Prevention and management of acute mountain sickness and high altitude pulmonary edema and cerebral edema.
Reading File
Finding Sources
Finding Sources
Searching PubMed
"acute mountain sickness" AND "high altitude" AND prevention management
Reading File
Reading File
Finding Sources
Searching PubMed
"high altitude" AND ("pulmonary edema" OR "cerebral edema")
Reading File
Reading File
Finding Sources
Reading File
Loading Image
Loading Image
Reading File
Searching the Web
Wilderness Medical Society 2024 altitude illness guidelines acetazolamide dexamethasone dosing
Loading Image
I now have all the information needed for a comprehensive answer. Let me compile it.
High-Altitude Illness: Prevention and Management
High-altitude illness (HAI) encompasses three related but distinct syndromes — acute mountain sickness (AMS), high-altitude cerebral edema (HACE), and high-altitude pulmonary edema (HAPE) — all triggered by hypobaric hypoxia on ascent. AMS and HACE represent a continuum of neurological illness; HAPE is primarily a pulmonary disorder that can occur independently.
1. Definitions and the Altitude Threshold
| Altitude | Clinical Relevance |
|---|---|
| >2500 m (~8200 ft) | HAPE may develop in susceptible individuals |
| >3000 m (~9800 ft) | AMS becomes common; standard ascent rules apply |
| >5000 m | Retinal hemorrhages occur frequently even without symptoms |
2. Acute Mountain Sickness (AMS)
Clinical Features
AMS is a neurologic syndrome developing 6–12 hours after ascent. Diagnosis is clinical, using the Lake Louise Scoring System (headache plus at least one of nausea/vomiting, fatigue, dizziness, difficulty sleeping). It must be distinguished from exhaustion, dehydration, hypothermia, alcoholic hangover, and hyponatremia.
- Mild AMS: Headache + mild symptoms, no functional impairment
- Moderate AMS: Moderate headache, significant fatigue, nausea, reduced activity
- Severe AMS / HACE: Ataxia, altered consciousness — encephalopathy
Risk Factors
- Rate of ascent — the single most important risk factor
- Prior history of altitude illness
- Physical exertion (but not lack of fitness)
- Sleep hypoxia/desaturation
- Young age (>50 years old may be relatively protected)
- Respiratory tract infections; dehydration; neck irradiation damaging carotid bodies
3. High-Altitude Cerebral Edema (HACE)
Clinical Features
HACE is the severe end of the AMS spectrum — an encephalopathy characterized by:
- Ataxia (truncal)
- Altered consciousness (confusion, lethargy, coma)
- Diffuse, not focal, neurological deficits
- Papilledema, retinal hemorrhages
- Untreated: fatal within 24 hours
Pathophysiology
Hypobaric hypoxia → hypoxic cerebral vasodilation → ↑ cerebral blood flow → ↑ capillary pressure → vasogenic edema. VEGF, histamine, arachidonic acid, nitric oxide, and adenosine all increase blood-brain barrier permeability. Venous outflow obstruction also contributes. Vasogenic edema may progress to cytotoxic (intracellular) edema in severe HACE.
MRI finding: T2 hyperintensity in the splenium of the corpus callosum and posterior white matter is the hallmark (Fig. 475-1). Hemosiderin deposits here represent long-lasting "footprints" of HACE.
