Protein calorie malnutrition in critically ill patient from harrison
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
I now have excellent content from Harrison's (Chap. 346 — Enteral and Parenteral Nutrition, and Chap. 345 — Malnutrition and Nutritional Assessment). Let me compile the comprehensive answer.
Protein-Calorie Malnutrition in the Critically Ill Patient
Harrison's Principles of Internal Medicine, 22nd Edition (2025)
1. Overview and Prevalence
Malnutrition occurs in 30–50% of hospitalized patients depending on the clinical setting, diagnosis, and diagnostic criteria used. In the ICU specifically, the combination of reduced intake, systemic inflammation, and heightened catabolism makes critically ill patients uniquely vulnerable to protein-calorie malnutrition (PCM).
Adverse outcomes of malnutrition include:
- Poor wound healing
- Compromised immune status
- Impaired organ function
- Increased length of hospital stay and readmissions
- Higher mortality and increased healthcare costs
2. Two Distinct Diseases (Pathophysiological Classification)
Harrison's describes two fundamentally different starvation diseases relevant to the critically ill:
a) Simple Starvation-Related Malnutrition (SRM)
- Occurs with low or absent food intake without significant inflammation
- Body adapts by progressively reducing metabolic rate
- Protein catabolism is relatively modest (~3–5 g N/day)
- Energy comes primarily from fat oxidation
- Body protein is preserved as long as energy needs are met by fat
b) Cytokine/Inflammation-Driven Malnutrition (CDM)
- Driven by systemic inflammation (e.g., sepsis, trauma, burns, major surgery)
- Characterized by obligatory, uncontrollable muscle protein catabolism
- A highly protein-catabolic patient may excrete ≥15 g N/day in urine even without dietary protein — more than three times faster than simple fasting
- Since 1 g N lost reflects 6.25 g protein: 15 g N/day = 94 g protein/day lost
- This is the form that dominates in the critically ill
3. Protein Requirements in the Critically Ill
- Healthy adult minimum: 0.65 g/kg/day; recommended: 0.80 g/kg/day
- Critically ill/highly catabolic patient: requirements are markedly increased, driven by:
- Increased amino acid losses (wound exudates, fistulas, inflammatory diarrhea, renal replacement therapy)
- Marked muscle protein catabolism from the metabolic response to systemic inflammation accompanying major injury, serious infections, and intense immune activation
Factors that further increase protein requirement:
- High-dose glucocorticoid therapy
- Sepsis, trauma, burns
- Renal replacement therapy (removes amino acids from circulation)
Factors affecting protein retention:
- Positive energy balance (extra carbohydrate) improves body protein retention
- Negative energy balance reduces efficiency of protein turnover and increases dietary protein requirements
4. Metabolic Response to Critical Illness
The critically ill patient undergoes a profound metabolic shift:
| Feature | Simple Starvation | Critical Illness (CDM) |
|---|---|---|
| Metabolic rate | Reduced (adaptive) | Increased or normal |
| Protein catabolism | Modest (3–5 g N/day) | Severe (≥15 g N/day) |
| Fat utilization | Predominant | Present but protein loss uncontrolled |
| Response to feeding | Anabolism achievable | Anabolism largely blocked during acute phase |
| Albumin/prealbumin | Relatively preserved | Unreliable (lowered by inflammation) |
Key point: In patients with high-level inflammation (CRP >50 mg/L), nutrition care is primarily supportive, not therapeutic for correcting protein deficits. Anabolic nutrition goals become achievable only as inflammation subsides.
5. Diagnosis (GLIM Criteria, 2019)
The Global Leadership Initiative on Malnutrition (GLIM) criteria require at least one phenotypic + one etiologic criterion:
Phenotypic criteria:
- Non-volitional weight loss
- Low BMI
- Reduced muscle mass
Etiologic criteria:
- Reduced food intake or assimilation (≤50% of energy requirement for >1 week, OR any reduction >2 weeks)
- Disease burden/inflammatory condition — acute illness (major infection, burns, trauma, closed head injury) or chronic disease
CRP as supportive lab:
- Mild inflammation: 3.0–9.9 mg/L
- Moderate: 10–50 mg/L
- Severe: >50 mg/L
Note: Albumin and prealbumin are unreliable markers of nutritional status in the presence of inflammation. They reflect acute-phase response, not protein stores.
