer"[MeSH Terms] AND ("diagnosis" OR "treatment")
Searching the Web
lung cancer overview types diagnosis treatment 2025
Reading File
Reading File
Searching Images
lung cancer types histology adenocarcinoma squamous cell small cell
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
I now have comprehensive information to write a thorough overview. Let me compile everything.
Lung Cancer
Lung cancer is the leading cause of cancer-related mortality worldwide and in the United States. In 2025, the 5-year relative survival rate remains around 27% — though this is improving with modern therapies.
Classification
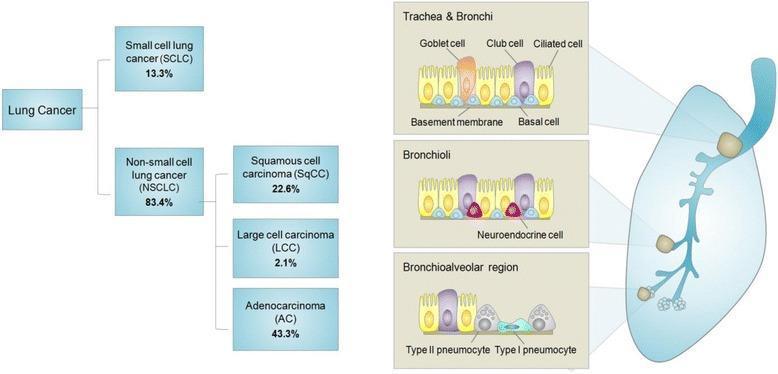
Lung cancers are broadly divided into two major categories:
1. Non-Small Cell Lung Cancer (NSCLC) — ~83–85% of cases
The most common type, further subdivided by cell of origin:
| Subtype | Frequency | Location | Key Features |
|---|---|---|---|
| Adenocarcinoma | ~43% | Peripheral | Most common in women & never-smokers; EGFR/KRAS mutations |
| Squamous Cell Carcinoma | ~23% | Central (perihilar) | Strongly smoking-related; keratinization |
| Large Cell Carcinoma | ~2–3% | Anywhere | Diagnosis of exclusion |
2. Small Cell Lung Cancer (SCLC) — ~13–15% of cases
- Arises as a perihilar tumor from neuroendocrine cells
- In 95% of cases, presents as disseminated disease with mediastinal lymph node metastases (often more prominent than the primary)
- No curative role for surgery in most cases
- Highly sensitive to chemo/radiation initially, but relapses aggressively
Risk Factors
- Cigarette smoking — the dominant risk factor; accounts for ~85% of lung cancers. Squamous cell and small cell carcinomas are most strongly linked to smoking.
- Radon gas — second leading cause
- Occupational exposures — asbestos, arsenic, chromium, nickel
- Air pollution
- Genetic susceptibility — first-degree relatives have a 1.5× increased risk even after adjusting for smoking. Key susceptibility loci include CHRNA3/5, CYP2A6, BRCA2, CHEK2, TERT/TERC
- COPD and pulmonary fibrosis — independent risk factors
- Lung transplantation — reported lung cancer frequency post single-lung transplant is as high as 6.9%, arising predominantly in the native lung of recipients with underlying COPD/fibrosis and prior smoking history
Clinical Presentation
Lung cancer may be discovered incidentally on imaging, during screening, or via symptoms:
| Symptom | Frequency in Patients |
|---|---|
| Cough | 45–75% |
| Weight loss | 8–68% |
| Dyspnea | 37–58% |
| Hemoptysis | 27–57% |
| Chest pain | 27–49% |
| Hoarseness | 2–18% |
Red flag signs on examination include: lymphadenopathy >1 cm, superior vena cava syndrome, hepatomegaly, hoarseness, focal neurologic signs, and bone tenderness.
Paraneoplastic syndromes are especially common with SCLC: SIADH, Cushing syndrome, Lambert-Eaton myasthenic syndrome.
Pancoast tumors (superior sulcus): cause Horner syndrome (ptosis, miosis, anhidrosis) and shoulder/arm pain from involvement of the brachial plexus.