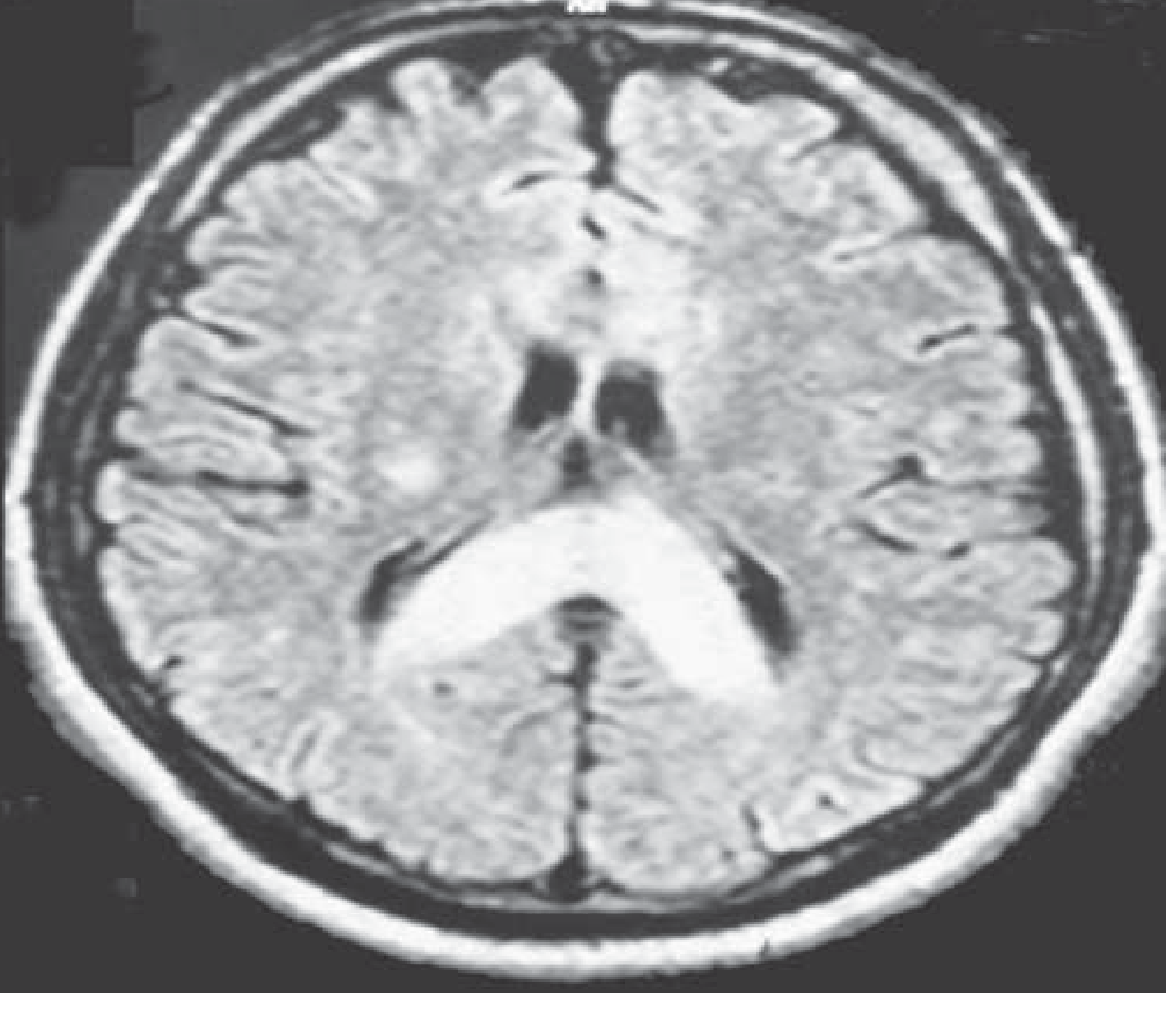
T2 MRI of HACE: marked swelling with hyperintense signal in the posterior body and splenium of the corpus callosum. — Harrison's Principles of Internal Medicine 22E, Fig. 475-1
4. High-Altitude Pulmonary Edema (HAPE)
Clinical Features
HAPE is a non-cardiogenic pulmonary edema developing within 2–4 days after arrival at altitude. It rarely occurs after >4–5 days at the same altitude (adaptation renders vasculature less susceptible). HAPE is the most common fatal altitude illness (0.5–2% of those rapidly ascending).
- Initial: reduced exercise tolerance, dry cough
- Progression: tachypnea, tachycardia at rest, blood-tinged frothy sputum, cyanosis
- Crackles at bases; right ventricular strain on ECG
- Hypoxemia + respiratory alkalosis
- CXR: patchy/localized opacities, NOT Kerley B lines or bat-wing pattern
Risk factors specific to HAPE: Male sex, rapid ascent, prior HAPE history, cold temperatures, respiratory infections, mitral stenosis, primary pulmonary hypertension, unilateral absence of pulmonary artery. Patent foramen ovale is 4× more common in HAPE-susceptible individuals (likely a marker, not cause).