6. Specialized Nutritional Support (SNS) in the Critically Ill
Indications for SNS
Instrumental SNS (EN or PN) is indicated when all four conditions apply:
- Nutrient ingestion will likely remain inadequate for many days
- There is important muscle loss (of any cause)
- Patient's nutrient requirements are increased (inflammatory/protein-catabolic state)
- SNS has a reasonable prospect of improving clinical outcome or quality of life
Enteral Nutrition (EN) — Preferred Route
Indications: Unable to eat enough, functional GI tract is accessible, and optimized voluntary nutrition is impossible.
- Common in: impaired consciousness, severe dysphagia, mechanical ventilation
Contraindications (absolute): Intestinal ischemia, mechanical obstruction, peritonitis, GI hemorrhage
Relative contraindications: High-dose pressor therapy (risk of bowel ischemia), severe coagulopathy, paralytic ileus, hypotension, diarrhea, nausea/vomiting
Intensive EN is best avoided during very grave and unstable illness.
Initiation and monitoring:
- Nasogastric tube; head of bed raised to ≥30° to prevent aspiration
- Standard formula: start at 50 mL/h, advance by 25 mL/h every 4–8 h to goal rate
- Intragastric bolus option: 200–400 mL over 15–60 min with 4-hourly residual checks
Complications of EN:
- Aspiration pneumonia (most dangerous — especially in ventilated patients)
- Diarrhea, hyperglycemia, fluid/electrolyte derangements
- Failure to achieve nutritional goal
Parenteral Nutrition (PN) — When EN Not Feasible
- Contains: crystalline amino acids, glucose, lipid emulsions, minerals, electrolytes, micronutrients — infused directly into bloodstream
- Indicated when EN is contraindicated, impractical, or failing to meet nutritional goals
7. Refeeding Syndrome
A specific complication when refeeding severely malnourished/adapted patients:
Mechanism:
- Carbohydrate refeeding → insulin secretion → cellular uptake of phosphate, potassium, magnesium
- Results in dangerous hypophosphatemia, hypokalemia, hypomagnesemia
Cardiovascular consequences:
Three causes of left heart failure during refeeding:
- Abrupt increase of intravascular volume (fluids + glucose → insulin-mediated renal Na retention)
- Increased cardiac demand on an atrophic left ventricle (insulin-mediated ↑ resting energy expenditure)
- Myocardial deficiencies of K, P, Mg
Prevention:
- Severely limit sodium provision
- Introduce carbohydrate slowly
- Monitor serum phosphate, potassium, magnesium frequently
- Provide appropriate electrolyte supplements
8. Special Clinical Situations in the ICU
| Condition | Key Consideration |
|---|---|
| Renal failure | Minimize non-essential amino acids; standard protein targets in patients receiving renal replacement therapy |
| Liver failure | Generous energy and protein despite encephalopathy risk; spread protein across day; meticulous attention to fluid, electrolyte, and acid-base balance |
| Major surgery | 7–10 days preoperative SNS for malnourished patients; early postoperative PN if unable to eat |
| Cancer | SNS improves quality of life and therapy tolerance; not prescribed in advanced disease with no effective anticancer therapy |
Summary
In critically ill patients, cytokine-driven malnutrition (CDM) dominates — characterized by uncontrolled muscle protein catabolism driven by systemic inflammation, not merely by inadequate intake. Protein requirements are markedly elevated (far above the standard 0.8 g/kg/day). Albumin and prealbumin are unreliable guides in inflammation. The GLIM criteria provide a practical diagnostic framework. During the acute inflammatory phase, nutritional support is primarily supportive; anabolic nutrition becomes therapeutic as inflammation resolves. EN is preferred over PN when the gut is accessible. Refeeding syndrome is a critical hazard during nutritional repletion.
Source: Harrison's Principles of Internal Medicine, 22nd Edition (2025) — Chaps. 345 (Malnutrition and Nutritional Assessment) and 346 (Enteral and Parenteral Nutrition), pp. 2659–2680
Definition,causes, type, clinical features, management
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Loading Image
Reading File
Now I have comprehensive material. Let me compile the full answer.
Protein-Calorie Malnutrition (PCM) — Definition, Causes, Types, Clinical Features, Management
Sources: Harrison's Principles of Internal Medicine 22E (2025); Robbins & Cotran Pathologic Basis of Disease; Sleisenger & Fordtran's GI and Liver Disease
Definition
Protein-Calorie Malnutrition (PCM) — also called Protein-Energy Malnutrition (PEM) or Severe Acute Malnutrition (SAM) — is a state resulting from an inadequate intake of dietary protein and/or calories to meet the body's metabolic needs, leading to depletion of body protein and fat stores with impairment of physiological functions.
