Hyperplasia,polyp diagnosis,ectopic pregnancy treatment and abortion in detail
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
I now have comprehensive material from all four topics. Let me compose the detailed response.
1. Hyperplasia
Definition & Pathogenesis
Endometrial hyperplasia is defined as an abnormal proliferation of endometrial glands relative to the stroma, resulting in an increased gland-to-stroma ratio compared with normal proliferative endometrium. It is an important cause of abnormal uterine bleeding and a frequent precursor to endometrial carcinoma.
The fundamental driver is prolonged unopposed estrogenic stimulation, which may arise from:
- Obesity — peripheral conversion of androgens to estrogens in adipose tissue
- Menopause / Perimenopause — anovulatory cycles
- Polycystic ovarian syndrome (PCOS)
- Functioning granulosa cell tumors of the ovary
- Exogenous estrogen — unopposed estrogen replacement therapy
- Excessive cortical stromal function (cortical stromal hyperplasia)
Molecular basis: Inactivation of the PTEN tumor suppressor gene is the key early molecular event, found in >20% of endometrial hyperplasias. PTEN normally suppresses the PI3K/AKT pathway; loss of PTEN causes overactivation of this growth pathway and enhances estrogen receptor–driven gene expression, promoting endometrial overgrowth. Patients with Cowden syndrome (germline PTEN mutation) have high rates of endometrial carcinoma.
Classification (WHO / EIN System)
| WHO Category | EIN Equivalent | Malignant Potential | Treatment |
|---|---|---|---|
| Hyperplasia without atypia (simple or complex) | Benign endometrial hyperplasia | ~1–3% | Medical (progestins) |
| Atypical hyperplasia (simple or complex) | EIN (Endometrial Intraepithelial Neoplasia) | ~30% progression; ~40–50% concurrent carcinoma | Surgical (preferred) or medical |
