Acute respiratory distress syndrome management
"acute respiratory distress syndrome"[MeSH Terms] AND management
ARDS management guidelines 2024 2025 lung protective ventilation
"acute respiratory distress syndrome"[MeSH Terms] AND "mechanical ventilation"[MeSH Terms]
ARDS management guidelines ATS ESICM 2024
"ARDS" AND "prone positioning" OR "corticosteroids" OR "ECMO"
Acute Respiratory Distress Syndrome (ARDS): Management
Definition & Severity Classification (Berlin Definition 2012)
- Bilateral radiographic opacities not fully explained by effusions, atelectasis, or nodules
- Respiratory failure not fully explained by cardiac failure or fluid overload
- PaO₂/FiO₂ ≤ 300 with PEEP or CPAP ≥ 5 cm H₂O
- Onset within 1 week of a known clinical insult
| Severity | PaO₂/FiO₂ | PEEP |
|---|---|---|
| Mild | ≤ 300 but > 200 mmHg | ≥ 5 cm H₂O |
| Moderate | ≤ 200 but > 100 mmHg | ≥ 5 cm H₂O |
| Severe | ≤ 100 mmHg | ≥ 5 cm H₂O |
Core Management Principles
- Aggressive treatment of the underlying inciting cause
- Avoiding iatrogenic complications
- Lung-protective mechanical ventilation
1. Lung-Protective Ventilation (LPV) — Strong Recommendation (ATS 2024 & ESICM 2023)
ARDSNet Protocol Parameters
| Parameter | Target |
|---|---|
| Tidal volume (VT) | 4–8 mL/kg predicted body weight (PBW) — start at 6 mL/kg |
| Plateau pressure (Pplat) | ≤ 30 cm H₂O |
| Driving pressure | Minimize (target < 15 cm H₂O) |
| Oxygenation goal | PaO₂ 55–80 mmHg or SpO₂ 88–95% |
| pH goal | 7.30–7.45 |
| Initial RR | Approximate baseline minute ventilation, not > 35 bpm |
- If Pplat > 30 cm H₂O → decrease VT by 1 mL/kg steps (minimum 4 mL/kg PBW)
- If Pplat < 25 cm H₂O and VT < 6 mL/kg → increase VT by 1 mL/kg steps
PEEP/FiO₂ Table (Low PEEP Strategy as Starting Point)
| FiO₂ | 0.3 | 0.4 | 0.5 | 0.6 | 0.7 | 0.8 | 0.9 | 1.0 |
|---|---|---|---|---|---|---|---|---|
| PEEP | 5 | 5–8 | 8–10 | 10 | 10–14 | 14 | 14–18 | 18–24 |
Permissive Hypercapnia
- Raised intracranial pressure (trauma, mass lesion)
- Acute cerebrovascular disease
- Acute myocardial ischemia
- Severe pulmonary hypertension / right ventricular failure
- Uncorrected severe metabolic acidosis
- Pregnancy
2. Prone Positioning — Strong Recommendation for Moderate/Severe ARDS
- ATS 2024: > 12 hours/day; ESICM 2023: > 16 hours/day
- Indicated when PaO₂/FiO₂ ≤ 150
- Redistributes perfusion to better-ventilated lung regions, reduces dorsal atelectasis, and improves V/Q matching
- The PROSEVA trial (2013) showed a 16% absolute mortality reduction in severe ARDS
- PRONECMO RCT (Schmidt et al., JAMA 2023, PMID 38038395) evaluated prone positioning combined with VV-ECMO
3. Neuromuscular Blocking Agents (NMBAs)
| Guideline | Recommendation |
|---|---|
| ATS 2024 | Conditional for use in first 48 hours, severe ARDS (P/F < 100) |
| ESICM 2023 | Strong recommendation against routine continuous infusions |
- Early use of cisatracurium (French ACURASYS trial) showed mortality benefit in severe ARDS
- The subsequent ROSE trial (2019) failed to replicate this benefit
- Individualized use is therefore favored over routine application — consider in severe dyssynchrony
