Octeriotide
Checking Date & Time
Reading File
Finding Sources
Searching PubMed
octreotide somatostatin analogue clinical uses
Reading File
Reading File
Reading File
Loading Image
Here is a comprehensive overview of octreotide (you may have meant "octreotide" — a common misspelling):
Octreotide
Structure & Mechanism
Octreotide is a synthetic octapeptide analogue of somatostatin (SST-14), engineered to retain the core pharmacophore of the native hormone while having a dramatically longer half-life.
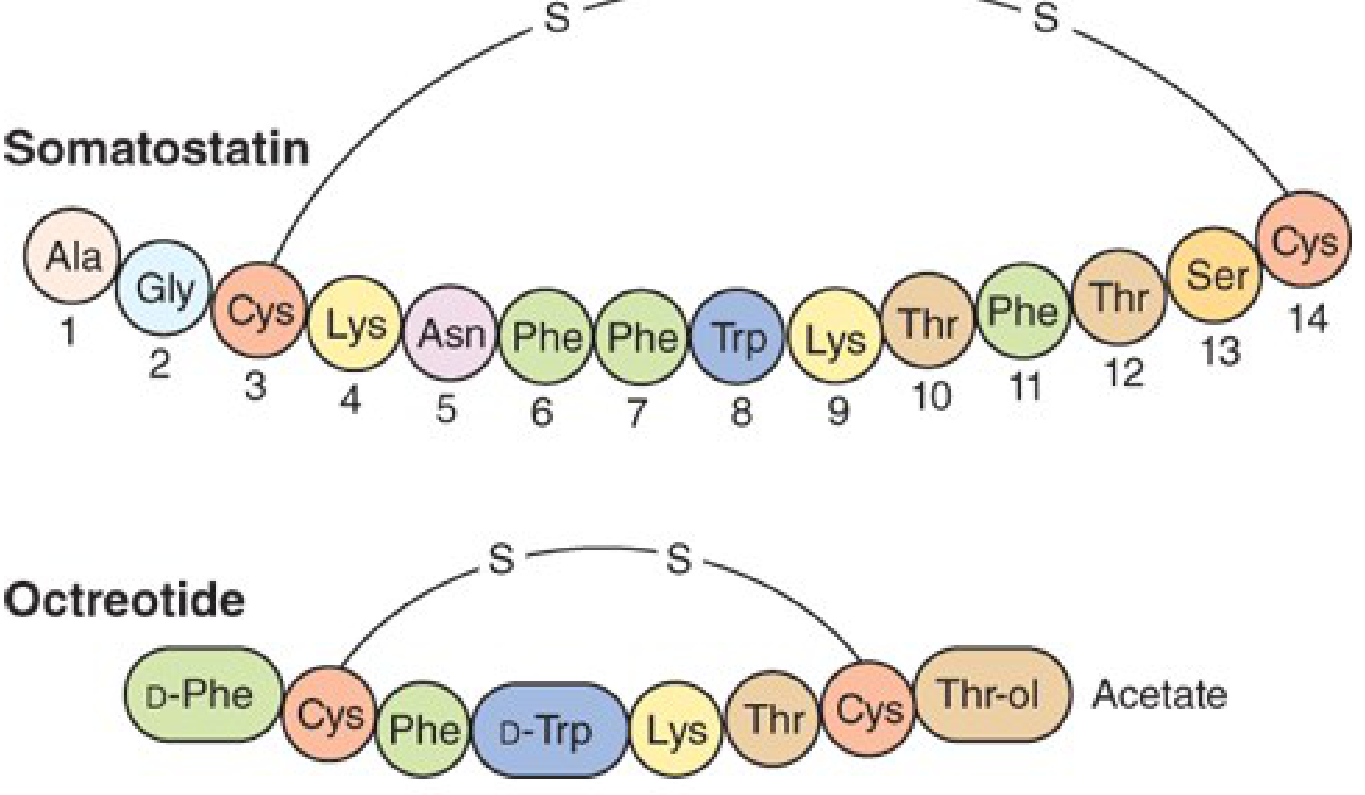
Key structural features:
- 8 amino acids (vs. 14 in native somatostatin)
- Contains D-Phe and D-Trp (D-amino acid substitutions that resist proteolysis)
- Maintains a disulfide bridge between two Cys residues
- Binds preferentially to SST receptor subtypes 2 and 5 (SSTR2, SSTR5)
Potency comparison vs. somatostatin:
- 45× more potent at inhibiting GH release
- Only 2× more potent at suppressing insulin secretion → much lower hypoglycemia risk
- Plasma t½ ~80–90 min (30× longer than somatostatin's 3-min t½)
Pharmacokinetics
| Parameter | Short-acting SC | Long-acting (LAR) |
|---|---|---|
| Route | Subcutaneous or IV | IM depot |
| Dose | 50–200 µg q8h | 10–40 mg q4 weeks |
| t½ (SC) | ~80–90 min | Sustained release |
| Duration of action | 6–12 h | ~4 weeks |
| Peak effect (SC) | ~30 min | — |
The LAR (long-acting release) formulation is a slow-release microsphere preparation injected into alternate gluteal muscles every 4 weeks. It is initiated only after short-acting octreotide has been shown to be effective and tolerated.
Pharmacological Actions
Somatostatin (and octreotide) suppress secretion across multiple systems:
- Pituitary: Inhibits GH and TSH release
- Pancreas: Inhibits insulin, glucagon, and exocrine secretion
- GI tract: Inhibits gastrin, secretin, motilin, VIP; reduces intestinal secretion
- Vascular: Reduces portal and splanchnic blood flow
- Tumors: Can reduce size of GH-secreting adenomas (though growth resumes when stopped)
Clinical Uses
1. Endocrine Tumors (Neuroendocrine)
- Acromegaly — reduces GH/IGF-1 levels; goal: GH <2.5 ng/mL after OGTT, IGF-1 within normal range for age/sex. Can also shrink adenoma size.
