Laryngeal carcinoma
Reading File
Finding Sources
Finding Sources
Searching PubMed
"laryngeal carcinoma"[MeSH Terms] AND ("treatment"[MeSH Terms] OR "diagnosis"[MeSH Terms])
Reading File
Reading File
Reading File
Searching PubMed
"laryngeal cancer"[MeSH Terms] AND "treatment"[MeSH Terms]
Reading File
Searching Images
laryngeal carcinoma squamous cell cancer larynx
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
I now have comprehensive information. Let me compile a thorough answer.
Laryngeal Carcinoma
Epidemiology
Laryngeal cancer accounts for ~13,150 new diagnoses per year in the US (2018 data), with ~3,710 deaths. The male-to-female ratio is 3.9:1. Over 90% of cases occur in patients >40 years old. Race is an independent prognostic factor — African American patients present younger and have lower overall survival. Worldwide, laryngeal cancer incidence and mortality are declining, likely related to decreased tobacco use.
- 85–95% of all laryngeal malignancies are squamous cell carcinoma (SCC)
- Site distribution in the US: Glottis 51% > Supraglottis 33% > Subglottis 2% (14% uncategorized)
- Geographic variation: Glottic > Supraglottic in US, UK, Canada; reversed in France, Italy, Spain, Finland
— Cummings Otolaryngology Head and Neck Surgery
Risk Factors
| Factor | Notes |
|---|---|
| Tobacco | Dose-dependent; most important risk factor |
| Alcohol | Synergistic with tobacco — multiplicative increase in risk |
| Laryngopharyngeal reflux (LPR) | Independent risk factor for laryngeal/pharyngeal cancer |
| Occupational toxins | Higher incidence in unskilled manual workers with high alcohol + tobacco exposure |
| HPV | Role implicated, especially in younger patients |
| Diet | Low fruit/vegetable consumption increases risk |
The combination of tobacco and alcohol has a multiplicative (not merely additive) carcinogenic effect.
Histology
Normal tissue:
- Supraglottis: ciliated pseudostratified columnar epithelium
- Glottis: stratified squamous epithelium
- Subglottis: ciliated pseudostratified columnar epithelium
Squamous Cell Carcinoma Grades:
- Well-differentiated: keratinization, intercellular bridges, pleomorphic nuclei
- Moderately differentiated: less keratinization, more atypical nuclei
- Poorly differentiated: minimal keratinization, minimal intercellular bridges, numerous atypical nuclei
Verrucous carcinoma (1–2% of laryngeal cancers):
- Exophytic, well-differentiated keratinizing epithelium
- Pushing (not infiltrative) margins
- Does not metastasize unless foci of conventional SCC are present
- Surgical excision preferred; no nodal dissection needed; radiation not required
Non-SCC malignancies (rare): adenocarcinoma, adenoid cystic carcinoma, neuroendocrine tumors, mucoepidermoid carcinoma, chondrosarcoma, metastatic lesions
TNM Staging (AJCC 8th Edition)
Supraglottic
| Stage | Description |
|---|---|
| T1 | Limited to one subsite |
| T2 | >1 subsite or spread outside supraglottis; no vocal fold fixation |
| T3 | Vocal fold fixation OR invasion of postcricoid/pre-epiglottic space/paraglottic space/inner cortex of thyroid cartilage |
| T4a | Invades thyroid cartilage or beyond larynx (moderately advanced) |
| T4b | Invades prevertebral fascia, encases carotid, or invades mediastinum (very advanced) |
Nodal metastasis at presentation: 25–50%. Spreads typically to base of tongue or pre-epiglottic space. Level II–IV nodal involvement typical.
Glottic
| Stage | Description |
|---|---|
| T1a | One vocal fold, normal mobility |
| T1b | Bilateral vocal folds, normal mobility |
| T2 | Extends to subglottis or supraglottis, OR impaired mobility |
| T3 | Vocal fold fixation OR invasion of paraglottic space/inner cortex thyroid cartilage |
| T4a | Invades cricoid or thyroid cartilage or beyond larynx |
| T4b | Invades prevertebral fascia, encases carotid, or invades mediastinum |
Glottic cancer has very low rates of nodal metastasis (T1–2: 0–5%) due to sparse lymphatic drainage of the true vocal cords.