4. Corticosteroids — Conditional Recommendation (ATS 2024)
- ATS 2024 conditionally recommends corticosteroids; ESICM 2023 did not formally address this
- A recent trial suggested decreased mortality, shorter ventilation duration, and shorter hospital stay
- Key uncertainties: Optimal agent, dose, timing, and duration remain undefined
- Practical advantages: Low cost, widely available
5. Lung Recruitment Maneuvers (LRMs)
- Both ATS 2024 and ESICM 2023: Strong recommendation AGAINST prolonged LRMs
- ART trial showed increased mortality with aggressive recruitment
- ESICM: weak recommendation against even brief LRMs
6. High-Frequency Oscillatory Ventilation (HFOV)
- ATS 2024: Strong recommendation AGAINST (OSCILLATE trial showed harm)
- Not recommended as a rescue strategy
7. Venovenous ECMO (VV-ECMO) for Severe ARDS
| Guideline | Recommendation |
|---|---|
| ATS 2024 | Conditional recommendation for use |
| ESICM 2023 | Strong recommendation for use in an ECMO-capable center |
- Indicated when severe, refractory hypoxemia persists despite optimal conventional management
- Best outcomes when initiated early, at high-volume centers (EOLIA trial data)
- Combining prone positioning with ECMO is being studied (PRONECMO trial)
8. Fluid Management
- Conservative fluid strategy preferred once hemodynamic stability is established
- FACTT trial (ARDSNet): Conservative strategy improved oxygenation and reduced ventilator days without increasing non-pulmonary organ failure
- Avoid fluid overload — worsens pulmonary edema
- Diuretics may be used to achieve net-negative fluid balance
9. Pharmacological Adjuncts (Limited Evidence)
| Agent | Status |
|---|---|
| Inhaled nitric oxide (iNO) | Transient improvement in oxygenation; no mortality benefit |
| Inhaled prostacyclin | Similar transient oxygenation benefit; may be used as bridge to ECMO |
| Surfactant | Not recommended in adults |
| β₂ agonists (salbutamol) | Trials showed no benefit (BALTI-2); not recommended |
| Vitamin C / thiamine | Insufficient evidence |
10. Supportive ICU Care
- Sedation: Target lightest effective sedation using validated scales (RASS); minimize benzodiazepines
- Early mobilization: Reduces ICU-acquired weakness; passive and active physiotherapy
- Nutritional support: Enteral nutrition preferred; initiate within 24–48 hours
- DVT prophylaxis: Low-molecular-weight heparin unless contraindicated
- Stress ulcer prophylaxis: PPIs or H₂ blockers
- Infection control: Treat inciting infection aggressively; VAP bundle (HOB 30–45°, oral care, cuff pressure, circuit management)
Summary: ATS 2024 vs. ESICM 2023 Comparison
| Intervention | ATS 2024 | ESICM 2023 |
|---|---|---|
| Protective ventilation (VT 4–8 mL/kg PBW) | ✅ Strong FOR | ✅ Strong FOR |
| Early prone positioning (moderate/severe) | ✅ Strong FOR (>12 h/day) | ✅ Strong FOR (>16 h/day) |
| VV-ECMO (severe) | ✅ Conditional FOR | ✅ Strong FOR (ECMO center) |
| Prolonged LRMs | ❌ Strong AGAINST | ❌ Strong AGAINST |
| NMBA (cisatracurium, severe, <48 h) | ✅ Conditional FOR (P/F <100) | ❌ Strong AGAINST routine use |
