Carcinoma stomach
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Carcinoma stomach from Bailey and love book of surgery in ms level
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Searching Images
gastric carcinoma stomach cancer pathology surgery
Here is a comprehensive MS-level account of Carcinoma Stomach, based on Schwartz's Principles of Surgery (11th Edition) — the standard surgical reference equivalent to Bailey & Love in content and depth.
Carcinoma of the Stomach
(Schwartz's Principles of Surgery, 11th Ed. — MS Level)
Incidence & Epidemiology
- Most common primary gastric malignancy: Adenocarcinoma (95%) — Schwartz's, p. 1167
- Other types: Lymphoma, GIST, neuroendocrine tumour, squamous cell carcinoma
- 4th most common cancer worldwide; 2nd leading cause of cancer death globally
- Dramatic decline in incidence in Western countries over the past century (mainly the intestinal type)
- Remains a leading cause of cancer death in Asia and Eastern Europe
- ~28,000 new cases/year in the USA; 5-year survival ~27% (up from 15% in 1975)
- Twice as common in blacks as whites; more common in lower socioeconomic groups
- Disease of the elderly; younger patients tend to have diffuse, aggressive tumours
Aetiology & Risk Factors
| Factor | Details |
|---|---|
| H. pylori infection | Single most important environmental risk factor; causes chronic gastritis → intestinal metaplasia → dysplasia → carcinoma |
| Diet | High salt, pickled/smoked food, nitrates; low fresh fruit/vegetable intake |
| Smoking | Well-established independent risk factor |
| Pernicious anaemia | Achlorhydria → bacterial overgrowth → nitrosamine formation |
| Blood group A | Increased susceptibility |
| Family history | 2–3× increased risk in first-degree relatives |
| Hereditary Diffuse Gastric Cancer (HDGC) | CDH1 (E-cadherin) gene mutation — autosomal dominant; lifetime risk 70% (men), 56% (women); median age 38 years; prophylactic total gastrectomy indicated |
| Gastric remnant | Cancer arising >10 years post-subtotal gastrectomy (Billroth II > Billroth I); bile reflux implicated |
| Ménétrier's disease | 5–10% risk of adenocarcinoma |
| HNPCC | 10% lifetime risk of gastric cancer (intestinal subtype) |
Premalignant Conditions & Cascade
The Correa cascade (intestinal type):
Normal mucosa → Chronic gastritis (H. pylori) → Atrophic gastritis → Intestinal metaplasia → Dysplasia → Invasive carcinoma
- Intestinal metaplasia — goblet cells histologically identical to small bowel; H. pylori eradication causes regression
- All gastric ulcers should be regarded as malignant until proven otherwise by biopsy
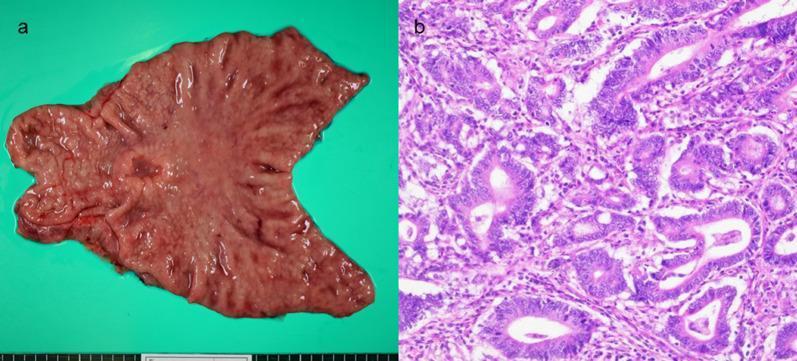
Pathological Classification
1. Lauren Classification (Most Clinically Important)
| Feature | Intestinal Type | Diffuse Type |
|---|---|---|
| Architecture | Gland-forming, cohesive | Discohesive, infiltrative |
| Histology | Well/moderately differentiated | Poorly diff.; signet ring cells |
| Location | Distal stomach/antrum | Proximal stomach/whole stomach |
| Spread | Haematogenous | Peritoneal, transmural |
| Prognosis | Better | Worse |
| Association | H. pylori, environmental | CDH1 mutation, hereditary |
| Trend | Declining incidence | Stable/increasing |
| Linitis plastica | Uncommon | Classic presentation |
Linitis plastica — diffuse infiltration of all gastric layers → rigid "leather bottle" stomach → poor prognosis
