Organophosphorus poisoning
Reading File
Finding Sources
Finding Sources
Searching PubMed
"organophosphate poisoning" AND management
Reading File
Reading File
Finding Sources
Reading File
Reading File
Loading Image
Reading File
I now have all the information needed for a comprehensive answer. Let me compile it:
Organophosphorus (Organophosphate) Poisoning
Epidemiology & Sources
Organophosphorus (OP) compounds are among the most common causes of pesticide-related deaths globally, with >150,000 pesticide poisoning deaths annually worldwide. Common agents include diazinon, acephate, malathion, parathion, and chlorpyrifos. Exposure occurs via:
- Deliberate self-poisoning (most common globally)
- Accidental agricultural/industrial exposure
- Contaminated food
- Chemical warfare agents (nerve agents: sarin, soman, VX) share the same mechanism
Routes of absorption: inhalation, dermal, transconjunctival, mucous membrane, GI
Mechanism
OPs irreversibly inhibit acetylcholinesterase (AChE) via phosphorylation of its active serine site → ACh accumulates at all cholinergic synapses → overwhelming stimulation of muscarinic and nicotinic receptors.
Aging: The OP-AChE bond undergoes permanent, irreversible stabilization ("aging") — timing varies from minutes (soman) to days (malathion). Once aging occurs, new enzyme synthesis is required (takes weeks). This is why antidotes must be given early.
Clinical Syndromes
Four syndromes follow OP exposure:
1. Acute Poisoning
Most patients are symptomatic within 8 hours, nearly all within 24 hours. Symptom onset is fastest with inhalation, slowest with transdermal absorption.
Cholinergic effects on the nervous system:
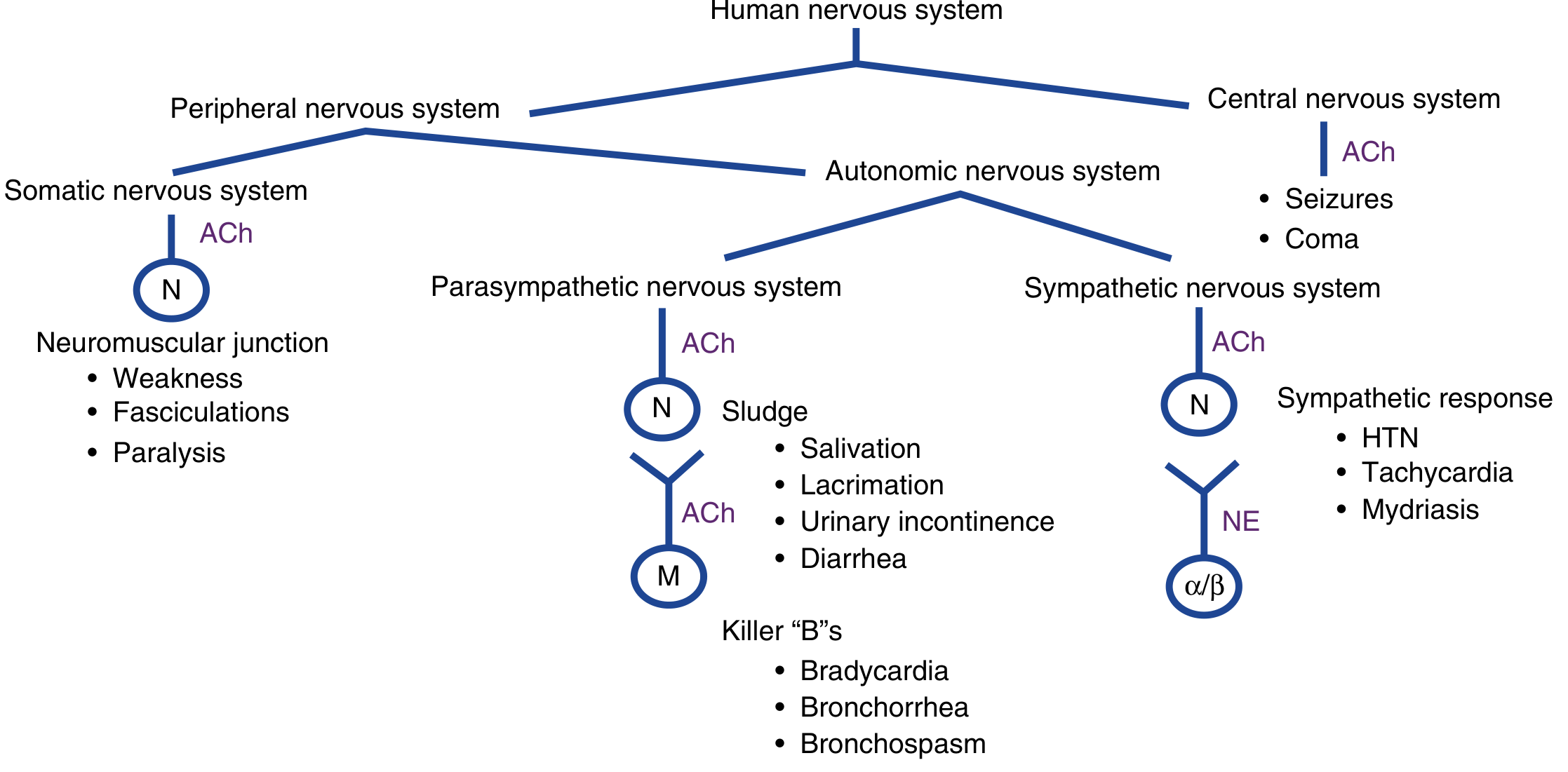
Muscarinic effects (mnemonic: SLUDGE/DUMBELS)
| SLUDGE | DUMBELS |
|---|---|
| Salivation | Defecation |
| Lacrimation | Urination |
| Urinary incontinence | Miosis |
| Defecation | Bradycardia, bronchorrhea, bronchospasm |
| GI cramps | Emesis |
| Emesis | Lacrimation |
| Salivation |
"Killer Bs" = Bradycardia, Bronchorrhea, Bronchospasm — the lethal triad
Nicotinic effects (NMJ & sympathetic ganglia)
- Muscle fasciculations, cramps, weakness (including diaphragm)
- Mydriasis, pallor
- Tachycardia, hypertension
CNS effects
- Anxiety, restlessness, emotional lability
- Tremor, headache, dizziness
- Confusion, delirium, hallucinations
- Seizures, coma
Cardiac
- ST-segment changes, peaked T waves, AV block, QTc prolongation
2. Intermediate Syndrome
- Occurs 24–96 hours after resolution of the cholinergic crisis
- Proximal limb weakness, neck flexor weakness, cranial nerve palsies, respiratory muscle paralysis
- Does not respond to atropine or pralidoxime