Subglottic
- Rare, aggressive, poorly differentiated
- Higher risk of level VI node spread and superior mediastinal metastasis
- May present with airway obstruction, stridor, dyspnea
N-staging update (AJCC 8th Ed.)
Extranodal extension (ENE) incorporated:
- N2a: Pathologic ENE in single ipsilateral node <3 cm
- N3b: ENE in node >3 cm or multiple ipsilateral/bilateral nodes with ENE
Distant Metastasis
Hematogenous spread: Lungs > Liver > Skeletal system
Lymphatic spread: Mediastinum
Higher risk with advanced stage, neck disease, locoregional recurrence, supraglottic/subglottic primary
Clinical Presentation
- Glottic: Hoarseness (early symptom due to vocal cord involvement) → airway compromise
- Supraglottic: Dysphagia, throat pain, referred otalgia, neck mass — often presents late
- Subglottic: Stridor, dyspnea, airway obstruction
Diagnosis & Imaging
Direct laryngoscopy + biopsy is the primary diagnostic tool.
CT is the first-line imaging modality — widely available, reproducible, evaluates:
- Paraglottic space involvement
- Cartilage invasion
- Lymph node status
- Deep soft tissue extension
MRI advantages: superior soft tissue contrast, multiplanar display, no ionizing radiation. Disadvantages: slower, prone to motion artifact, costly, poor cortical bone imaging.
PET/CT: Recommended at 10–12 weeks post-treatment to assess response and differentiate scar/fibrosis from residual tumor (earlier timing increases false-positive rate).
Endoscopic Image: Supraglottic SCC
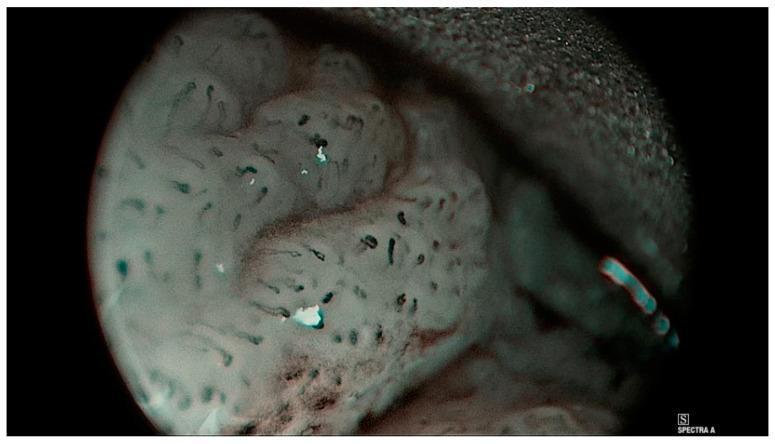
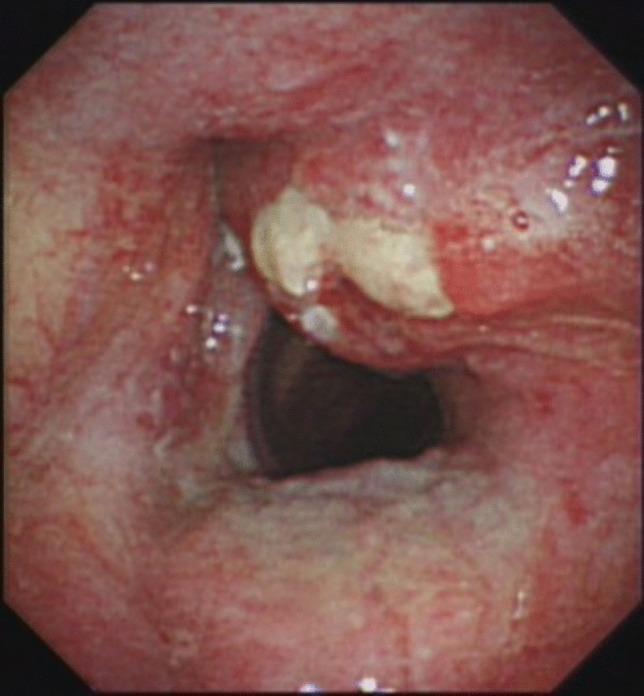
Treatment
Early Laryngeal Cancer (Stage I/II)
Single-modality treatment:
Radiation Therapy:
- 6-week course, total 60–70 Gy
- Local control: 90–98% for T1 and select T2 lesions
- Higher failure (up to 30%) for T2 lesions with impaired vocal fold mobility
- Advantages: voice preservation, avoids tracheotomy, suitable for poor surgical candidates
- Disadvantages: mucositis, laryngeal edema, dysphagia, xerostomia, chondronecrosis risk, difficulty detecting recurrence
Transoral Laser Microsurgery (TLM):
- Replaced open partial laryngectomy as preferred surgical approach
- Transoral robotic partial laryngectomy increasingly reported
- Advantages: may avoid irradiation entirely; similar local control to radiation
- Disadvantages: poorer voice outcomes vs. radiation
- Contraindications: both arytenoid joints involved, infraglottic extension to cricoid, hyoid bone invasion, posterior arytenoid mucosa involvement
Advanced Laryngeal Cancer (Stage III/IV)
Combined modality approach. Organ preservation is the primary goal where functionally feasible.
