Latest guidelines for management of hypertension in pregnancy
Reading File
Finding Sources
Searching PubMed
"hypertension" AND "pregnancy" AND "management"
Searching the Web
hypertension in pregnancy management guidelines 2024 2025 ACOG ISSHP
Reading File
Reading File
Searching PubMed
"hypertensive disorders of pregnancy" AND "guidelines"
Reading File
Reading Article
Reading Article
Reading File
Reading File
Reading File
Finding Sources
Loading Image
I now have all the data needed for a comprehensive answer. Here is the complete evidence-based summary.
Management of Hypertension in Pregnancy — Current Guidelines
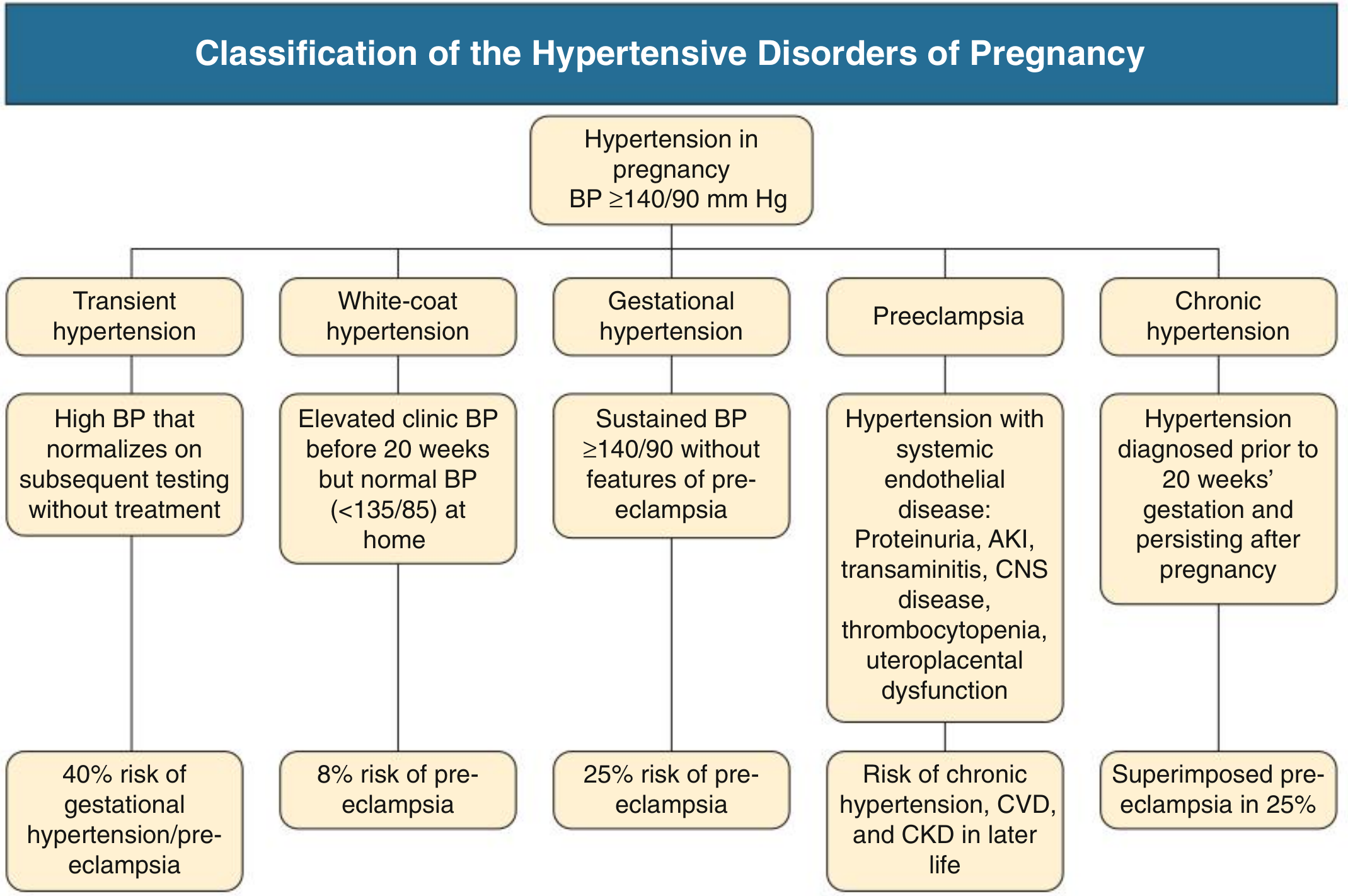
1. Classification
Hypertension in pregnancy is defined as BP ≥ 140/90 mmHg on two occasions. The disorders are classified as:
| Disorder | Definition | Progression risk |
|---|---|---|
| Transient hypertension | Elevated BP normalising on repeat testing without treatment | 40% risk of gestational HTN/preeclampsia |
| White-coat hypertension | Office BP ≥140/90, home BP <135/85 | 8% risk of preeclampsia |
| Gestational hypertension | New HTN after 20 weeks without features of preeclampsia | 25% progress to preeclampsia |
| Preeclampsia | New HTN after 20 weeks + systemic endothelial disease (see below) | Risk of future CKD, CVD |
| Chronic hypertension | HTN diagnosed before 20 weeks or persisting >12 weeks postpartum | 25% risk of superimposed preeclampsia |
Preeclampsia no longer requires proteinuria for diagnosis. It is defined as new HTN after 20 weeks with any of: proteinuria (uPCR >30 mg/mmol), AKI, elevated transaminases, CNS symptoms, thrombocytopenia, hepatic capsule distension, uteroplacental dysfunction (fetal growth restriction, abnormal umbilical artery Doppler, stillbirth).