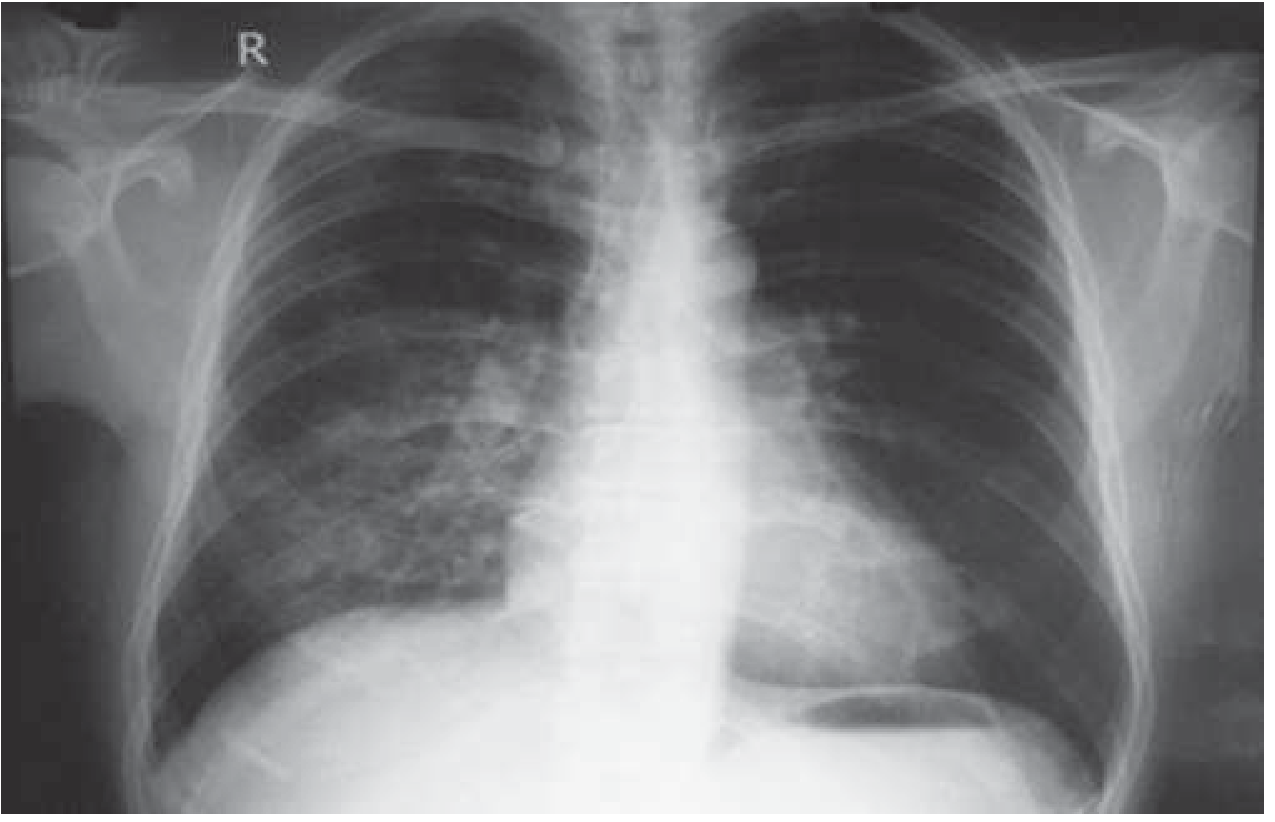
CXR of HAPE: opacity in the right middle and lower zones simulating pneumonic consolidation. Clears with descent and supplemental oxygen. — Harrison's 22E, Fig. 475-3
Pathophysiology
Hypoxia → uneven hypoxic pulmonary vasoconstriction → increased blood flow through unprotected vessels → elevated capillary pressure → fluid leak (non-cardiogenic). Inflammatory kinins increase capillary permeability. HAPE is a protein-rich edema with a normal pulmonary capillary wedge pressure (confirming non-cardiogenic origin). Impaired alveolar fluid clearance (reduced Na⁺/K⁺-ATPase activity in epithelial cells) further contributes. — Fishman's Pulmonary Diseases, Guyton & Hall
5. Prevention
Non-Pharmacological (All Altitude Illness)
- Gradual ascent is the cornerstone of prevention
- Above 3000 m: increase sleeping altitude by ≤300–500 m/day
- Rest day (no ascent) every 3–4 days, or every 1000 m gained
- Spend one night at an intermediate altitude before going higher
- Avoid overexertion on arrival
- Prior high-altitude exposure within preceding weeks confers protection
Pharmacological Prevention (2024 WMS Guidelines)
| Drug | Indication | Dose | Notes |
|---|---|---|---|
| Acetazolamide | AMS/HACE prevention (moderate-high risk) | 125 mg PO q12h (adult); children: 1.25 mg/kg q12h | Start day before ascent; continue 2–4 days at peak altitude. Facilitates acclimatization by inducing metabolic acidosis → stimulates ventilation. Contraindicated in sulfonamide allergy. |
| Dexamethasone | AMS/HACE prevention (adult only, 2nd line or high-risk) | 2 mg PO q6h or 4 mg q12h | Does NOT facilitate acclimatization — masks symptoms. Taper if used >5–7 days. Not recommended for prophylaxis in children. |