| Corticosteroids | ✅ Conditional FOR | Not addressed |
| High PEEP (moderate/severe) | ✅ Conditional FOR | No recommendation |
| HFOV | ❌ Strong AGAINST | Not addressed |
| ECCO₂R | Not addressed | ❌ Strong AGAINST |
- Fishman's Pulmonary Diseases and Disorders (Reilly & Christie — ARDS chapter), block 29
- Barash's Clinical Anesthesia, 9e — ARDS section, block 20
- ATS 2024 Clinical Practice Guideline (Qadir N et al., AJRCCM 2024;209:24–36)
- ESICM 2023 Guidelines (Grasselli G et al., Intensive Care Med 2023, PMID 37326646)
- New Global ARDS Definition 2024 (Matthay MA et al., AJRCCM 2024;209:37–47)
- PRONECMO RCT (Schmidt M et al., JAMA 2023, PMID 38038395)
Acute respiratory distress syndrome management
"acute respiratory distress syndrome"[MeSH Terms] AND treatment
ARDS management ATS 2024 guidelines recommendations summary
"ARDS" AND ("prone positioning" OR "corticosteroids" OR "neuromuscular blockade" OR "ECMO")
ARDS management algorithm lung protective ventilation prone positioning
Acute Respiratory Distress Syndrome (ARDS): Management
Definition & Severity Classification (Berlin Definition, 2012)
- Bilateral opacities on CXR/CT not fully explained by effusions, atelectasis, or nodules
- Respiratory failure not fully explained by cardiac failure or fluid overload
- PaO₂/FiO₂ ≤ 300 mmHg with PEEP or CPAP ≥ 5 cm H₂O
- Acute onset within 1 week
| Severity | PaO₂/FiO₂ | PEEP |
|---|---|---|
| Mild | 200–300 mmHg | ≥ 5 cm H₂O |
| Moderate | 100–200 mmHg | ≥ 5 cm H₂O |
| Severe | < 100 mmHg | ≥ 5 cm H₂O |
A 2024 Global ARDS Definition (Matthay MA et al., AJRCCM 2024;209:37–47) expanded criteria to include patients on high-flow nasal oxygen and non-invasive ventilation, and accommodates resource-limited settings.
ATS 2024 Guideline Framework
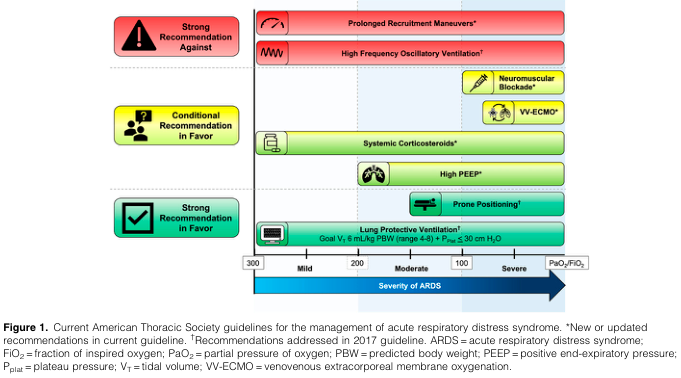
1. Treat the Underlying Cause
- Infectious: sepsis, bacterial/viral/fungal pneumonia (including SARS-CoV-2, Legionella, Pneumocystis), military tuberculosis
- Non-infectious: diffuse alveolar hemorrhage, acute eosinophilic pneumonia, vasculitis (Goodpasture syndrome), drug toxicity, lupus pneumonitis, post-bone marrow transplant
2. Lung-Protective Ventilation (LPV) — Strong Recommendation
NIH ARDSNet Protocol (Table 141-9, Fishman's Pulmonary Diseases)
- Mode: Volume-controlled Assist/Control
- Start VT at 8 mL/kg IBW if baseline > 8 mL/kg, reduce by 1 mL/kg every ≤ 2 hours to target 6 mL/kg IBW
- Initial RR to approximate baseline minute ventilation (max 35 bpm)
- Inspiratory flow > 80 L/min; target I:E ratio of 1:1.0–1.3
| FiO₂ | 0.3 | 0.4 | 0.5 | 0.6 | 0.7 | 0.8 | 0.9 | 1.0 |
|---|---|---|---|---|---|---|---|---|
| PEEP (cm H₂O) | 5 | 5–8 | 8–10 | 10 | 10–14 | 14 | 14–18 | 18–24 |
- Check Pplat at least every 4 h (0.5-s inspiratory pause)
- If Pplat > 30 → decrease VT by 1 mL/kg (minimum 4 mL/kg IBW)
- If Pplat < 25 and VT < 6 mL/kg → increase VT by 1 mL/kg until Pplat > 25 or VT = 6 mL/kg
- pH 7.15–7.30: increase RR (max 35 bpm)
- pH < 7.15: may increase VT in 1 mL/kg steps (Pplat goal may be exceeded as last resort)