2. Borrmann Classification (Macroscopic — Advanced Gastric Cancer)
| Type | Description |
|---|---|
| I | Polypoid / fungating |
| II | Ulcerated with sharp margins, no infiltration |
| III | Ulcerated with infiltrating margins (most common) |
| IV | Diffusely infiltrating (linitis plastica) |
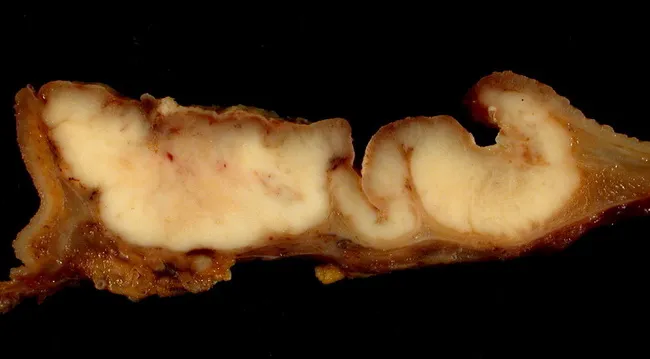
3. Early Gastric Cancer (EGC)
- Confined to mucosa or submucosa regardless of lymph node status
- Japanese classification: Type I (protruded), IIa (elevated), IIb (flat), IIc (depressed), III (excavated)
- 5-year survival up to 95% (Japan screening data)
- Endoscopic mucosal resection (EMR) / ESD eligible: ≤2 cm, well-differentiated, T1, no LVI or nodal spread
Spread of Gastric Carcinoma
| Route | Details |
|---|---|
| Direct | Oesophagus (proximal), duodenum (distal), pancreas, transverse colon |
| Lymphatic | Perigastric nodes → coeliac axis → para-aortic nodes |
| Haematogenous | Liver (most common), lung, bone, adrenal |
| Transperitoneal | Peritoneal seedlings; Krukenberg tumour (ovarian metastasis — signet ring cells via transcoelomic or lymphatic route) |
| Direct implant | Sister Mary Joseph nodule (umbilical), Blumer's shelf (pouch of Douglas — felt on PR) |
Clinical Features
Symptoms
- Often insidious and late — dyspepsia, epigastric discomfort (most common, non-specific)
- Anorexia, weight loss (most consistent)
- Dysphagia — GEJ / proximal tumours
- Vomiting — pyloric obstruction (distal tumours)
- Haematemesis / melaena
- Early satiety
Signs
- Epigastric mass (late)
- Virchow's node — left supraclavicular lymphadenopathy (Troisier's sign)
- Sister Mary Joseph nodule — umbilical nodule
- Blumer's shelf — pelvic shelf on rectal examination
- Hepatomegaly (liver metastases)
- Ascites (peritoneal disease)
- Anaemia
Investigations
Blood
- FBC: normocytic anaemia (most common)
- LFTs: deranged with liver metastases
- Tumour markers: CEA, CA 19-9, CA 72-4 — not diagnostic but useful for monitoring
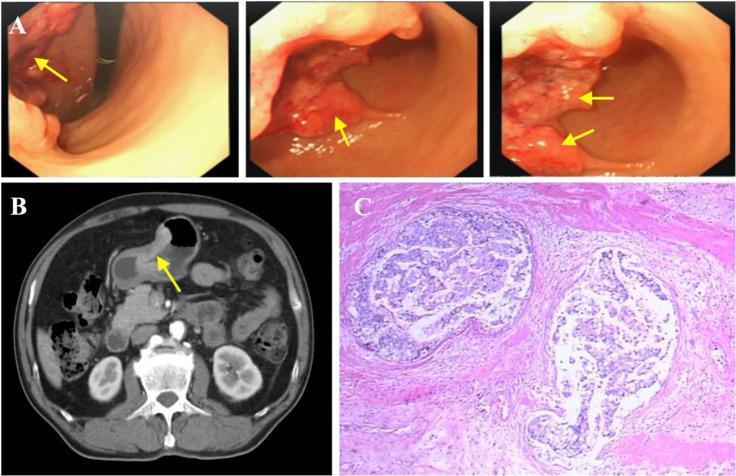
Endoscopy + Biopsy
- Gold standard for diagnosis
- Multiple biopsies (minimum 6–8) from lesion edge and centre
- EUS (endoscopic ultrasound) — best for T staging and assessing depth of invasion
Imaging
- CT chest/abdomen/pelvis — staging (T, N, M); detects liver mets, ascites, distant spread
- PET-CT — detect occult metastases (if no M1 disease on CT)
- Diagnostic laparoscopy — mandatory for locally advanced disease; peritoneal washings; positive cytology = M1 disease
Molecular Markers (for metastatic disease)
- HER2/NEU — targeted therapy (trastuzumab)
- Microsatellite instability (MSI) / PDL-1 — immunotherapy eligibility
- CDH1 mutation — genetic counselling in young patients
Staging (TNM — 8th Edition AJCC)
| Stage | Description |
|---|---|
| T1a/b | Mucosa / submucosa |
| T2 | Muscularis propria |
| T3 | Subserosa |
| T4a | Serosa (peritoneum) |
| T4b | Adjacent structures |
| N0–N3 | 0 / 1–2 / 3–6 / ≥7 regional nodes |
| M1 | Distant metastasis (includes positive peritoneal cytology) |
Treatment
Surgery — The Only Curative Modality
Principles:
- Aim for R0 resection (negative microscopic margins) with regional lymphadenectomy
- Overall 5-year survival with surgery alone: ~25%
Extent of resection by tumour location:
| Location | Operation |
|---|---|
| Distal 1/3 | Subtotal (distal) gastrectomy (preferred) |