| — | Endometrial adenocarcinoma, well-differentiated | Malignant | Surgical + staging |
The ACOG/SGO 2015 recommendation replaced the old WHO 4-category system with the 3-tier EIN schema to better guide clinical management.
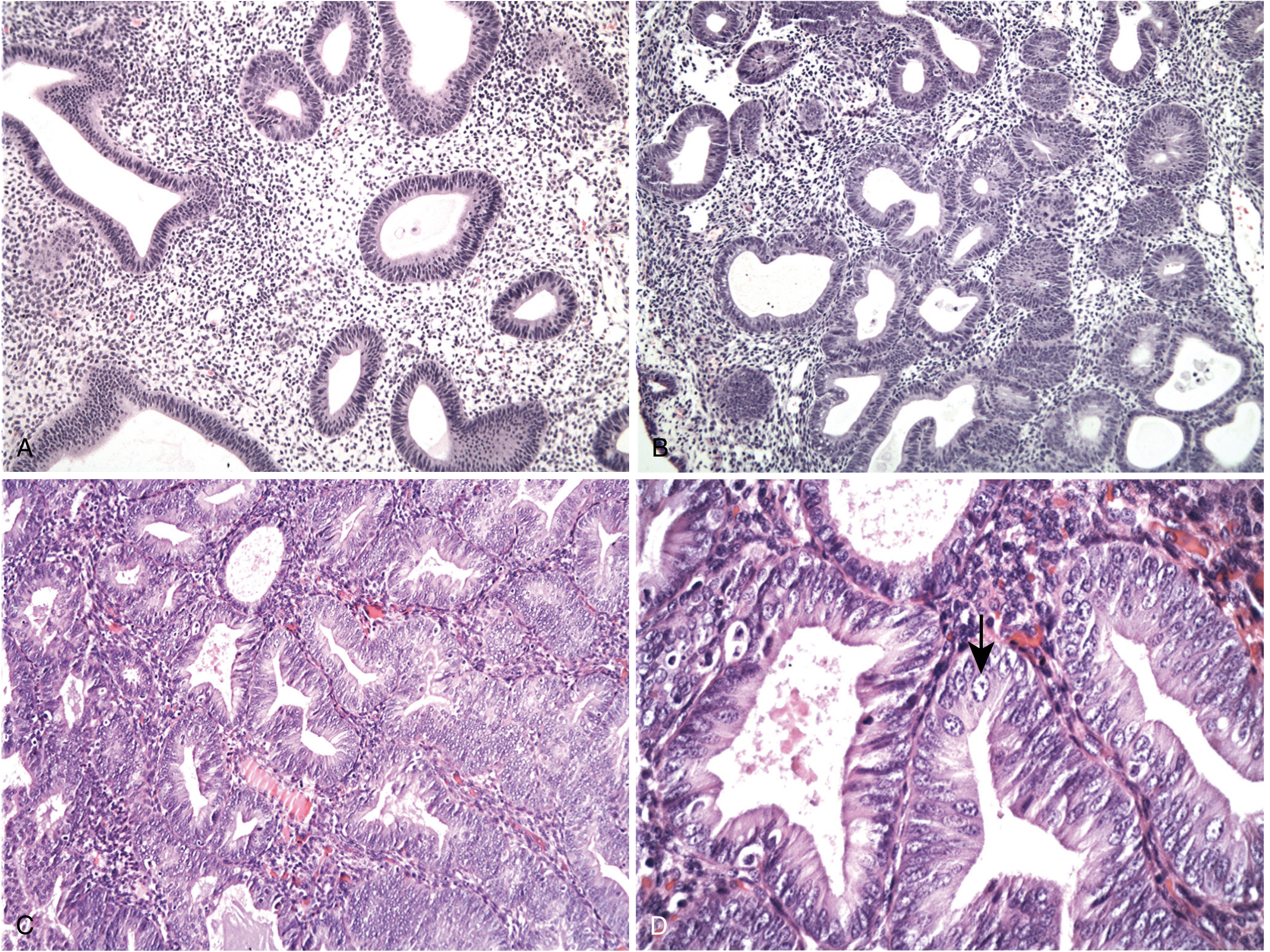
Morphology
Hyperplasia without atypia:
- Increased gland-to-stroma ratio with variation in gland size and shape
- May show cystic dilation or back-to-back glands focally, but some intervening stroma is retained
- Cytologic features similar to normal proliferative endometrium
- Low malignant potential (~1–3%)
Atypical hyperplasia / EIN:
- Complex patterns of glands with nuclear atypia — rounded, vesicular nuclei with prominent nucleoli
- Back-to-back glands, markedly reduced stroma
- High malignant potential; often concurrent with carcinoma
Management
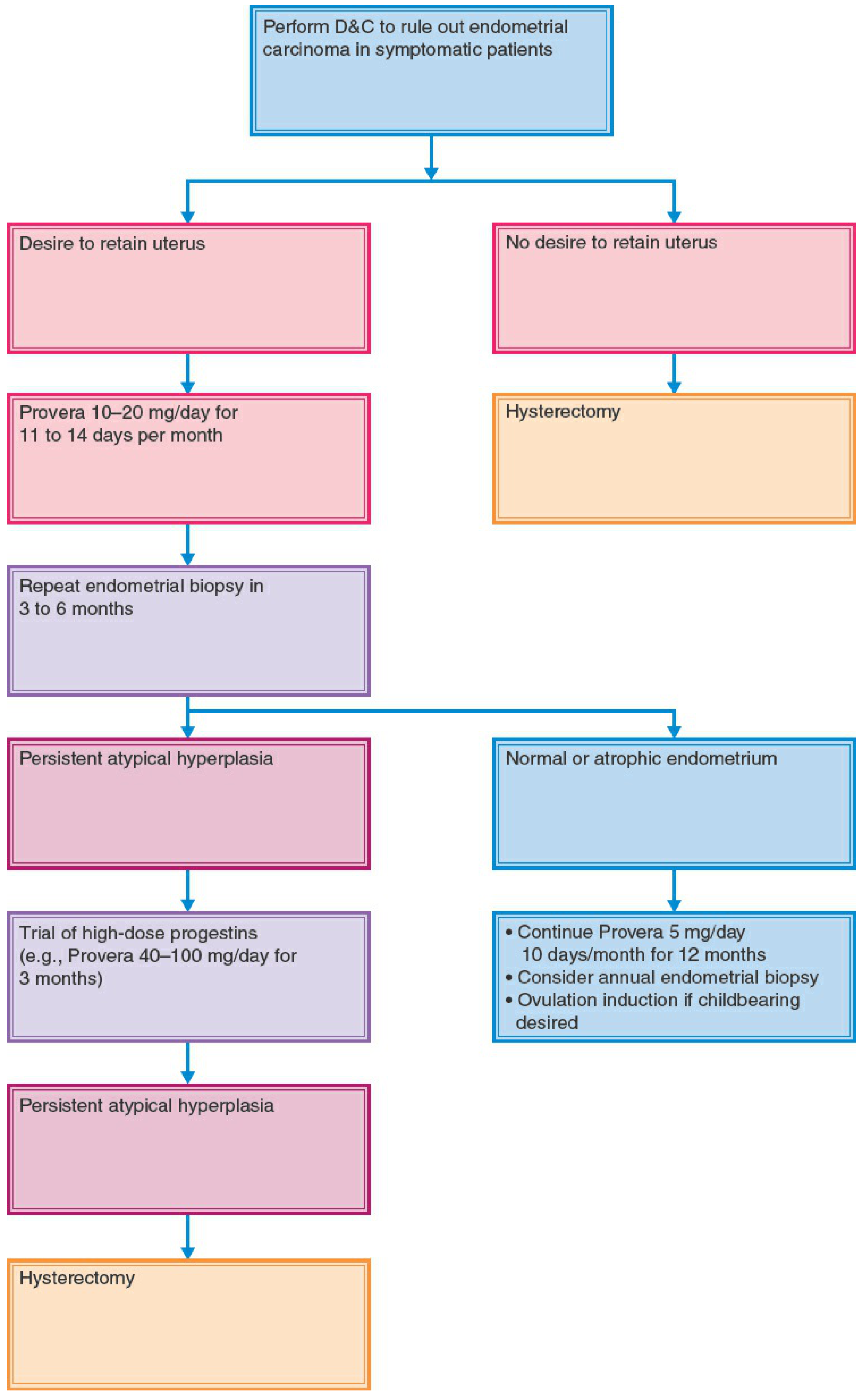
Hyperplasia without atypia (benign):
- Progestin therapy — oral (medroxyprogesterone acetate), parenteral, or levonorgestrel IUD
- Follow-up endometrial biopsy every 3 months
- Risk of progression is low; many regress
Atypical Hyperplasia / EIN:
- Hysterectomy is the definitive treatment, especially in postmenopausal women (rules out concurrent carcinoma)
- Progestin therapy (oral, parenteral, or LNG-IUD) may be used in women who desire fertility or are poor surgical candidates — with close follow-up (biopsy every 3 months); recurrence risk ~50%
- After successful pregnancy, lack of regression should prompt hysterectomy
2. Polyp Diagnosis
Types of Clinically Relevant Polyps
Endometrial Polyps
Definition: Benign focal overgrowths of endometrial glands and stroma projecting into the uterine cavity, often containing a central vascular stalk.