- Raised ICP, acute cerebrovascular disease, acute MI, severe pulmonary hypertension/RV failure, uncorrected metabolic acidosis, pregnancy, sickle cell anemia
- Driving pressure (Pplat − PEEP): recent evidence suggests this is the ventilator variable most strongly mediating lung injury — minimize < 15 cm H₂O
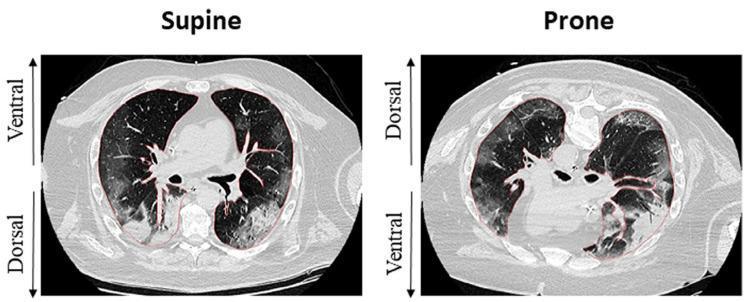
- "Baby lung" concept: CT shows ARDS is heterogeneous — only posterior dependent regions collapse; ventilation preferentially goes to a small "baby lung," making standard tidal volumes effectively over-distending
3. Prone Positioning — Strong Recommendation (Moderate/Severe ARDS)
- Threshold: PaO₂/FiO₂ < 150 mmHg
- Duration: ATS 2024 → > 12 hours/day; ESICM 2023 → > 16 hours/day
- The PROSEVA trial (2013, 466 patients): 28-day mortality 16% prone vs. 32.8% supine — a landmark result
- Mechanism: promotes homogeneous lung inflation from dorsal to ventral, improving V/Q matching and reducing both overdistension and atelectasis, thereby limiting ventilator-induced lung injury (VILI)
4. Neuromuscular Blocking Agents (NMBAs) — Conditional / Selective Use
| Guideline | Recommendation |
|---|---|
| ATS 2024 | Conditional FOR — severe ARDS (P/F < 100), first 48 h |
| ESICM 2023 | Strong AGAINST routine continuous infusion |
- ACURASYS trial (2010, n=340): cisatracurium for 48 h → 90-day mortality 31.6% vs. 40.7% placebo (significant only after adjustment)
- ROSE trial (2019, n=1006): cisatracurium vs. usual care with light sedation → 90-day mortality identical (42.5% vs. 42.8%)
- Key difference: ROSE used lighter sedation in controls; ACURASYS used heavy sedation throughout — suggesting the benefit may have been from avoiding over-sedation, not the paralytic itself
- Severe ventilator dyssynchrony
- Facilitating safe prone positioning
- Refractory severe hypoxemia
5. Corticosteroids — Conditional Recommendation (ATS 2024)
- ESICM 2023 did not formally address; ATS 2024 makes a conditional recommendation for use
- Theoretical benefits: reduce inflammation and fibrosis in acute and proliferative phases
- Risks: secondary infection, hyperglycemia, delirium, neuromuscular weakness
- LaSRS (ARDSNet, 2006): moderate-dose methylprednisolone in persistent ARDS (7–21 days) → no mortality benefit; worse outcomes if started after day 14
- DEXA-ARDS (2020, n=277, Spain): dexamethasone in moderate/severe ARDS (P/F < 200) → reduced ventilator days and improved oxygenation
- Current consensus supports early use (within first 14 days) in moderate/severe ARDS not responding to initial therapies
- Consider in moderate/severe ARDS early in course
- Always use in ARDS with specific non-ARDS indications: PCP pneumonia, acute eosinophilic pneumonia, septic shock
- Avoid if ARDS present > 14 days
- Optimal agent, dose, and duration remain undefined
6. Conservative Fluid Management
- Conservative fluid strategy (target CVP < 4 mmHg or PAOP < 8 mmHg) vs. liberal strategy
- No mortality difference at 60 days, but conservative strategy produced:
- ↓ Duration of assisted ventilation
- ↓ ICU length of stay
- No increase in shock, organ failure, or dialysis use