Normal BP in pregnancy falls to a nadir of ~113/69 mmHg at 18–19 weeks; the upper reference limit is 136/86 mmHg in mid-pregnancy and 144/95 mmHg at term.
2. Blood Pressure Thresholds for Treatment
Severe Hypertension (SBP ≥ 160 or DBP ≥ 105 mmHg)
Antihypertensive therapy is clearly indicated to prevent stroke and cardiovascular events. Urgent treatment is required.
Mild-to-Moderate Hypertension (140–159/90–104 mmHg)
This is the area of greatest guideline divergence:
- The ACOG Task Force historically recommended against routine antihypertensive use in women with chronic HTN and BP <160/105 mmHg without end-organ damage, citing concerns that aggressive BP lowering may impair fetal growth via reduced uteroplacental perfusion.
- The CHIPS Trial (Control of Hypertension in Pregnancy Study) showed that treating to a tight DBP target of 85 mmHg versus less-tight control (DBP 100 mmHg) was safe — no difference in pregnancy loss or high-level neonatal care — but significantly reduced maternal complications (severe HTN, thrombocytopenia, transaminitis).
- Current guidance therefore supports treating to a target DBP of 85 mmHg in most women to reduce maternal risk without harming the fetus.
The 2025 Circulation review (Countouris et al., PMID 39960983) highlights that national and international guidelines still differ in treatment thresholds and targets, and calls for more trials to establish optimal BP ranges peripartum and postpartum.
3. Safe Antihypertensive Agents in Pregnancy
| Drug | Dose | Notes |
|---|---|---|
| Methyldopa (α₂-agonist) | 250 mg BD, titrate up | Longest safety record; first-line in many guidelines |
| Labetalol (α₁/β-blocker) | Oral: 100 mg BD; IV: 20–40 mg boluses | Widely used; good acute and chronic control |
| Nifedipine (CCB) | 30 mg OD (SR) | Effective; avoid sublingual formulation |
| Hydralazine | 5–10 mg IV/IM, repeat q20 min | Acute severe HTN; common in low-resource settings |
| Amlodipine | 5–10 mg OD | Increasingly used; CCB class |
Contraindicated Agents
- ACE inhibitors (captopril, enalapril) — teratogenic (renal dysgenesis, oligohydramnios, fetal hypocalvaria); contraindicated in all trimesters
- Angiotensin receptor blockers (ARBs) — same fetotoxic profile; contraindicated
- Spironolactone — theoretical risk of inadequate male fetal virilisation; avoid (eplerenone may be used if essential)
- Thiazide diuretics — generally avoided (reduce uteroplacental perfusion); use only if volume overload is a concern
4. Management of Specific Scenarios
Chronic Hypertension
- Control BP before conception using pregnancy-safe agents; switch from unsafe drugs (ACEi, ARB) as soon as pregnancy is confirmed
- Follow closely for superimposed preeclampsia (sudden BP rise in previously well-controlled patient, new proteinuria, thrombocytopenia, elevated liver enzymes, headache, visual changes)
- Screen for secondary causes: primary hyperaldosteronism, renal artery stenosis, phaeochromocytoma, obstructive sleep apnoea (≥40% of pregnant women with HTN have OSA)
Gestational Hypertension
- Monitor BP closely; ~25% will progress to preeclampsia
- Initiate antihypertensives if BP ≥ 140/90 mmHg persists
Preeclampsia — Non-Severe
- Hospitalise or manage with close outpatient surveillance
- Initiate antihypertensives; aim for BP <150/100 mmHg
- Serial fetal assessment (growth scans, umbilical artery Doppler)
- Plan delivery at 37 weeks or earlier if features of severity
Severe Preeclampsia / Eclampsia
- Acute BP control: Labetalol IV (20 mg, escalate to 40 mg at 10 min if insufficient) or Hydralazine IV 5–10 mg q20 min or Nifedipine oral (avoid sublingual)