Clinical features:
- Most are asymptomatic (incidental finding)
- Can cause intermenstrual bleeding, heavy menstrual bleeding, irregular bleeding, postmenopausal bleeding, and dysmenorrhea
- Associated with tamoxifen use and infertility
- Incidence increases with age throughout reproductive years; prevalence ~5.8% in premenopausal women, ~11.8% in postmenopausal women
Diagnosis:
- Transvaginal ultrasound (TVUS): Suspected when endometrial thickening is seen; vascular "feeder vessel" pattern on Doppler helps distinguish polyps from fibroids and malignancy. Note: endometrial thickening is NOT specific — hyperplasia, polyp, and carcinoma all cause thickening.
- Saline infusion sonohysterography (SIS/SHG): Confirmation with saline contrast improves visualization of intracavitary lesions.
- Hysteroscopy: Gold standard — allows direct visualization and simultaneous removal.
- Endometrial biopsy / D&C: Tissue sampling for histologic confirmation.
- Hysterosalpingogram (HSG): Can detect filling defects.
Malignant potential:
- In premenopausal women: premalignant change 0.2–24%, malignancy 0–13%
- Higher risk in postmenopausal women with bleeding
- Up to 2.5% of polyps may harbor foci of endometrial carcinoma
- Spontaneous regression occurs in ~27% at 1 year (more likely for smaller polyps)
Management:
- Hysteroscopic polypectomy — treatment of choice; simultaneous diagnosis and treatment
- Removal may improve fertility in infertile patients
- Observation acceptable for small, asymptomatic polyps in premenopausal women
Cervical Polyps
- Pedunculated protrusions from the cervical canal; usually benign
- Cause: abnormal bleeding (postcoital, intermenstrual), vaginal discharge
- Postmenopausal cervical polyps frequently coexist with endometrial polyps; women on tamoxifen with cervical polyps have significantly higher risk of concomitant endometrial polyps
- Diagnosis: Direct visualization; tissue sent for histology
- Management: Simple avulsion/twisting at the base; hysteroscopy recommended when endometrial polyps are suspected
3. Ectopic Pregnancy — Treatment
Definition & Epidemiology
Implantation of a fertilized ovum outside the uterine cavity (most commonly the fallopian tube). Incidence: 0.64–2.0% of pregnancies. Most common obstetric cause of maternal death in the first trimester. In a series of 300 cases, 50% of patients had been evaluated at least twice before correct diagnosis.
Clinical Presentation (Classic Triad)
- Pelvic/abdominal pain
- Amenorrhea
- Abnormal vaginal bleeding
Also: adnexal mass, cervical tenderness, shoulder pain (diaphragmatic irritation from hemoperitoneum), syncope, back pain, anemia. Bradycardia with significant intraperitoneal bleeding is not unusual.
Risk Factors
- Prior salpingitis / PID
- Previous ectopic pregnancy
- Intrauterine contraceptive device (IUD)
- Tubal ligation
- Assisted reproductive technology (ART)
Diagnosis
- Serum β-hCG + transvaginal ultrasound (first-line)
- Discriminatory zone: when β-hCG >1,500–2,000 mIU/mL, an intrauterine gestational sac should be visible; absence suggests ectopic
- Culdocentesis: aspiration of non-clotting blood (>2 mL, hematocrit >3%) from the cul-de-sac indicates hemoperitoneum; positive in ~70% of proven ectopics
Treatment
A. Surgical Treatment
Indications:
- Hemodynamically unstable patient
- Ruptured ectopic
- Contraindications to methotrexate
- Failed medical management
Options:
- Salpingectomy (linear) — removal of the affected tube
- Lower risk of repeat ectopic (4% vs 10% with salpingostomy)
- Standard when contralateral tube is normal
- Salpingostomy (linear salpingotomy) — incision over the pregnancy, trophoblast removed, tube preserved
- Higher subsequent intrauterine pregnancy rate (RR 1.24) but more repeat ectopics
- Preferred when contralateral tube is damaged
- Laparoscopy is preferred over laparotomy (less morbidity, faster recovery)
B. Medical Treatment — Methotrexate
Methotrexate is a folic acid analog that inhibits dihydrofolate reductase, preventing DNA synthesis in rapidly dividing cells (trophoblast, bone marrow, intestinal mucosa). It is the first-line medical treatment for eligible patients.
~35% of ectopic pregnancies are candidates for methotrexate.