- Bottom line: Use conservative fluid strategy in ARDS patients not in shock (MAP < 60 or on vasopressors = exception — do not restrict fluids then)
- PAC vs. CVC for hemodynamic monitoring: outcomes identical, but PAC carries higher complication rate — CVC preferred
7. VV-ECMO — Conditional to Strong Recommendation (Severe ARDS)
| Guideline | Recommendation |
|---|---|
| ATS 2024 | Conditional FOR |
| ESICM 2023 | Strong FOR (in ECMO-capable center) |
- For severe, refractory ARDS failing all conventional measures
- Provides gas exchange support while allowing ultra-lung-protective ventilation (VT 1–3 mL/kg, low Pplat)
- Best outcomes at high-volume ECMO centers; early referral recommended
- PRONECMO RCT (JAMA 2023, PMID 38038395): evaluated prone positioning during VV-ECMO in severe ARDS
- Standard ARDS management continues in parallel during ECMO: steroids, prone positioning (coordinated carefully around cannulas), conservative fluids, early tracheostomy
8. Interventions Recommended AGAINST
| Intervention | Guideline | Reason |
|---|---|---|
| Prolonged Lung Recruitment Maneuvers | Both ATS & ESICM: Strong AGAINST | ART trial — increased mortality |
| High-Frequency Oscillatory Ventilation (HFOV) | ATS: Strong AGAINST | OSCILLATE trial — increased mortality |
| ECCO₂R | ESICM: Strong AGAINST | Insufficient benefit, complications |
| Surfactant | Not recommended in adults | Trials failed to show benefit |
| β₂ agonists (salbutamol) | Not recommended | BALTI-2 trial — no benefit |
| Routine iNO | Not recommended for mortality | Transient oxygenation benefit only |
9. Supportive ICU Care
| Domain | Intervention |
|---|---|
| Sedation | Lightest effective sedation (RASS target); avoid deep sedation; minimize benzodiazepines |
| Analgesia-first | Fentanyl/morphine before sedatives (analgosedation protocol) |
| Early mobilization | Passive/active physiotherapy; prevents ICU-acquired weakness |
| Nutrition | Enteral nutrition within 24–48 h; avoid overfeeding |
| DVT prophylaxis | LMWH unless contraindicated |
| Stress ulcer prophylaxis | PPI or H₂ blocker |
| VAP bundle | HOB 30–45°, oral chlorhexidine, cuff pressure checks, circuit management |
| Glycemic control | Target 140–180 mg/dL (7.8–10 mmol/L) |
Summary: ATS 2024 vs. ESICM 2023
| Intervention | ATS 2024 | ESICM 2023 |
|---|---|---|
| Lung-protective ventilation (VT 4–8 mL/kg PBW) | ✅ Strong FOR | ✅ Strong FOR |
| Prone positioning >12–16 h/day (mod/severe) | ✅ Strong FOR (>12 h) | ✅ Strong FOR (>16 h) |
| VV-ECMO (severe refractory) | ⚠️ Conditional FOR | ✅ Strong FOR (ECMO center) |
| Prolonged lung recruitment maneuvers | ❌ Strong AGAINST | ❌ Strong AGAINST |
| HFOV | ❌ Strong AGAINST | Not addressed |
| NMBA (cisatracurium, severe, <48 h) | ⚠️ Conditional FOR (P/F <100) | ❌ Strong AGAINST routine use |
| Systemic corticosteroids | ⚠️ Conditional FOR | Not addressed |
| High PEEP (moderate/severe) | ⚠️ Conditional FOR | No recommendation |
| ECCO₂R | Not addressed | ❌ Strong AGAINST |
- Fishman's Pulmonary Diseases and Disorders (Reilly & Christie) — ARDS chapter