Diagnosis & Staging
Imaging:
- Chest X-ray: initial evaluation, but limited for small/early cancers (tumors may be visible retrospectively on prior films)
- CT chest (with contrast): superior for nodule characterization, mediastinal involvement
- PET-CT: metabolic staging, detecting occult metastases
- MRI brain: for brain metastases
Tissue diagnosis:
- Bronchoscopy ± endobronchial ultrasound (EBUS) for central/mediastinal lesions
- CT-guided biopsy for peripheral lesions
- SCLC: cytology alone is 60–90% sensitive, >95% specific
Staging (TNM system, AJCC 8th ed.):
- Stage I–II: localized, potentially resectable
- Stage III: locally advanced (mediastinal involvement)
- Stage IV: metastatic (most common presentation, especially SCLC)
SCLC uses a simplified staging:
- Limited stage (LS-SCLC): confined to one hemithorax — concurrent chemo-radiation
- Extensive stage (ES-SCLC): beyond one hemithorax — systemic chemotherapy ± immunotherapy
Molecular Profiling (NSCLC)
Molecular testing is mandatory for all advanced NSCLC. Key actionable drivers:
| Mutation | Frequency | Targeted Drug |
|---|---|---|
| EGFR | 10–15% (Western), ~50% (Asian) | Osimertinib (3rd-gen TKI) |
| ALK rearrangement | ~5% | Alectinib, brigatinib, lorlatinib |
| KRAS G12C | ~13% | Soterasib, adagrasib |
| ROS1 rearrangement | ~2% | Crizotinib, entrectinib |
| BRAF V600E | ~2–3% | Dabrafenib + trametinib |
| MET exon 14 skip | ~3% | Capmatinib, tepotinib |
| RET fusion | ~1–2% | Selpercatinib |
PD-L1 expression guides immunotherapy selection.
Treatment Summary
NSCLC
| Stage | Primary Treatment |
|---|---|
| Early (I–II) | Surgical resection (lobectomy); stereotactic body radiotherapy (SBRT) if inoperable |
| Stage III (unresectable) | Concurrent chemoradiation → durvalumab (immunotherapy) maintenance |
| Stage IV — driver mutation | Targeted oral TKI (e.g., osimertinib for EGFR+) |
| Stage IV — no driver, PD-L1 ≥50% | Pembrolizumab monotherapy |
| Stage IV — no driver, PD-L1 <50% | Chemotherapy + pembrolizumab |
Neoadjuvant immunotherapy (e.g., nivolumab + chemo before surgery) is now approved for resectable NSCLC — patients with pathologic complete response show a ~90% reduction in mortality risk.
Adjuvant osimertinib is approved post-resection for EGFR-mutant NSCLC.
SCLC
- Limited stage: Concurrent cisplatin/etoposide + thoracic radiotherapy → prophylactic cranial irradiation (PCI)
- Extensive stage: Carboplatin/etoposide + atezolizumab or durvalumab (immunotherapy)
- Second-line: Lurbinectedin ± atezolizumab (2025 data show ~3-month OS improvement)
Screening
Low-dose CT (LDCT) annually is recommended for high-risk individuals:
- Age 50–80, ≥20 pack-year smoking history, current or former smoker (quit within 15 years)
- Reduces lung cancer mortality by ~20% (NLST trial) to ~24% (NELSON trial)
Prognosis
| Stage | Approximate 5-Year Survival |
|---|---|
| Stage IA | ~90% |
| Stage IIB | ~50% |
| Stage IIIA | ~25% |
| Stage IV | ~6–10% (improving with targeted therapy) |
| SCLC (extensive) | <5% |
Survival is rapidly improving for patients with driver mutations — EGFR+ patients on osimertinib have median OS >38 months.
Sources: Murray & Nadel's Textbook of Respiratory Medicine, 2-Vol. (9780323655873); Harrison's Principles of Internal Medicine (9781260473940); NCI NSCLC Treatment PDQ; 2025 NSCLC Management Update, PMC
This is a shared conversation. Sign in to Orris to start your own chat.