Candidate criteria (for methotrexate):
- Hemodynamically stable
- No evidence of rupture
- Unruptured tube <4 cm (relative contraindication if ≥4 cm)
- No fetal cardiac activity on ultrasound (relative contraindication if present)
Absolute contraindications:
| Contraindication |
|---|
| Intrauterine pregnancy |
| Hemodynamically unstable patient |
| Ruptured ectopic pregnancy |
| Breastfeeding |
| Hepatic, renal, or hematologic disorders |
| Known sensitivity to methotrexate |
| Immunodeficiency |
Dosing regimens:
- Single-dose: MTX 50 mg/m² IM — simpler, less toxic; β-hCG checked on days 4 and 7; if <15% decline, second dose given
- Multi-dose: MTX 1 mg/kg IM alternating with leucovorin (folinic acid rescue) — more effective for higher β-hCG levels but more side effects
- Two-dose protocol — intermediate option
Pre-treatment work-up:
- Serum β-hCG, CBC with differential, liver function tests, creatinine, blood type & screen
- Chest X-ray if pulmonary disease history
- Administer RhoGAM if Rh-negative
Patient instructions:
- Avoid alcohol, NSAIDs, folic acid-containing multivitamins, and sexual intercourse until β-hCG is negative
- Report severe/prolonged pain (lower abdominal pain is normal for 10–14 days)
- Report heavy vaginal bleeding
Follow-up:
- Weekly β-hCG until non-pregnant levels
- Tubal rupture can still occur even with falling β-hCG
- Cul-de-sac fluid is a common finding; does not mandate surgery unless hemodynamic instability or significant hematocrit drop occurs
- ~4–5% of women require surgery after failed methotrexate
Side effects (dose/frequency-dependent):
- GI: nausea, vomiting, stomatitis, abdominal pain (30–40%)
- Bone marrow suppression, alopecia, elevated liver enzymes, pneumonitis, dermatitis
- Leucovorin reduces side effects in multi-dose regimens
C. Expectant Management
- Considered only for very select patients with low, declining β-hCG (<1,000 mIU/mL), small ectopic, no pain, and strict compliance for close monitoring
- Risk of rupture persists
4. Abortion — In Detail
Definition & Types
Abortion = termination of pregnancy before viability (~20–24 weeks). Types:
- Spontaneous abortion (miscarriage) — involuntary loss
- Induced abortion — intentional termination (elective or therapeutic)
- Unsafe abortion — performed without necessary skills or in inadequate conditions
Epidemiology
- ~56.3 million abortions/year globally (2010–2014); 25% of all pregnancies
- ~25 million unsafe abortions annually; 47,000 women die per year from unsafe abortion
- In the US, ~88% of legal abortions occur in the first trimester (<13 weeks)
- Mortality rate for legal abortion: 0.7 per 100,000 procedures vs. maternal mortality 8.8 per 100,000 live births
Safety
- Risk increases exponentially with gestational age
- Aspiration abortion at ≤8 weeks: mortality 0.3/100,000 → rises to 6.7/100,000 at ≥18 weeks
- Even late D&E (>18 weeks) is safer than continuing pregnancy (maternal mortality comparison)
- Legal abortion is extremely safe; illegal abortion carries high complication and death rates
First-Trimester Methods (<13 weeks)
1. Vacuum Curettage (Aspiration Abortion)
- Most common method for first-trimester abortion
- Performed under local anesthesia ± moderate sedation; usually outpatient
- Cervical preparation: metal/plastic dilators OR misoprostol 400 μg vaginally/buccally 3–4 hours before
- Cannula: 5–12 mm plastic vacuum cannula with manual (modified 50-mL syringe, effective through 10 weeks) or electric pump
- Antibiotic prophylaxis: single dose doxycycline (taken night before procedure)
- Major complications (requiring hospitalization, surgery, or transfusion): 0.16%
- Incomplete abortion 0.33%
- Hemorrhage 0.13%
- Infection 0.27%
- Uterine perforation 0.01%
2. Medical Abortion (Medication Abortion)
Mifepristone + Misoprostol — FDA-approved, gold standard regimen:
| Step | Drug | Dose & Route |
|---|---|---|
| Day 1 | Mifepristone | 200 mg orally |
| Day 1–3 (24–48 h later) | Misoprostol | 800 μg buccally (or vaginally/sublingually) |
- Approved up to 70 days from last menstrual period (off-label evidence supports full first trimester)
- Efficacy: ~97% in early first trimester; slightly higher at earlier gestational ages
- Mifepristone: progesterone receptor antagonist — blocks progesterone, sensitizes uterus to prostaglandins, causes decidual breakdown
- Misoprostol: PGE1 analog — causes uterine contractions and cervical softening
- Main effects/side effects: Uterine cramping, bleeding, nausea, vomiting, diarrhea
- Serious adverse events: ~0.3% (excess bleeding requiring transfusion; infection even rarer)
- Mortality risk: 0.00063% — 14 times lower than risk of live birth
Absolute contraindications to mifepristone/misoprostol:
- Ectopic pregnancy (treat separately)
- IUD in place (remove first)
- Chronic adrenal failure
- Long-term corticosteroid therapy
- Allergy to mifepristone, misoprostol, or prostaglandins
- Hemorrhagic disorder
- Inherited porphyria
Complications of medical abortion:
- Heavy/prolonged bleeding: up to 8% bleed for ≥30 days
- Need for surgical curettage: 2% at ≤49 days, 3% at 50–56 days, 5% at 57–63 days
- Infection (rare); Clostridium sordellii deaths prompted prophylactic doxycycline use and switch from vaginal to buccal misoprostol
Alternative regimens (when mifepristone unavailable):
- Methotrexate + misoprostol — effective but slower
- Misoprostol alone — 800 μg vaginally, repeated at 24 h if needed; ~91% efficacy up to 56 days
Second-Trimester Methods (13–24 weeks)
1. Dilation and Evacuation (D&E)
Most commonly used midtrimester method in the US.