- Seizure prophylaxis/treatment: Magnesium sulfate IV — documented efficacy for seizure prevention and treatment; no adverse maternal/fetal effects at therapeutic doses; continue for 24 h postpartum. Treat all women with severe preeclampsia or CNS manifestations (headache, visual disturbance, altered consciousness)
- ~20% of eclampsia occurs >48 h postpartum — magnesium coverage must extend into the postpartum period
- Definitive treatment: Delivery — if severe features are present and fetus is sufficiently mature. If preterm, hospitalisation ± corticosteroids for fetal lung maturation and temporising antihypertensive therapy may be used to gain fetal maturity
HELLP Syndrome
- Haemolysis, Elevated Liver enzymes, Low Platelets
- AKI incidence 10–25% in HELLP; urgent delivery indicated when AKI occurs
- Differentiate from TTP (plasmapheresis beneficial in TTP but not HELLP) and AFLP
5. Prevention of Preeclampsia
| Intervention | Evidence |
|---|---|
| Low-dose aspirin (75–150 mg/day), started before 16 weeks | Reduces preeclampsia risk by ~20% in high-risk women (ASPRE trial); recommended by ISSHP, NICE, ACOG |
| Calcium supplementation (1.5–2 g/day) | Reduces preeclampsia risk in calcium-deficient populations (Cochrane meta-analysis) |
| First-trimester combined screening (MAP, uterine artery PI, PlGF, PAPP-A) | Identifies high-risk women at 11–13 weeks for targeted aspirin prophylaxis |
High-risk features warranting aspirin: prior preeclampsia, chronic HTN, diabetes, CKD, antiphospholipid syndrome, SLE, obesity (BMI >30), multiple gestation, age >40, nulliparity with >1 moderate risk factor.
6. Postpartum Management
- BP may spike in the first 3–5 days postpartum even in women who were normotensive intrapartum
- Methyldopa, labetalol, nifedipine, and enalapril/captopril are compatible with breastfeeding
- ACEi/ARBs can be used postpartum (no longer teratogenic concern)
- All women with HDP should have cardiovascular risk review at 6–12 weeks postpartum — HDP is an independent risk factor for future hypertension, ischaemic heart disease, stroke, and CKD
- Home BP monitoring programs have been shown to improve BP ascertainment across diverse populations and lower BP in the months after delivery
7. Investigations at Diagnosis
- Urine protein:creatinine ratio (PCR) or albumin:creatinine ratio (ACR), 24-h urine protein
- FBC (thrombocytopenia in HELLP/preeclampsia)
- Serum creatinine, LFTs, uric acid
- Coagulation screen if HELLP suspected
- Placental growth factor (PlGF) — a low PlGF or high sFlt-1/PlGF ratio predicts preeclampsia with high accuracy and can be used to rule out preeclampsia in suspected cases
- Fetal ultrasound: growth biometry, umbilical artery Doppler
Key Evidence Updates (2023–2025)
- [PMID 39960983] Countouris et al., Circulation 2025: Reviews current standards and notes that guidelines still differ on treatment thresholds; highlights the value of postpartum HTN clinics and home BP monitoring for long-term cardiovascular risk reduction.
- [PMID 37391211] Wu et al., BMJ 2023: Comprehensive review confirming PlGF-based testing improves identification of high-risk preeclampsia; affirms aspirin prophylaxis and confirms delivery timing evidence for non-severe preeclampsia.
- CHIPS Trial (landmark RCT): Tight DBP target 85 mmHg is safe and reduces maternal complications without increasing fetal harm — supports more aggressive treatment of mild-to-moderate HTN than older guidelines suggested.
Sources: Brenner & Rector's The Kidney, 10th ed.; Comprehensive Clinical Nephrology, 7th ed.; Goodman & Gilman's Pharmacological Basis of Therapeutics, 13th ed.; Countouris et al., Circulation 2025 [PMID 39960983]; Wu et al., BMJ 2023 [PMID 37391211].
This is a shared conversation. Sign in to Orris to start your own chat.