The WHO defines SAM as:
- Weight-to-height ratio ≥3 standard deviations below the median growth standard, OR
- Visible wasting, OR
- Presence of nutritional edema
There are two protein compartments at stake:
- Somatic compartment — skeletal muscle proteins (depleted more in marasmus)
- Visceral compartment — liver and organ proteins (depleted more in kwashiorkor)
Causes
Primary Causes (Inadequate Intake)
| Category | Examples |
|---|---|
| Poverty / food insecurity | Most common cause globally; war, famine, refugee settings |
| Early weaning | Infant fed carbohydrate-only diet after displacement by new sibling |
| Fad diets | Replacement of milk by rice-based beverages |
| Eating disorders | Anorexia nervosa, bulimia nervosa |
Secondary Causes (Increased Loss / Demand)
| Category | Examples |
|---|---|
| GI malabsorption | Chronic diarrhea, short bowel syndrome, protein-losing enteropathy |
| Protein loss | Nephrotic syndrome, extensive burns, large wounds/fistulas |
| Chronic disease | Cancer, COPD, congestive heart failure, CKD, IBD |
| Systemic inflammation / critical illness | Sepsis, trauma, major surgery, burns — drives cytokine-mediated catabolism |
| Drug therapy | Drugs interfering with nutrient absorption or function |
| Total parenteral nutrition | Iatrogenic if improperly formulated |
| Neurological | Dysphagia post-stroke, advanced dementia |
| Psychiatric | Depression, psychotic disorders |
| Elderly/bedridden | >50% of nursing home residents in the US are malnourished |
Types
PCM exists on a spectrum between two classical poles:
Type 1: Marasmus ("Dry" PCM)
- Caused by severe global caloric deficiency (both protein and energy)
- Somatic compartment predominantly affected
- Adaptive catabolism provides amino acids as energy
- Visceral proteins relatively spared → albumin normal or only slightly reduced
- Subcutaneous fat mobilized as fuel
Type 2: Kwashiorkor ("Wet" PCM)
- Caused by protein deprivation relatively greater than caloric reduction
- Carbohydrate-dominated diet (e.g., post-weaning)
- Visceral compartment severely depleted → marked hypoalbuminemia → edema
- Often precipitated by superimposed infection/acute stress
Type 3: Marasmic-Kwashiorkor (Mixed)
- Features of both types with wasting and edema
- Occurs when a severely wasted child develops an acute infectious illness
Type 4: Nutritional Dwarfism (Stunting)
- Chronic mild-to-moderate PCM
- Weight < 60% for age; normal weight-for-height
- Growth retardation without acute wasting
In Adults / Critically Ill (Harrison's Classification):
| Type | Mechanism |
|---|---|
| SRM (Starvation-Related Malnutrition) | Low intake, no significant inflammation; adaptive metabolic reduction |
| CDM (Cytokine/Inflammation-Driven Malnutrition) | Obligatory muscle catabolism from systemic inflammation; dominant in ICU |
Waterlow Classification of Severity
| Parameter | Normal | Mild | Moderate | Severe |
|---|---|---|---|---|
| Weight-for-height (Wasting) | 90–100% | 80–89% | 70–79% | <70% |