Cervical preparation (essential prior to D&E):
- Osmotic dilators:
- Laminaria japonica (seaweed stems) or Dilapan-S hydrophilic rods inserted into cervical canal
- Absorb water → swell → mechanical dilation + endogenous prostaglandin synthesis
- Removed the following day; sufficient dilation achieved for specialized forceps + large-bore vacuum cannula
- Misoprostol — can replace osmotic dilators; 1,200 μg multidose regimen reported with <2% major complications in experienced hands
- Mifepristone — as adjunct or replacement for osmotic dilators; no difference in complications vs. osmotic dilators + misoprostol
Procedure: Combination of:
- Cervical dilation + specialized forceps to remove fetal parts
- Large-bore vacuum cannula to evacuate placenta
- Ultrasound guidance is helpful
Intact D&E (for late second trimester):
- Wide cervical dilation → membrane rupture → assisted breech delivery → decompression of after-coming fetal head → intact delivery of fetus
- Feticidal agents often used before late second-trimester procedures
Feticidal agents:
- Intra-amniotic or intrafetal digoxin — 1.5 mg intra-amniotic; induces fetal demise within 24 hours
- Intracardiac potassium chloride (KCl) — both are efficacious with minimal maternal adverse effects
2. Labor Induction Methods
For late midtrimester, labor-induction protocols include:
- Serial osmotic dilators + misoprostol to induce labor → assisted expulsion
- High-dose oxytocin protocols
Summary Table
| Topic | Key Points |
|---|---|
| Hyperplasia | Unopposed estrogen → PTEN mutation → glandular overgrowth; EIN (atypical) has 30% cancer progression; treat with progestins or hysterectomy |
| Polyp Diagnosis | TVUS (screening) → sonohysterography → hysteroscopy (gold standard); coexistent endometrial + cervical polyps common with tamoxifen |
| Ectopic Pregnancy Tx | Methotrexate IM (medical, if stable/unruptured/≤4 cm); laparoscopic salpingectomy (surgical); β-hCG monitoring weekly |
| Abortion | First trimester: mifepristone 200 mg + misoprostol 800 μg (97% effective, ≤70 days); vacuum aspiration (most common surgical); Second trimester: D&E with cervical preparation |
Sources: Robbins & Cotran Pathologic Basis of Disease (9th ed.); Berek & Novak's Gynecology; Roberts and Hedges' Clinical Procedures in Emergency Medicine; Goldman-Cecil Medicine (International Ed.)
Make osce case on the above topic
Here are comprehensive OSCE cases covering all four topics:
🏥 OSCE CASES: Hyperplasia, Polyp, Ectopic Pregnancy & Abortion
CASE 1 — Endometrial Hyperplasia
Station Information
Time allowed: 10 minutes
Setting: Gynaecology outpatient clinic
Candidate instructions: You are a junior doctor. Please take a focused history, interpret the investigations provided, and discuss management with the examiner.
Patient Vignette
Mrs. Fatima, 52 years old, presents with irregular, heavy vaginal bleeding for 6 months. She is postmenopausal (LMP 2 years ago) and had started hormone replacement therapy (estrogen alone) 18 months ago for menopausal symptoms without consulting a specialist. She has a BMI of 34 kg/m² and was diagnosed with type 2 diabetes 5 years ago.
History to Elicit
| Domain | Expected Findings |
|---|---|
| Presenting complaint | Heavy, irregular PV bleeding; no pain |
| HRT history | Estrogen-only HRT for 18 months |
| Menstrual history | Postmenopausal × 2 years |
| Past medical history | T2DM, obesity, hypertension |
| Family history | Mother had endometrial cancer |
| Drug history | Metformin, amlodipine, estrogen HRT |
| Social history | Non-smoker, married, 2 children |
Examination Findings
- BMI: 34 kg/m²
- Abdomen: Soft, no masses, no tenderness
- Speculum: Atrophic changes; minimal blood-stained discharge
- Bimanual: Uterus slightly bulky, no adnexal masses
Investigations Provided
| Investigation | Result |
|---|---|
| Transvaginal ultrasound (TVUS) | Endometrial thickness 14 mm (>4 mm postmenopausal = abnormal) |
| Endometrial biopsy (Pipelle) | Atypical hyperplasia / EIN |
| CBC | Hb 9.8 g/dL (microcytic) |
| HbA1c | 8.1% |
| CA-125 | 28 U/mL (normal) |
OSCE Questions & Model Answers
Q1. What is the most likely diagnosis and what is the classification of this condition?
Endometrial Hyperplasia with Atypia (EIN — Endometrial Intraepithelial Neoplasia). Using the WHO/EIN classification:
- Benign endometrial hyperplasia (no atypia) — low malignant risk (~1–3%)
- EIN / Atypical hyperplasia — premalignant; ~30% progression to carcinoma; ~40–50% may have concurrent carcinoma
- Endometrial adenocarcinoma
Q2. What are the risk factors in this patient?
- Unopposed estrogen HRT (no progestogen added)
- Obesity (peripheral androgen → estrogen conversion)
- Type 2 diabetes (hyperinsulinemia stimulates estrogenic activity)
- Postmenopausal state
- Family history of endometrial cancer
Q3. What is the molecular basis of endometrial hyperplasia?
PTEN tumor suppressor gene inactivation — found in >20% of hyperplasias. PTEN normally inhibits the PI3K/AKT signaling pathway. When PTEN is lost, PI3K/AKT becomes overactive and enhances estrogen receptor-driven gene expression, promoting endometrial overgrowth. Cowden syndrome (germline PTEN mutation) carries high risk of endometrial carcinoma.
Q4. What is the definitive management for this postmenopausal patient?
Total hysterectomy is the definitive treatment — it removes the risk of concurrent carcinoma (present in 40–50% of EIN cases) and prevents progression. Bilateral salpingo-oophorectomy is also typically performed. Surgical staging may be indicated if carcinoma is found.
Q5. If this were a 28-year-old woman desiring fertility, how would you manage differently?