| Nifedipine (ER) | HAPE prevention (prior HAPE history only) | 30 mg ER q12h or 20 mg ER q8h | Start day before ascent; continue 4–7 days or until descent. Lowers pulmonary artery pressure. |
| Tadalafil | HAPE prevention (alternative) | 10 mg q12h | 2nd-line for HAPE prophylaxis in HAPE-susceptible individuals. |
| Sildenafil | HAPE prevention (alternative) | 50 mg q8h | 3rd-line option for HAPE prophylaxis. |
| Ibuprofen | High-altitude headache | 600 mg q8h | Effective for headache treatment; some evidence for AMS prevention. |
Key point: Acetazolamide is ineffective for HAPE prophylaxis. Do not rely on it alone in HAPE-susceptible individuals. Nifedipine (or tadalafil) is the drug of choice for HAPE prophylaxis.
6. Treatment
The management algorithm below illustrates the decision framework:
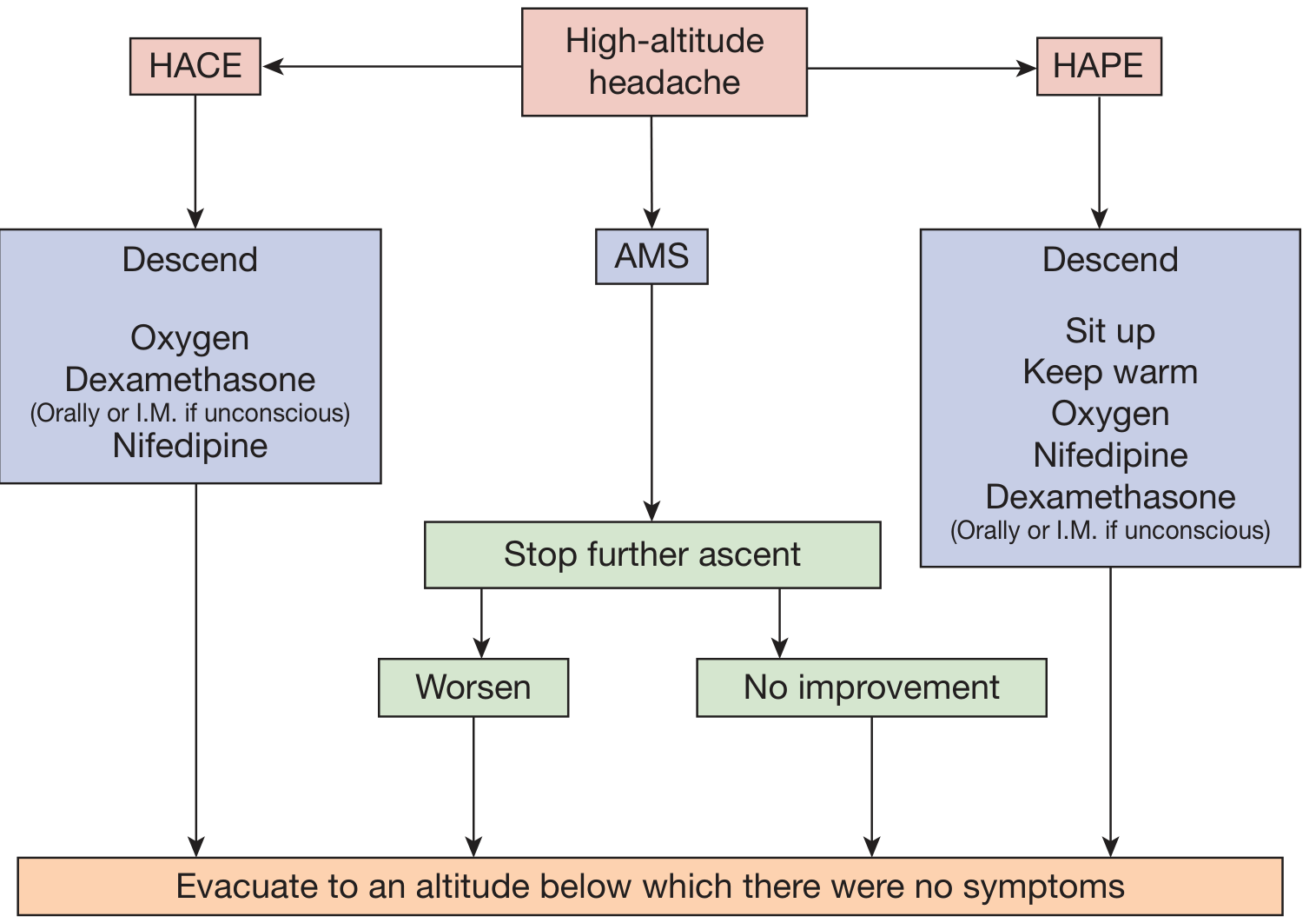
Overview of the management pathway for acute altitude illnesses. — Fishman's Pulmonary Diseases, Fig. 91-6
Treatment Table (from Harrison's 22E, Table 475-1)
| Condition | Management |
|---|---|
| AMS — Mild | Stop further ascent · Acetazolamide 250 mg q12h · Descend if no improvement in 24 h |
| AMS — Moderate | Immediate descent if worsening · Low-flow O₂ if available · Acetazolamide 250 mg q12h and/or dexamethasone 4 mg q6h · Hyperbaric therapy |
| HACE | Immediate descent or evacuation · O₂ 2–4 L/min · Dexamethasone 8 mg PO/IM/IV, then 4 mg q6h · Hyperbaric chamber if descent not possible |
| HAPE | Immediate descent or evacuation · Minimize exertion, keep warm · O₂ 4–6 L/min (target SpO₂ >90%) · Nifedipine 30 mg ER q12h (adjunct) · Hyperbaric chamber if descent not possible |
Condition-Specific Treatment Details
AMS:
- The cornerstone is stopping ascent; this alone allows acclimatization to catch up