Concurrent chemoradiation (CRT):
- Standard for most T3 lesions
- Cisplatin is the key radiosensitizing agent
Total laryngectomy (TL) + adjuvant therapy:
- For T4 disease with cartilage invasion or nonfunctional larynx
- Also used for salvage after CRT failure
Landmark Trials:
| Trial | Key Finding |
|---|---|
| VA Laryngeal Study (1991) | Induction chemo (cisplatin/5-FU) → RT vs. TL + postop RT → equivalent 2-year survival (68% both groups); 64% laryngeal preservation in chemo arm |
| RTOG 91-11 (2003) | Concurrent CRT > induction chemo + RT > RT alone for laryngeal preservation; T4 with cartilage invasion fared worse with nonsurgical approach |
Neck management:
- Glottic T1–T2: No elective neck treatment (0–5% occult disease)
- Glottic T3–T4: Treat neck, especially with transglottic extension
- Supraglottic: Bilateral neck treatment (elective bilateral level II–IV dissection or bilateral radiation fields; 30–50% occult disease even in early disease)
- Post-CRT neck: Planned neck dissection NOT necessary if complete response on PET/CT at 12 weeks (Level I evidence)
Adjuvant Therapy
Indications for adjuvant radiation or chemoradiation after surgery: positive surgical margins, extranodal extension — meta-analysis shows improved overall survival in these settings.
Voice Rehabilitation After Total Laryngectomy
| Method | Notes |
|---|---|
| Tracheoesophageal prosthesis (TEP) | Best voice outcomes; first-line |
| Esophageal speech | Difficult to learn; uses trapped/expelled air |
| Electrolarynx | Mechanical quality; less preferred by patients |
Complications
Surgery:
- Pharyngocutaneous fistula (risk ↑ with prior radiation; ↓ with regional/free flap)
- Stomal stenosis (may indicate stomal recurrence)
- Dysphagia from pharyngeal stenosis
Chemoradiation:
- Esophageal stenosis
- Nonfunctional larynx (may require tracheotomy, G-tube, or salvage TL)
- Chondritis (especially risk with post-CRT biopsy)
Surveillance After Treatment
- Year 1: every 1–3 months
- Year 2: every 2–4 months
- Years 3–5: every 4–6 months
- Thereafter: every 6–12 months
- Thyroid function tests every 6–12 months (20–65% hypothyroidism incidence depending on treatment)
Recurrent/Metastatic Disease
- ~50% of recurrences can be salvaged
- Signs: increased edema, impaired vocal fold mobility, mass lesion, ulceration, worsening dysphonia/dysphagia
- PET/CT at 10–12 weeks post-therapy for assessment
- Biopsy confirmation required for suspected recurrence
- Salvage often requires TL (multifocal/insidious recurrence patterns)
- Systemic therapy: cisplatin-based regimens; anti-EGFR (cetuximab); checkpoint inhibitors (pembrolizumab, nivolumab) for recurrent/metastatic PD-L1+ disease
Sources: K J Lee's Essential Otolaryngology (10th Ed.) • Cummings Otolaryngology Head and Neck Surgery
This is a shared conversation. Sign in to Orris to start your own chat.