- Progestin therapy — levonorgestrel IUD (Mirena, first choice) or oral medroxyprogesterone acetate / norethisterone
- Endometrial biopsy every 3 months to monitor response
- Recurrence risk ~50%; once pregnancy achieved or regression fails → hysterectomy
- Multidisciplinary discussion with oncology and reproductive medicine
Q6. What histological features distinguish EIN from benign hyperplasia?
- Nuclear atypia: Rounded, vesicular nuclei with prominent nucleoli
- Back-to-back glands with minimal intervening stroma
- Loss of normal gland architecture
- Increased mitotic activity
Marking Scheme
| Domain | Marks |
|---|---|
| Relevant history (risk factors, HRT, family Hx) | /4 |
| Correct interpretation of TVUS + biopsy | /3 |
| Correct diagnosis + classification | /3 |
| Management — hysterectomy rationale | /4 |
| Fertility-sparing alternative with follow-up | /3 |
| Molecular pathology (PTEN) | /2 |
| Communication & professionalism | /1 |
| Total | /20 |
CASE 2 — Polyp Diagnosis
Station Information
Time allowed: 8 minutes
Setting: Gynaecology outpatient clinic
Candidate instructions: Review the history and investigations below. Answer the examiner's questions and counsel the patient about her diagnosis.
Patient Vignette
Mrs. Aisha, 44 years old, presents with intermenstrual bleeding and occasional post-coital bleeding for 4 months. Her periods are regular (28-day cycle, 5 days, moderate flow). She is on tamoxifen following breast cancer surgery 2 years ago. She has no other complaints. She is nulliparous and would like to conceive in the future.
Investigations Provided
| Investigation | Result |
|---|---|
| TVUS | Endometrial thickness 12 mm; a 1.5 cm echogenic lesion with a vascular pedicle (feeder vessel) in the uterine cavity |
| Saline infusion sonohysterography | 1.5 cm intracavitary polypoid lesion, well-defined |
| Cervical smear | Normal |
| Pregnancy test | Negative |
| Endometrial biopsy (pipelle) | Insufficient sample |
OSCE Questions & Model Answers
Q1. What is the most likely diagnosis?
Endometrial polyp (AUB-P under the PALM-COEIN classification of abnormal uterine bleeding).
Q2. What is the gold standard investigation for confirming this diagnosis?
Hysteroscopy — allows direct visualization of the uterine cavity and simultaneous removal of the polyp (hysteroscopic polypectomy). Histological analysis of the removed tissue confirms diagnosis and excludes malignancy.
Q3. Why is this patient at increased risk of endometrial polyps?
Tamoxifen — a selective estrogen receptor modulator (SERM) used in breast cancer treatment. It acts as an estrogen antagonist in breast tissue but has partial estrogen agonist effects on the endometrium, leading to endometrial proliferation, polyps, and increased risk of endometrial carcinoma. Women on tamoxifen with a cervical polyp should also be evaluated for concurrent endometrial polyps.
Q4. What is the PALM-COEIN classification? Where does polyp fit?
PALM-COEIN classifies causes of abnormal uterine bleeding:
- PALM (Structural): Polyp, Adenomyosis, Leiomyoma, Malignancy + Hyperplasia
- COEIN (Non-structural): Coagulopathy, Ovulatory dysfunction, Endometrial, Iatrogenic, Not yet classified Polyp = AUB-P
Q5. What is the malignant potential of endometrial polyps?
- Premenopausal women: premalignant change 0.2–24%; malignancy 0–13%
- Postmenopausal women with bleeding: higher risk
- Overall: up to 2.5% may harbor endometrial carcinoma
- This patient's tamoxifen use further increases her risk → all polyps in tamoxifen users should be removed and sent for histology
Q6. This patient wants to conceive. Does the polyp need to be removed?
Yes. Evidence suggests that hysteroscopic polypectomy improves pregnancy rates in infertile patients. Even asymptomatic polyps should be removed in women desiring conception. It also provides tissue for histology to exclude malignancy, which is important given her tamoxifen use.
Q7. What is the difference between endometrial and cervical polyps?
| Feature | Endometrial Polyp | Cervical Polyp |
|---|---|---|
| Origin | Endometrial glands + stroma | Endocervical epithelium |
| Symptoms | Intermenstrual, HMB, postmenopausal bleeding | Postcoital, intermenstrual bleeding, discharge |
| Diagnosis | TVUS, SIS, Hysteroscopy | Direct visualization (speculum exam) |
| Management | Hysteroscopic polypectomy | Avulsion / twisting at base |
| Association | Tamoxifen, obesity, HRT | Often coexist with endometrial polyps |
Marking Scheme
| Domain | Marks |
|---|---|
| Correct diagnosis (AUB-P) | /2 |
| PALM-COEIN classification | /3 |
| Tamoxifen link explained | /3 |
| Gold standard investigation | /2 |
| Malignant potential discussion | /3 |
| Fertility implications | /2 |
| Endometrial vs cervical polyp differentiation | /3 |
| Communication | /2 |
| Total | /20 |
CASE 3 — Ectopic Pregnancy
Station Information
Time allowed: 10 minutes
Setting: Emergency Gynaecology Unit
Candidate instructions: A 27-year-old woman is brought to the emergency department. Assess her, interpret investigations, and outline management.
Patient Vignette
Miss Zara, 27 years old, presents with sudden-onset sharp right iliac fossa pain for 6 hours, associated with light vaginal bleeding for 3 days. She reports 6 weeks of amenorrhea. She feels dizzy and has had one episode of vomiting. She has a history of pelvic inflammatory disease (PID) 2 years ago and currently uses a copper IUD for contraception.