- Acetazolamide 250 mg q12h speeds acclimatization
- Analgesics (ibuprofen, acetaminophen) for headache
- Descent for worsening or failure to improve after 24 h
HACE:
- Descent is the definitive treatment — even 300–1000 m produces dramatic improvement
- Dexamethasone 8 mg loading dose then 4 mg q6h (blocks VEGF upregulation, reduces vasogenic edema)
- O₂ 2–4 L/min
- Gamow bag (portable hyperbaric chamber, 2 psi) if descent is impossible — simulates descent of ~1500 m and produces spectacular improvement, "buying time"
- Nifedipine and PDE-5 inhibitors have no role in HACE treatment
- Acetazolamide is an adjunct to dexamethasone in HACE but not primary treatment
HAPE:
- Descent ≥1000 m or until symptoms resolve is definitive
- Supplemental O₂ to SpO₂ >90% is highly effective and may reverse HAPE without descent
- Nifedipine 30 mg ER (or 10 mg sublingual acutely) — reduces pulmonary artery pressure
- Tadalafil, sildenafil — PDE-5 inhibitors reduce pulmonary vascular resistance and have shown benefit in HAPE treatment
- CPAP has been used where available
- Nitric oxide + O₂ has been reported
- Gamow bag if no O₂ or descent available
- Patient should sit upright; keep warm (cold worsens hypoxic vasoconstriction)
- Dexamethasone is used in HAPE but is not primary treatment; often given when HACE coexists
7. Hyperbaric Therapy (Gamow Bag)
A portable fabric hyperbaric chamber inflated to ~2 psi above ambient pressure simulates descent of ~1500 m. It produces rapid symptomatic improvement in both HACE and HAPE and is particularly valuable in remote settings where immediate descent is not possible. It does not replace definitive descent — once removed, symptoms may return unless acclimatization has occurred.