| Z-score | ±Z | −1.1 to −2 Z | −2.1 to −3 Z | <−3 Z |
| Height-for-age (Stunting) | 95–105% | 90–94% | 85–89% | <85% |
Clinical Features
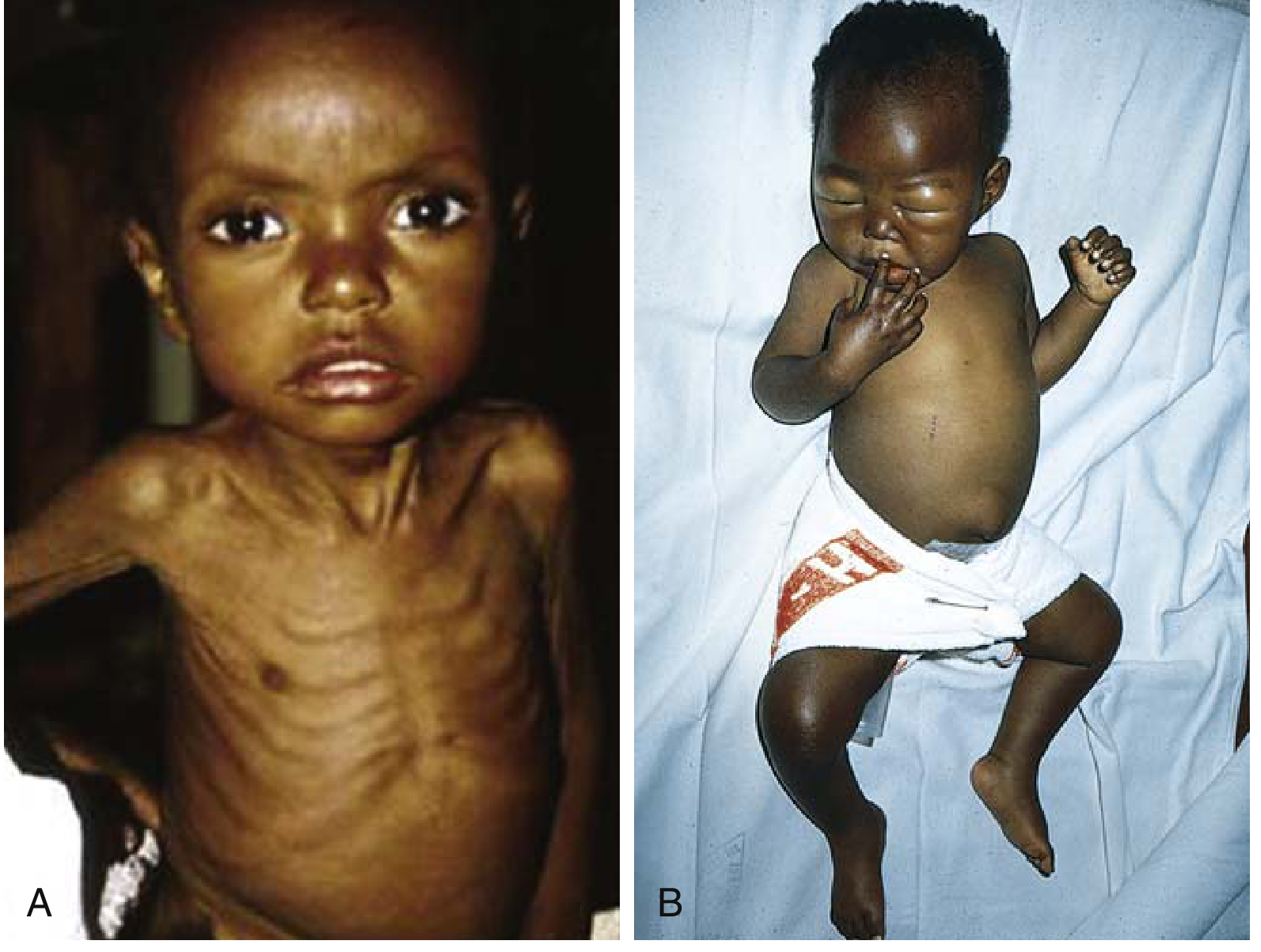
Comparative Clinical Features
| Feature | Kwashiorkor | Marasmus |
|---|---|---|
| Edema | Present (hallmark) | Absent |
| Weight for age | 60–80% of normal | <60% of normal |
| Muscle wasting | Mild (masked by edema) | Severe, obvious |
| Subcutaneous fat | Relatively spared | Markedly depleted |
| Appetite | Poor | Good |
| Mood/behavior | Apathetic alone; irritable when held | Alert |
| Serum albumin | Markedly low | Normal or slightly low |
| Liver | Enlarged, fatty (impaired lipoprotein synthesis) | Normal |
| Skin | "Flaky paint" — alternating hyperpigmentation, desquamation, hypopigmentation | Loose, wrinkled |
| Hair | Depigmented bands ("flag sign"), easily pluckable, straight | May be sparse |
| Abdomen | Protuberant (muscle weakness + hepatomegaly + intestinal distension) | Scaphoid (sunken) |
| Face | "Moon face" (edematous) | "Old man face" (wasted) |
Features Common to Both Types
- Growth failure / weight loss
- Anemia — multifactorial (iron, folate, protein deficiency; anemia of chronic inflammation)
- Immune deficiency — particularly T-cell-mediated immunity → recurrent infections
- Vitamin deficiencies — vitamins A, C, folate, B12, zinc
- Hypothermia — due to loss of insulating subcutaneous fat and reduced metabolic rate
- Poor wound healing
- Bone marrow hypoplasia — reduced red cell precursors
Morphological Changes (Pathology)
- Liver: Fatty change in kwashiorkor (reduced apolipoprotein synthesis → VLDL impairment); not in marasmus
- Small intestine (kwashiorkor): Decreased crypt mitoses → mucosal atrophy, villous loss, disaccharidase deficiency
- Bone marrow: Hypoplastic; mixed anemia (microcytic, normocytic, or macrocytic)
- Brain (severe infantile PCM): Cerebral atrophy, reduced neurons, impaired myelination
- Thymus and lymphoid tissue: Atrophic (more in kwashiorkor)
In Critically Ill Adults (Harrison's)
- Hypoalbuminemia — marker of inflammation, not nutrition per se
- ECF expansion — edema may conceal the true degree of muscle wasting
- BMI unreliable in presence of edema/obesity
- Urinary nitrogen loss ≥15 g N/day in severe catabolic states → loss of ~94 g protein/day
- Low serum prealbumin — reflects systemic inflammation (not nutritional status)
Management
Step 1: Screening and Diagnosis
- Use validated screening tools (NRS-2002, MUST, MNA)
- Diagnose using GLIM criteria: ≥1 phenotypic + ≥1 etiologic criterion
- Assess severity: weight loss, BMI, muscle mass
- Identify underlying cause (inflammation vs. inadequate intake)
Step 2: Treat the Underlying Cause
- Manage infection, sepsis, trauma, malabsorption
- Modify drugs that impair nutrient absorption
- Address barriers to voluntary food intake (dysphagia, pain, nausea, depression)
Step 3: Nutritional Repletion
A. Optimized Voluntary Nutrition (First-line when feasible)