Examination Findings
| Finding | Result |
|---|---|
| BP | 94/60 mmHg |
| Pulse | 112 bpm |
| Temperature | 36.8°C |
| Abdomen | Right iliac fossa tenderness, guarding |
| Speculum | Small amount of dark blood; IUD strings visible |
| Bimanual | Marked cervical motion tenderness; right adnexal tenderness; no definite mass palpable |
Investigations
| Investigation | Result |
|---|---|
| Urine pregnancy test | Positive |
| Serum β-hCG | 3,800 mIU/mL |
| Transvaginal ultrasound | No intrauterine gestational sac; 2.8 cm right adnexal mass with ring-of-fire sign; moderate free fluid in pouch of Douglas |
| Hb | 9.2 g/dL |
| Blood group | O Rh-negative |
OSCE Questions & Model Answers
Q1. What is the diagnosis? Justify with the investigations.
Ruptured right tubal ectopic pregnancy. Evidence:
- Positive pregnancy test + amenorrhea = pregnant
- β-hCG 3,800 > discriminatory zone (1,500–2,000) → intrauterine sac should be visible if IUP
- No intrauterine gestational sac on TVUS
- Right adnexal mass with "ring of fire" (vascular trophoblastic ring on Doppler)
- Free fluid in pouch of Douglas = hemoperitoneum
- Hemodynamic instability (BP 94/60, HR 112) = ruptured
Q2. What is your immediate management?
EMERGENCY — Stabilize and take to theatre immediately:
- IV access × 2 large-bore cannulae
- IV fluid resuscitation (crystalloid bolus; cross-match 4 units blood)
- Oxygen — high-flow
- Urgent bloods: FBC, U&E, coagulation, group & crossmatch
- Inform anaesthetics and theatre — emergency laparoscopy / laparotomy
- Administer Anti-D immunoglobulin (patient is Rh-negative)
- Remove IUD intraoperatively
Q3. What surgical options are available?
- Laparoscopic salpingectomy (preferred) — remove the affected tube; lower repeat ectopic rate (4% vs 10%)
- Laparoscopic salpingostomy — incise tube, remove trophoblast, preserve tube; preferred if contralateral tube is damaged; higher subsequent IUP rate (RR 1.24) but higher repeat ectopic rate
- Laparotomy — if laparoscopy unavailable or patient too unstable
Q4. Would methotrexate be appropriate here? Why or why not?
No. Absolute contraindications to methotrexate in this patient:
- Hemodynamically unstable (BP 94/60, HR 112)
- Evidence of rupture (free fluid/hemoperitoneum) Methotrexate is only appropriate for hemodynamically stable, unruptured ectopic pregnancies <4 cm without cardiac activity.
Q5. If this were a stable patient with a 3 cm unruptured ectopic and β-hCG of 1,200 mIU/mL, how would you manage medically?
Methotrexate 50 mg/m² IM (single dose protocol):
- Pre-treatment: CBC, LFTs, creatinine, blood group, RhoGAM if Rh-negative
- β-hCG on day 1, 4, 7
- If <15% decline between day 4 and 7 → second dose
- Avoid: alcohol, NSAIDs, folic acid supplements, sexual intercourse until β-hCG negative
- Weekly β-hCG until undetectable
- Return if severe pain, heavy bleeding, or dizziness (signs of rupture)
- ~95–96% success rate in eligible patients
Q6. What are the risk factors for ectopic pregnancy in this patient?
- Prior PID / salpingitis — tubal scarring and impaired ciliary motility
- IUD use — does not cause ectopic but if pregnancy occurs with IUD in situ, higher proportion are ectopic
- Previous ectopic pregnancy, tubal surgery, smoking, assisted reproduction
Q7. Why is bradycardia clinically significant in ectopic pregnancy?
Paradoxical bradycardia (vasovagal response) can occur with significant intraperitoneal bleeding — it should NOT falsely reassure the clinician. Blood in the peritoneal cavity does NOT consistently correlate with peritoneal signs, blood pressure, or pulse rate.
Marking Scheme
| Domain | Marks |
|---|---|
| Correct diagnosis with justification | /4 |
| Immediate emergency management (ABCDE) | /4 |
| Anti-D administration | /1 |
| Surgical options with comparison | /3 |
| Why methotrexate is contraindicated here | /2 |
| Methotrexate protocol for stable patient | /3 |
| Risk factors identified | /2 |
| Communication / safety netting | /1 |
| Total | /20 |
CASE 4 — Abortion
Station Information
Time allowed: 10 minutes
Setting: Family planning / sexual health clinic
Candidate instructions: Counsel this patient about her options. Demonstrate knowledge of abortion methods and legal/ethical considerations.
Patient Vignette
Miss Priya, 23 years old, presents requesting termination of pregnancy. She is 8 weeks pregnant (confirmed by TVUS). She is otherwise healthy, not on regular medications, has no allergies, and has a copper IUD in situ (placed 2 years ago — IUD failure). She is not in a stable relationship and feels unable to continue the pregnancy. She is anxious and wants to know her options.
Counselling Framework (to be demonstrated)
- Non-judgmental, empathetic approach
- Confirm gestational age and exclude ectopic
- Explain all options: continue pregnancy, adoption, termination
- If termination chosen: explain methods available at 8 weeks
- Discuss risks, aftercare, contraception
- Document informed consent
OSCE Questions & Model Answers
Q1. What are the options for termination of pregnancy at 8 weeks?