- Carcinoid tumors — controls secretory diarrhea, flushing, and wheezing
- VIPoma — suppresses watery diarrhea (WDHA syndrome)
- Glucagonoma, gastrinoma, insulinoma, ACTH-secreting tumors
2. GI Bleeding
- Esophageal variceal bleeding — reduces portal pressure and splanchnic flow; decreases all-cause mortality and transfusion requirements (used alongside endoscopy)
- IV infusion used acutely; t½ effect on portal pressure is not prolonged, so continuous infusion is preferred
3. Diarrhea
Dose-dependent effect on motility:
- Low doses (50 µg SC) → stimulates motility (used for intestinal pseudo-obstruction, SIBO, scleroderma)
- High doses (100–250 µg SC) → inhibits motility → treats:
- Secretory diarrhea (HIV-associated, chemotherapy/radiation-induced, diabetic)
- Dumping syndrome / post-vagotomy diarrhea
- Short-bowel syndrome
4. Other
- Pancreatic fistula — inhibits pancreatic secretion
- Somatostatin receptor scintigraphy — radiolabeled octreotide (octreoscan) localizes neuroendocrine tumors with SSTR and predicts treatment response
Adverse Effects
| System | Effect |
|---|---|
| GI | Nausea, vomiting, abdominal cramping, flatulence, steatorrhea (fat-soluble vitamin deficiency risk) |
| Biliary | Gallbladder sludge/stones in 20–30% at 1 year, up to 50% at 5 years (gallbladder contractility ↓); symptomatic gallstones ~1%/year |
| Glycemic | Hyperglycemia (more common) or mild hypoglycemia — due to altered insulin/glucagon/GH balance |
| Thyroid | Hypothyroidism with prolonged use |
| Cardiac | Sinus bradycardia (~25%), conduction disturbances (~10%) |
| Nutritional | Vitamin B12 deficiency with long-term use |
| Injection site | Pain, especially with LAR formulation |
Comparison with Other SST Analogues
| Drug | Receptor Selectivity | Formulation | Notes |
|---|---|---|---|
| Octreotide | SSTR2, SSTR5 | SC q8h or IM monthly (LAR) | Most widely used |
| Lanreotide | SSTR2, SSTR5 | Deep SC q4 weeks (autogel) | Comparable efficacy to octreotide LAR |
| Pasireotide | SSTR1,2,3,5 (esp. SSTR5) | SC or IM (LAR) | Greater biochemical control of acromegaly (31.3% vs. 19.2% vs. octreotide LAR); higher hyperglycemia risk (~50%) |
Sources: Katzung's Basic and Clinical Pharmacology, 16th Ed. — p. 1051, 1713 | Goodman & Gilman's Pharmacological Basis of Therapeutics — p. 1493–1495
Pls send infusion titration
Checking Date & Time
Asking for Preferences
Finding Sources
Searching the Web
octreotide IV infusion titration protocol variceal bleeding dose mcg/hour
Reading File
Reading File
Reading File
Here is the octreotide infusion titration protocol for variceal/acute GI bleeding:
Octreotide IV Infusion — Variceal Bleeding
Adults
| Step | Action | Dose |
|---|---|---|
| 1. Loading bolus | IV push over 3–5 min | 50 µg IV bolus |
| 2. Start infusion | Begin immediately after bolus | 50 µg/hour continuous IV infusion |
| 3. Duration | Continue for | 3–5 days after confirmed variceal bleeding |
- Initiate as soon as variceal bleeding is suspected — do not wait for endoscopic confirmation
- Combine with urgent endoscopy (variceal band ligation preferred; sclerotherapy if visualization is poor)
- No routine dose escalation described for adults — 50 µg/h is the standard maintenance rate
Pediatrics (weight-based)
| Step | Action | Dose |
|---|---|---|
| 1. Loading bolus | IV | 1–2 µg/kg (max 50 µg) |
| 2. Start infusion | Continuous IV | 1–2 µg/kg/hour |
| 3. Titration | Increase hourly by 1 µg/kg if bleeding continues | Up to max 4 µg/kg/hour |
- Monitor blood glucose closely — hyperglycemia is the main adverse effect in children
- If octreotide unavailable: vasopressin 0.002–0.005 unit/kg/min, titrated up to 0.01 unit/kg/min (consult pediatric GI)
- ICU admission required
Clinical Context & Mechanism
- Octreotide reduces splanchnic blood flow by inhibiting glucagon release → lowers portal pressure and azygos blood flow
- Effect on portal pressure is not prolonged despite continuous infusion — this is why it must run alongside endoscopic intervention, not replace it
- Comparable efficacy to terlipressin and sclerotherapy; preferred in the US (terlipressin not universally available)
- Long-acting (LAR) formulation is not effective for acute variceal bleeding
Monitoring
| Parameter | Frequency |
|---|---|
| Blood glucose | Every 4–6 hours (hyperglycemia or hypoglycemia risk) |
| Heart rate | Continuous (sinus bradycardia in ~25%) |
| BP | Routine monitoring |
| Bleeding status | Ongoing clinical assessment |
Sources: Mulholland & Greenfield's Surgery, 7th Ed. — AASLD 2016-based guidance | Tintinalli's Emergency Medicine — Pediatric dosing | Sleisenger & Fordtran's GI & Liver Disease | Rosen's Emergency Medicine
This is a shared conversation. Sign in to Orris to start your own chat.