8. Special Populations
| Population | Key Point |
|---|---|
| Coronary artery disease | VO₂ max falls at altitude; exercise treadmill test before high-altitude travel; strongly positive test = contraindication |
| Hypertension | Continue antihypertensives; most well-controlled hypertensives remain stable; prazosin or nifedipine for labile hypertension |
| Asthma | Generally better tolerated at altitude (fewer allergens); carry all medications including oral glucocorticoids |
| Pregnancy | ≤3000 m acceptable for low-risk; avoid higher altitudes where SpO₂ drops steeply |
| Sickle cell disease | Vaso-occlusive crisis risk even at 2500 m; use supplemental O₂ |
| Obesity | Higher risk of AMS (pronounced nocturnal hypoxemia); avoid rapid ascent |
| Migraine | Increased AMS likelihood; high-altitude headache responds to O₂ (migraine does not) |
| Cerebrovascular disease | TIA: avoid high altitude for ≥3 months; known cerebral aneurysm: contraindication |
Recent Evidence (2024 WMS Guidelines — PMID 37833187)
The 2024 Wilderness Medical Society update (Luks et al., Wilderness Environ Med, 2024) confirmed the following key updates vs. prior guidelines:
- Acetazolamide prophylaxis dose lowered to 125 mg q12h (previously 250 mg; equally effective, fewer side effects)
- Salmeterol is no longer recommended for HAPE prophylaxis (weak benefit, moderate-quality evidence against)
- A new risk-stratification model was introduced: moderate-high risk = prior altitude illness, OR sleeping altitude ≥2800 m on Day 1, OR ascent rate ≥500 m/day above 3000 m
- For high-risk ascent profiles (>5000 m), 250 mg q12h acetazolamide may still be appropriate
Sources: — Harrison's Principles of Internal Medicine, 22nd Ed. (2025), Ch. 475 · — Guyton and Hall Textbook of Medical Physiology, Ch. 44 · — Fishman's Pulmonary Diseases and Disorders, Ch. 91 · 2024 WMS Altitude Illness Guidelines (Luks AM et al., Wilderness Environ Med 2024)
Nifedipine vs tadalafil
Finding Sources
Searching PubMed
"nifedipine" AND "tadalafil" AND "high altitude pulmonary edema"
Reading File
Reading File
Reading Article
Searching PubMed
"tadalafil" AND "high altitude pulmonary edema" prevention
Searching PubMed
"tadalafil" AND "nifedipine" AND "altitude" AND randomized
Finding Sources
Reading File
Reading Article
I have comprehensive data for a direct, well-sourced comparison. Here it is:
Nifedipine vs. Tadalafil for HAPE Prevention (and Treatment)
Both drugs target the same pathophysiological bottleneck — excessive hypoxic pulmonary vasoconstriction (HPV) — but through different mechanisms, and their clinical profiles differ in practically relevant ways.
Mechanism of Action
| Nifedipine | Tadalafil | |
|---|---|---|
| Drug class | Dihydropyridine calcium channel blocker | PDE-5 inhibitor |
| Mechanism | Blocks L-type voltage-gated Ca²⁺ channels → relaxes pulmonary arterial smooth muscle → reduces pulmonary artery pressure | Inhibits PDE-5 → ↑ cGMP → prolongs nitric oxide–mediated vasodilation → reduces pulmonary vascular resistance |
| Nitric oxide pathway | Indirect (reduces vasoconstriction independently of NO) | Directly potentiates endogenous NO signalling |
| Why relevant to HAPE | Counters the uneven HPV that causes overperfusion and capillary stress failure | Restores the impaired NO-dependent vasodilation seen in HAPE-susceptible individuals (who have reduced exhaled NO at altitude) |
Crucially, HAPE-susceptible people have lower exhaled nitric oxide and higher endothelin-1 levels — this is why PDE-5 inhibitors like tadalafil have a mechanistic rationale beyond just nonspecific vasodilation. — Harrison's 22E, Ch. 475
Prevention Efficacy
The key landmark RCT (Maggiorini et al., Ann Intern Med, 2006; same ascent profile to ~4500 m in HAPE-susceptible subjects) directly compared tadalafil and dexamethasone against placebo:
- Tadalafil reduced HAPE incidence by ~65%
- Dexamethasone reduced it by ~78% (surprisingly superior — possibly via sympathetic suppression + increased endothelial NO availability)
For nifedipine, earlier RCTs demonstrated ~70–100% reduction in HAPE at the same altitude with the same ascent profile (Berger 2022, PMID 35511718 cites these historical data as the benchmark). The 2025 pilot RCT (George et al., PMID 39872192) in military personnel with prior HAPE showed 0% HAPE recurrence with nifedipine, tadalafil, or acetazolamide vs. ~13% with placebo on reascent — all three were equivalent in that small sample (n=30/group).