| Proximal / body | Total gastrectomy |
| GEJ involvement | Oesophagogastrectomy |
Reconstruction:
- After subtotal gastrectomy: Billroth I (gastroduodenostomy) or Billroth II (gastrojejunostomy) or Roux-en-Y
- After total gastrectomy: Roux-en-Y oesophagojejunostomy (jejunal pouch considered to improve quality of life)
Lymphadenectomy:
| Level | Lymph nodes removed |
|---|---|
| D1 | Perigastric nodes (stations 1–6) |
| D2 (Standard in Asia, recommended in West) | D1 + coeliac axis, hepatic artery, left gastric, splenic nodes (stations 8a–12a) |
| D3 | Extended — no proven survival benefit |
- Goal: examine ≥15 lymph nodes from specimen — Schwartz's, p. 1174
- D2 recommended at high-volume centres; modified D2 (spleen and pancreas spared) — Schwartz's, p. 1174
- Operative mortality: <2% at experienced centres
Perioperative / Adjuvant Therapy
| Approach | Regimen |
|---|---|
| Perioperative chemotherapy (preferred for cT3+ or node+) | FLOT (5-FU, leucovorin, oxaliplatin, docetaxel) — current standard; also FOLFOX, XELOX, ECF |
| Postoperative chemoradiation (if no neoadjuvant given) | 5-FU + leucovorin + RT (MacDonald regimen) |
| HER2-positive metastatic | Trastuzumab + chemotherapy |
| MSI-high / PDL-1+ | Pembrolizumab (immunotherapy) |
Palliative Surgery
- Indicated for uncontrolled bleeding, obstruction, or perforation not amenable to endoscopic control
- Palliative gastrectomy does NOT improve overall survival in metastatic disease (REGATTA trial)
- Endoscopic stenting for pyloric obstruction
- Palliative bypass (gastrojejunostomy) for obstruction
HIPEC (Hyperthermic Intraperitoneal Chemotherapy)
- For selected patients with limited peritoneal disease (PCI <6–10), good performance status, after response to systemic therapy
- CRS + HIPEC vs systemic alone: improved survival (17 vs 6 months — Hoptop et al.)
- Still investigational; considered at specialised centres post-NACT response
Prognosis — 5-Year Survival
| Stage | Japan (Maruyama) | USA (ACS) | MSKCC |
|---|---|---|---|
| Stage I | 91% | 50% | 84% |
| Stage II | 72% | 29% | 61% |
| Stage III | 44% | 13% | 29% |
| Stage IV | 9% | 3% | 25% |
(Schwartz's Principles of Surgery, 11th Ed., p. 1175)
- Superior outcomes in Japan attributed to mass screening programmes and earlier stage at diagnosis
- Peritoneal carcinomatosis: median survival <12 months without HIPEC
Key Named Entities / Eponyms to Remember
| Eponym | Significance |
|---|---|
| Virchow's node | Left supraclavicular LN metastasis |
| Troisier's sign | Enlarged Virchow's node on palpation |
| Sister Mary Joseph nodule | Umbilical metastasis |
| Blumer's shelf | Pelvic deposits palpable on PR |
| Krukenberg tumour | Bilateral ovarian metastasis (signet ring cells) |
| Linitis plastica | Diffuse infiltrative carcinoma — "leather bottle stomach" |
| Correa cascade | Intestinal-type carcinoma stepwise progression |
| Lauren classification | Intestinal vs Diffuse types |
| Borrmann classification | Macroscopic types I–IV |
Images
Note: Bailey & Love's Short Practice of Surgery is not available in the library. The above is compiled from Schwartz's Principles of Surgery (11th Ed.), the equivalent standard reference for surgical MS examinations, covering identical core content at the same depth.
This is a shared conversation. Sign in to Orris to start your own chat.