- Managed with mechanical ventilation
3. Organophosphate-Induced Delayed Polyneuropathy (OPIDP)
- Onset 2–3 weeks post-exposure
- Distal sensorimotor polyneuropathy ("dying-back" neuropathy)
- Due to inhibition of neuropathy target esterase (NTE)
- Not related to AChE inhibition; no treatment available
4. Chronic Toxicity
- Subclinical cognitive and neuropsychiatric symptoms from repeated low-level exposures (e.g., agricultural workers)
Diagnosis
Primarily clinical — based on history + cholinergic toxidrome. Confirmed by:
- RBC acetylcholinesterase activity (more specific; reflects synaptic AChE)
- Plasma pseudocholinesterase (butyrylcholinesterase) (more sensitive but less specific)
-
50% reduction in either is diagnostic; symptoms typically appear at ≥20–50% reduction
Management
Goals
- Decontamination
- Supportive care (airway priority)
- Reversal of ACh excess (atropine)
- Reversal of toxin-AChE binding (oximes)
Step 1 — Decontamination
- Remove and discard all clothing (dermal absorption risk to caregivers)
- Flush exposed skin copiously with water (primary method)
- Staff must use Level C PPE: full-face air-purifying respirator, chemical-resistant suit, nitrile/butyl rubber gloves
- Equipment (not skin) may be washed with 5% hypochlorite
- Activated charcoal and gastric lavage are not beneficial (rapid absorption + profuse emesis)
- No role for hemodialysis or enhanced elimination
Step 2 — Airway & Supportive Care
- Aggressive suction of secretions, oxygenation, mechanical ventilation if needed
- For RSI: prefer rocuronium 1 mg/kg (non-depolarizing, not metabolized by cholinesterases)
- ⚠️ If succinylcholine is used: expect prolonged paralysis 4–6 hours (AChE inhibited)
- Avoid β-blockers (may potentiate bradycardia)
- Seizures: benzodiazepines after airway secured
- Tachyarrhythmias: treat underlying cholinergic excess, do NOT treat symptomatically with β-blockers
Step 3 — Antidote Therapy
Atropine (Muscarinic antagonist — competitive)
| Parameter | Detail |
|---|---|
| Mechanism | Competitive antagonist at muscarinic receptors (does NOT reverse nicotinic/NMJ effects) |
| Initial dose (adult) | 1.2–3 mg IV (0.05 mg/kg in children); 2–6 mg IM if no IV access |
| Titration | Double dose every 5 minutes until endpoints achieved |
| Endpoints | ✅ Chest clear on auscultation ✅ HR >80 bpm ✅ SBP >80 mmHg ✅ Secretions dried |
| Not endpoints | Pupil dilation, tachycardia — do NOT use to stop dosing |
| Maintenance | 10–20% of total loading dose per hour as infusion |
| Total dose | May require 100s of mg in severe poisoning (up to 200–500 mg in first hour) |