- Sabiston Textbook of Surgery — VV-ECMO in ARDS chapter
- Barash's Clinical Anesthesia, 9e
- ATS 2024 Clinical Practice Guideline — Qadir N et al., AJRCCM 2024;209:24–36 (PMID 38032683)
- ESICM 2023 Guidelines — Grasselli G et al., Intensive Care Med 2023 (PMID 37326646)
- New Global ARDS Definition 2024 — Matthay MA et al., AJRCCM 2024;209:37–47
- PRONECMO RCT — Schmidt M et al., JAMA 2023 (PMID 38038395)
Non invasive ventilation in ARDS
"noninvasive ventilation"[MeSH Terms] AND "acute respiratory distress syndrome"[MeSH Terms]
non-invasive ventilation NIV HFNO CPAP ARDS evidence guidelines 2023 2024
"high flow nasal" OR "NIV" OR "CPAP" AND "ARDS" AND "intubation"
"high flow nasal cannula" AND "acute hypoxemic respiratory failure" AND ("ARDS" OR "intubation")
"patient self-inflicted lung injury" OR "P-SILI" AND "spontaneous breathing" AND ARDS
P-SILI patient self-inflicted lung injury NIV ARDS spontaneous breathing 2023 2024
non-invasive ventilation HFNC NIV ARDS algorithm flowchart
Non-Invasive Ventilation in ARDS
Overview
- High-Flow Nasal Cannula (HFNC)
- Continuous Positive Airway Pressure (CPAP) — via facemask or helmet
- Bilevel NIV (BiPAP/IPAP-EPAP) — via facemask or helmet
Why ARDS Is Different: Shunt Physiology
- Supplemental oxygen alone is largely ineffective — shunted blood cannot be oxygenated regardless of FiO₂
- PEEP (positive end-expiratory pressure) is the key therapeutic tool — it recruits flooded/collapsed alveoli and reduces shunt fraction
- As a result, most ARDS patients ultimately require invasive mechanical ventilation with lung-protective parameters
1. High-Flow Nasal Cannula (HFNC)
Mechanism of Action
- Delivers heated, humidified oxygen/air mixture at flows up to 60–80 L/min, eliminating room-air dilution
- Generates a modest but functional PEEP effect (approximately 1–3 cm H₂O in a closed-mouth patient, proportional to flow)
- Washout of nasopharyngeal dead space reduces CO₂ rebreathing
- Reduces inspiratory effort and work of breathing
- Better tolerated than facemask NIV (no claustrophobia, patient can speak/eat)
Typical Settings
- Initial: FiO₂ 50%, flow 40 L/min
- Titrate FiO₂ to target SpO₂; increase flow for dyspnea relief
- Maximum: FiO₂ 100%, flow 60 L/min
Key Trial Evidence
-
FLORALI trial (Frat JP et al., NEJM 2015, n=310): In non-intubated patients with acute hypoxemic respiratory failure without hypercapnia, altered mental status, or shock:
- HFNC → lower 90-day mortality HR vs. facemask NIV or standard oxygen
- Subgroup with P/F ≤ 200 showed particular benefit with HFNC vs. NIV
- NIV arm had the highest mortality in this P/F ≤ 200 subgroup — a pivotal finding
-
FLORALI-2 trial (2019): HFNC vs. NIV delivered via helmet — no significant difference in intubation rate; helmet NIV appeared more promising than facemask NIV
-
Murray & Nadel's notes: "NIV may generate harmful large tidal volumes in hypoxemic respiratory failure and lead to worsened injury to the lung... the higher mortality in patients undergoing NIV in the FLORALI study and in patients with a P/F ratio lower than 150 in the LUNG-SAFE series."