Summary: Both drugs are roughly equivalent in efficacy (~65–100% HAPE risk reduction). Nifedipine has the longer evidence base; tadalafil data are robust enough for guideline endorsement. — Harrison's 22E; 2024 WMS Guidelines
Dosing
| Nifedipine | Tadalafil | |
|---|---|---|
| Prevention | 30 mg ER q12h (or 20 mg ER q8h) | 10 mg q12h |
| Treatment (adjunct) | 30 mg ER q12h, or 10 mg SL acutely if needed | 10 mg q12h (or sildenafil 50 mg q8h) |
| Start | Day before ascent | Day before ascent |
| Duration | Continue 4–7 days or until descent | Continue 4–7 days or until descent |
Note: Harrison's 22E footnotes the treatment table that tadalafil 10 mg BID, sildenafil 50 mg TID, and dexamethasone 8 mg BID are all acceptable for HAPE prevention alongside nifedipine 30 mg ER BID.
Practical Clinical Comparison
| Feature | Nifedipine | Tadalafil |
|---|---|---|
| Guideline position (2024 WMS) | 1st-line HAPE prophylaxis | 2nd-line |
| Evidence quality | Strong / moderate quality | Strong / low quality |
| Systemic hypotension | More likely — especially with SL dose; monitor BP | Less systemic vasodilation; better tolerated hemodynamically |
| Reflex tachycardia | Yes (dihydropyridine effect) | Minimal |
| Headache | Less common | More common (PDE-5-mediated vasodilation) — can confound AMS diagnosis |
| Dosing convenience | BID (ER formulation) | BID — similar |
| Role in HACE/AMS | None | None |
| Use in treatment | Yes — sublingual 10 mg acutely, then ER form | Acceptable adjunct; less evidence than nifedipine for acute treatment |
| Drug interactions | CYP3A4 inhibitors increase levels | Nitrates (absolute CI), some antihypertensives |
| Cost/availability | Generally cheaper, widely available | More expensive, less available in remote areas |
Key Takeaway (2024 WMS Guidelines)
For HAPE-susceptible individuals (prior HAPE history) who cannot ascend gradually:
- 1st line: Extended-release nifedipine 30 mg q12h
- 2nd line: Tadalafil 10 mg q12h
- 3rd line: Dexamethasone 8 mg q12h (reserved; adverse effect profile requires monitoring)
- Do not use: Acetazolamide alone (no significant effect on pulmonary artery pressure; HAPE incidence not statistically reduced; see Berger 2022 RCT, PMID 35511718)
The choice between nifedipine and tadalafil in practice often comes down to tolerability: tadalafil's main advantage is less systemic hypotension, making it preferable in patients who are already vasodilated, volume-depleted, or taking other antihypertensives. Its main drawback is the headache it causes, which can obscure early AMS symptoms. Nifedipine is preferred where cost and availability are concerns, and it is the only agent with an acute sublingual rescue option (10 mg SL) when treatment of established HAPE is needed and descent is delayed.
— Harrison's Principles of Internal Medicine, 22nd Ed. (2025) · Goldman-Cecil Medicine · Fishman's Pulmonary Diseases · 2024 WMS Clinical Practice Guidelines
This is a shared conversation. Sign in to Orris to start your own chat.