⚠️ Tachycardia is NOT a contraindication to atropine — it often reflects bronchospasm/hypoxia which atropine reverses. Atropine does NOT reverse muscle paralysis — respiratory support is always required.
Glycopyrrolate (alternative): Does not cross the BBB, so avoids CNS toxicity. Dosing less well-defined; no proven benefit over atropine.
Pralidoxime (2-PAM) — Oxime (AChE reactivator)
| Parameter | Detail |
|---|---|
| Mechanism | Nucleophilic displacement of OP from AChE active site → enzyme reactivation |
| Effect | Reverses muscarinic + nicotinic + CNS effects; critically reverses muscle paralysis |
| Timing | Must be given before aging — ideally within hours of exposure |
| WHO dose | 30 mg/kg IV bolus → infusion of 8 mg/kg/hr |
| Duration | Continue 24–48 hours while monitoring AChE levels |
| Draw blood | Obtain AChE levels before administering if possible (do not delay for this) |
| Limitations | Current evidence insufficient to confirm mortality reduction (controversial) |
| Not indicated | Asymptomatic patients; isolated carbamate poisoning with minimal symptoms |
Key distinction: Pralidoxime is NOT useful for carbamate poisoning because carbamate–AChE binding is spontaneously reversible without oxime.
Step 4 — Adjuncts
- Benzodiazepines for seizures (first-line after atropine for cholinergic seizures)
- Magnesium sulfate and calcium channel blockers: some evidence of benefit, not routine
- Avoid: succinylcholine (prolonged paralysis), ester-type local anesthetics, β-blockers
Drugs to AVOID
| Drug | Reason |
|---|---|
| Succinylcholine | Metabolized by cholinesterases → prolonged block |
| β-blockers | Potentiate bradycardia |
| Ester-type local anesthetics | Hydrolyzed by cholinesterases → toxicity |
| Morphine, phenothiazines | May precipitate respiratory failure |
Carbamates vs. Organophosphates
| Feature | Organophosphates | Carbamates |
|---|---|---|
| AChE binding | Irreversible (aging) | Reversible (spontaneous) |
| Duration | Days–weeks | Hours |
| Oxime therapy | Indicated (if given early) | Not routinely indicated |
| Overall severity | Generally more severe | Generally milder |
Recent Evidence Note
A 2025 systematic review (PMID: 38797902) evaluated glycopyrrolate as an alternative to atropine in OP poisoning — findings support its use but dosing remains unstandardized, and it has not replaced atropine as first-line therapy.
Sources: Tintinalli's Emergency Medicine, 9e | Rosen's Emergency Medicine, 10e | Katzung's Basic & Clinical Pharmacology, 16e | Bradley & Daroff's Neurology in Clinical Practice | Morgan & Mikhail's Clinical Anesthesiology, 7e
This is a shared conversation. Sign in to Orris to start your own chat.