ESICM 2023 Recommendation on HFNC
- Suggests HFNC over conventional oxygen in non-intubated patients with P/F 100–300
- The recommendation for CPAP/NIV vs. HFNC remains unresolved
Monitoring for HFNC Failure: The ROX Index
| ROX Value | Interpretation |
|---|---|
| < 3.85 | High risk of HFNC failure → prepare for intubation |
| ≥ 4.88 at 2, 6, or 12 h | Lower risk of intubation |
2. CPAP (Continuous Positive Airway Pressure)
Mechanism
- Applies a single continuous positive pressure throughout inspiration and expiration
- Directly recruits alveoli, reduces shunt, improves oxygenation
- Does not reduce work of breathing in the same way as pressure-support NIV
Interface Options
- Facemask CPAP: standard, widely available
- Helmet CPAP: full-head interface, better tolerance for prolonged use, superior seal, no dead-space problem at high-flow rates
Evidence in ARDS

- Unable to make a recommendation for or against CPAP/NIV vs. conventional oxygen in general AHRF (high-level evidence for mortality, moderate for intubation)
- Suggests CPAP over COT to reduce intubation risk in COVID-19 ARDS specifically (low-certainty evidence)
- Cannot recommend CPAP to reduce mortality in COVID-19 AHRF (moderate-certainty evidence)
3. BiPAP/NIV (Bilevel Positive Airway Pressure)
Mechanism
- IPAP (inspiratory positive airway pressure): reduces work of breathing, augments tidal volume
- EPAP (expiratory positive airway pressure = PEEP): prevents end-expiratory collapse, recruits alveoli
Settings (Initial Guidance, Rosen's EM)
- Start IPAP 10 cm H₂O, EPAP 5 cm H₂O
- Titrate by 1–2 cm H₂O based on clinical response
- Maximum IPAP 20 cm H₂O (above this: discomfort, gastric insufflation)
- Full-face mask preferred over nasal mask (lower leak rate, better compliance)
Facemask vs. Helmet NIV
- Helmet NIV delivers better PEEP transmission, avoids facial pressure injury, and is better tolerated for prolonged use
- Helmet RCT (Patel et al., single-centre, n=83): Helmet NIV vs. facemask NIV in non-intubated ARDS → significantly reduced requirement for invasive ventilation and reduced mortality
- However, evidence base for helmet NIV remains limited to small trials
4. Patient Self-Inflicted Lung Injury (P-SILI): The Critical Concept
Mechanism
- The "baby lung" (small fraction of recruitable alveoli) receives the entirety of each tidal breath
- High transpulmonary pressure swings → regional lung overstretch and injury
- NIV can add a pressure boost on top of an already large spontaneous breath → further amplifies stress
- This creates a vicious cycle: lung injury → worse compliance → higher respiratory drive → worse P-SILI
Why HFNC and Helmet NIV Are Safer
- HFNC blunts respiratory drive by improving oxygenation and dead-space washout, with lower imposed tidal volumes
- Helmet CPAP provides sustained PEEP without adding inspiratory pressure boost → less amplification of effort-driven tidal volumes
- Facemask BiPAP adds inspiratory pressure during already-vigorous efforts → greatest P-SILI risk
5. Clinical Decision Framework by ARDS Severity
| Severity | P/F Ratio | NIV Approach | Evidence/Recommendation |
|---|---|---|---|
| Mild | 200–300 | HFNC, CPAP, or standard O₂ | Safe; reasonable first-line trial |
| Moderate | 100–200 | HFNC preferred over facemask NIV; close monitoring | Moderate support (FLORALI subgroup) |
| Severe | < 100 | Proceed to intubation | NIV/HFNC carry high failure risk and risk of P-SILI |
| Moderate-severe with specific indications | < 150 | Short trial of helmet CPAP/NIV acceptable | Limited RCT data; requires expert centre |
6. Indications for Urgent Intubation (Do Not Delay)
- Worsening hypoxemia despite NIV/HFNC (SpO₂ declining, FiO₂ requirement rising)
- High respiratory rate persisting > 30–35 bpm
- Worsening ROX index (< 3.85 or declining trajectory)
- Altered mental status / agitation — inability to protect airway
- Haemodynamic instability / shock
- Increasing work of breathing / accessory muscle use — impending fatigue
- Hypercapnia developing (not typical in early ARDS, but signals exhaustion)
- No improvement in P/F ratio after 1–2 hours of NIV
Delayed intubation in ARDS is associated with increased mortality. Patients who fail NIV and are then intubated have worse outcomes than those intubated early.