At 8 weeks (within first trimester), two main options:A. Medical Abortion (Medication Abortion):
- Mifepristone 200 mg orally + Misoprostol 800 μg buccally 24–48 hours later
- FDA-approved up to 70 days (10 weeks) from LMP
- Efficacy: ~97% in early first trimester
- Completely outpatient; can self-administer misoprostol at home
- Side effects: cramping, bleeding (intended effects), nausea, vomiting, diarrhea
- Serious adverse events: ~0.3%
B. Surgical Abortion — Vacuum Curettage (Aspiration):
- Manual vacuum aspiration (MVA) or electric vacuum aspiration (EVA)
- Single outpatient visit; local anaesthesia ± sedation
- Cervical preparation: misoprostol 400 μg vaginally/buccally 3–4 hours before
- Major complications: 0.16%; overall complication rate 1.26%
- Prophylactic doxycycline given preoperatively
Q2. What are the contraindications to medical abortion with mifepristone/misoprostol?
- Ectopic pregnancy (must be excluded first — critical)
- IUD in place — must be removed before starting treatment
- Chronic adrenal failure
- Long-term corticosteroid therapy
- Allergy to mifepristone, misoprostol, or other prostaglandins
- Hemorrhagic disorder / anticoagulation
- Inherited porphyria
Q3. This patient has an IUD in place. What should be done?
The IUD must be removed before initiating medical abortion. Proceeding with mifepristone/misoprostol while an IUD is in situ is a contraindication. The IUD should be removed at the clinic visit before medication is dispensed.
Q4. How does mifepristone work?
Mifepristone is a selective progesterone receptor modulator / competitive progesterone antagonist — it binds to progesterone receptors with high affinity but acts as an antagonist. This blocks the effect of natural progesterone on the endometrium, causing:
- Decidual breakdown and endometrial shedding
- Cervical softening
- Sensitization of the uterus to prostaglandins Misoprostol (PGE1 analogue) then induces uterine contractions and expulsion of the pregnancy.
Q5. What complications should you warn her about?
| Complication | Details |
|---|---|
| Bleeding | Heavy/prolonged in up to 8%; may last 30 days; surgical curettage needed in 2–5% |
| Incomplete abortion | Requires surgical completion |
| Infection | Rare; prophylactic doxycycline used |
| Failed abortion | <3%; confirmed at follow-up ultrasound |
| Ectopic | Must be excluded before treatment — risk of treatment failure if ectopic undetected |
Q6. What second-trimester methods are used (13–24 weeks)?
Dilation and Evacuation (D&E) — most common midtrimester method:
- Requires cervical preparation first:
- Laminaria japonica or Dilapan-S osmotic dilators (inserted 12–24 hours before; absorb water → swell → dilate cervix mechanically + prostaglandin release)
- Misoprostol alone (multidose, e.g., 1,200 μg)
- Mifepristone as adjunct
- Combination of: specialized forceps + large-bore vacuum cannula
- Ultrasound guidance recommended
- Feticidal agents (for late second trimester): intra-amniotic digoxin (1.5 mg) or intracardiac KCl — both effective with minimal maternal adverse effects
Intact D&E: Wide dilation → membrane rupture → breech delivery → decompression of after-coming fetal head
Q7. How does abortion mortality compare to continuing pregnancy?
- Legal abortion mortality: 0.7 per 100,000 procedures
- Maternal mortality (live birth): 8.8 per 100,000 live births
- Medical abortion (mifepristone/misoprostol) mortality: 0.00063% — 14× lower than risk of live birth
- D&E beyond 18 weeks is still safer than continuing pregnancy to term
- Risk increases with gestational age: 0.3/100,000 at ≤8 weeks → 6.7/100,000 at ≥18 weeks
Q8. What contraception would you recommend after abortion?
- Contraception should be started immediately after abortion (same day)
- Options: combined oral contraceptive pill, progestogen-only pill, injectable, implant, IUD (can be inserted immediately after surgical abortion), condoms
- Counsel that fertility returns almost immediately after abortion
Marking Scheme
| Domain | Marks |
|---|---|
| Non-judgmental counselling approach | /2 |
| Explains both medical and surgical options at 8 weeks | /4 |
| IUD must be removed before medical abortion | /2 |
| Mechanism of mifepristone + misoprostol | /3 |
| Contraindications to medical abortion | /2 |
| Complications counselling | /3 |
| Second trimester D&E with cervical preparation | /2 |
| Contraception post-abortion | /2 |
| Total | /20 |
Quick-Reference Summary Card
| Station | Diagnosis | Key Investigation | Key Treatment |
|---|---|---|---|
| 1 — Hyperplasia | EIN / Atypical Hyperplasia | Endometrial biopsy → atypia | Hysterectomy (or LNG-IUD if fertility desired) |
| 2 — Polyp | Endometrial Polyp (AUB-P) | Hysteroscopy (gold standard) | Hysteroscopic polypectomy |
| 3 — Ectopic | Ruptured Right Tubal Ectopic | TVUS + β-hCG (>discriminatory zone, no IUP) | Emergency laparoscopic salpingectomy + Anti-D |
| 4 — Abortion | First-trimester termination | TVUS (confirm IUP, gestational age) | Mifepristone 200 mg + Misoprostol 800 μg buccal |
Sources: Berek & Novak's Gynecology; Robbins & Cotran Pathologic Basis of Disease; Roberts and Hedges' Clinical Procedures in Emergency Medicine; Goldman-Cecil Medicine
This is a shared conversation. Sign in to Orris to start your own chat.