- Preferred — empowers patient, supports mobilization
- Address specific barriers: food texture, palatability, timing
- Oral nutritional supplements
B. Enteral Nutrition (EN) — preferred when gut is accessible
Standard initiation:
- Nasogastric tube; head of bed at ≥30°
- Polymeric formula: start at 50 mL/h, advance by 25 mL/h every 4–8 h to goal rate
- Intragastric bolus option: 200–400 mL every 4 h
Formula types:
| Formula | Use |
|---|---|
| Standard polymeric (1–2 kcal/mL, 50–70 g protein/L) | Most patients |
| Fiber-enriched | Bowel dysfunction |
| Elemental/semi-elemental | Maldigestion, malabsorption |
| Immune-enhancing (arginine, omega-3, glutamine) | Major elective GI surgery |
| Disease-specific (renal, hepatic, diabetic) | Organ-specific adaptation |
Contraindications to EN: Intestinal ischemia, obstruction, peritonitis, GI hemorrhage; high-dose pressors (relative)
C. Parenteral Nutrition (PN) — when EN not feasible
- Contains: crystalline amino acids + glucose + lipid emulsions + electrolytes + micronutrients
- Central PN for full caloric needs; peripheral PN for supplemental needs
- Indicated when EN is contraindicated, impractical, or failing to meet nutritional goals
Protein and Energy Targets
| Patient Type | Protein Requirement | Energy |
|---|---|---|
| Healthy adult | 0.65–0.80 g/kg/day | ~36 kcal/kg/day |
| Critically ill / catabolic | Markedly increased (1.2–2.0+ g/kg/day) | Measured by indirect calorimetry or predictive equation |
| Renal failure (no RRT) | Protein-restricted (individualized) | Adequate energy |
| Liver failure | Generous protein + energy; spread over day | — |
Step 4: Prevent and Manage Refeeding Syndrome
Risk: Patients with severe adapted starvation (SRM) started on nutrition
| Electrolyte | Danger | Prevention |
|---|---|---|
| Phosphate | Hypophosphatemia → cardiac/skeletal muscle dysfunction, hemolysis | Monitor frequently; supplement |
| Potassium | Hypokalemia → arrhythmias | Monitor; supplement |
| Magnesium | Hypomagnesemia | Monitor; supplement |
| Sodium | Volume overload | Severely limit sodium provision |
Cardiac complications of refeeding:
- Abrupt expansion of intravascular volume (glucose → insulin → renal Na retention)
- Increased cardiac demand on atrophic left ventricle
- Myocardial K/P/Mg deficiency
Prevention: Start carbohydrate slowly; limit sodium; monitor and supplement electrolytes from day 1
Monitoring
- Serum electrolytes (especially phosphate, K, Mg) — daily during refeeding
- Nitrogen balance — if available
- BMI, muscle circumference
- CRP (tracks inflammation trajectory)
- Serum albumin — useful over weeks to months after inflammation resolves (t½ ~2 weeks)
Sources: Harrison's Principles of Internal Medicine 22E (2025), Chaps. 345–346, pp. 2659–2680; Robbins & Cotran Pathologic Basis of Disease, Chap. 9, pp. 411–413; Sleisenger & Fordtran's GI and Liver Disease, Chap. 5
This is a shared conversation. Sign in to Orris to start your own chat.