7. Special Populations
Immunocompromised Patients
- Early evidence favoured NIV to avoid intubation-related infections
- More recent large trials (including HIGH trial, n=778): HFNC vs. standard oxygen — no mortality difference
- When NIV fails in haematological malignancies: mortality > 70%
- Current guidance: Individualised approach; avoid dogmatic NIV use; helmet interface may be preferable
COVID-19 ARDS
- Large observational data supports HFNC and CPAP as safe early approaches in mild-moderate COVID ARDS
- CPAP conditionally preferred over standard O₂ for reducing intubation (ESICM 2023)
- Awake prone positioning combined with HFNC/NIV showed promising results in COVID ARDS to reduce intubation rate
Post-Operative Patients
- NIV/CPAP well established for post-operative respiratory failure (abdominal/thoracic surgery)
- Reduces intubation rate (33% vs. 46% in one large RCT) but no mortality benefit
- Prophylactic CPAP ≥ 6 hours post-surgery reduces pulmonary complications
8. Summary Table
| Modality | PEEP Generated | Work of Breathing Relief | P-SILI Risk | ARDS Evidence | Best For |
|---|---|---|---|---|---|
| Standard O₂ | None | None | Low | Inadequate alone | Very mild ARDS only |
| HFNC | Modest (1–3 cm H₂O) | Moderate | Low | Best (FLORALI) | Mild-moderate ARDS, 1st line |
| CPAP — Facemask | Set directly | None | Low-moderate | Moderate (COVID data) | Mild ARDS, resource-limited |
| CPAP — Helmet | Set directly | None | Low | Limited RCT | Moderate ARDS trial at expert centres |
| BiPAP — Facemask | EPAP | IPAP support | Highest | Harmful in P/F < 150 | Generally avoid in moderate-severe ARDS |
| BiPAP — Helmet | EPAP | IPAP support | Moderate | Promising small RCT | Select moderate ARDS at expert centres |
Key Take-Home Points
- HFNC is the preferred non-invasive approach in mild-to-moderate ARDS (P/F > 150–200)
- Facemask BiPAP should generally be avoided in moderate-severe ARDS due to P-SILI risk — the FLORALI data shows higher mortality in P/F < 150 with facemask NIV
- Helmet NIV/CPAP is the most promising NIV modality for moderate ARDS — better tolerance, lower P-SILI risk — but evidence is limited
- P-SILI is the unifying physiological concept explaining why NIV can harm in severe ARDS
- The ROX index (SpO₂/FiO₂ ÷ RR) guides real-time HFNC monitoring: < 3.85 = high failure risk
- Do not delay intubation — non-invasive failure in ARDS carries substantially higher mortality than early intubation
- In all cases, close monitoring at 1–2 hour intervals is mandatory
- Fishman's Pulmonary Diseases and Disorders — ARDS chapter (Reilly & Christie)
- Murray & Nadel's Textbook of Respiratory Medicine — NIV & ARDS section
- Rosen's Emergency Medicine — HFNC and ROX index
- ESICM 2023 ARDS Guidelines — Grasselli G et al., Intensive Care Med 2023 (PMID 37326646)
- Grieco DL et al. — NIV & HFNC in hypoxemic failure/ARDS, Intensive Care Med 2021 (PMID 34232336)
- Lassola S, Giani M, Bellani G — Noninvasive Respiratory Support in ARDS, Clin Chest Med 2024 (PMID 39443002)
- Marongiu I et al. — P-SILI evidence & monitoring, J Clin Med 2024 (PMID 39064059)