H pylori eradication therapy
Reading File
Finding Sources
Searching PubMed
"Helicobacter pylori" AND "eradication therapy"
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
I now have comprehensive information to provide a thorough answer. Let me compile it.
H. pylori Eradication Therapy
Goals & Definitions
Eradication is defined as a negative test ≥4 weeks after completing therapy. Successful eradication reduces ulcer recurrence from 50–90% (with acid suppression alone) to <5% and significantly lowers gastric cancer risk. An acceptable first-line cure rate is ≥90%.
Choosing a Regimen: Local Resistance Matters
The critical threshold is clarithromycin resistance in the local population:
| Clarithromycin Resistance | Recommended First-Line |
|---|---|
| <15% (low) | Clarithromycin-based triple therapy OR bismuth quadruple |
| >15% (high) | Avoid clarithromycin-based triple therapy → use bismuth-based or non-bismuth quadruple |
Patients with prior personal exposure to clarithromycin or macrolides should be treated as high-resistance regardless of local rates.
First-Line Regimens
1. Standard Triple Therapy (for low-resistance areas only)
Duration: 14 days (7–10 days is inferior; 14 days improves eradication by ~5%)
| Regimen | Drugs | Doses |
|---|---|---|
| PAC | PPI + Amoxicillin + Clarithromycin | PPI standard dose BD; Amoxicillin 1 g BD; Clarithromycin 500 mg BD |
| PCM (penicillin allergy) | PPI + Clarithromycin + Metronidazole | PPI BD; Clarithromycin 250–500 mg BD; Metronidazole 400 mg BD |
- Double-dose PPI (e.g., omeprazole 40 mg BD) increases eradication rates by ~10%
- PPI raises gastric pH → H. pylori enters replicative state → more susceptible to antibiotics
- Cure rates with standard triple therapy now often <80% in many countries due to rising clarithromycin resistance
2. Bismuth-Based Quadruple Therapy (preferred in high-resistance areas; also recommended as universal first-line by some guidelines)
Duration: 14 days
| Component | Drug | Dose |
|---|---|---|
| PPI | Any standard PPI | Standard dose BD |
| Bismuth | Bismuth subsalicylate or subcitrate | 120–300 mg QID |
| Antibiotic 1 | Tetracycline HCl | 500 mg QID (or via Pylera/Helidac) |
| Antibiotic 2 | Metronidazole | 400 mg TID (or 375 mg QID in Pylera) |
- Eradication rates 85–90% first-line; 79–88% as second-line
- Metronidazole resistance has less clinical impact than clarithromycin resistance (resistance is only partial)
- Drawback: up to 17–34 pills/day; Pylera (combination capsule) simplifies to 14 pills/day
3. Non-Bismuth Quadruple (Concomitant) Therapy
PPI + Amoxicillin 1 g BD + Clarithromycin 500 mg BD + Metronidazole 400 mg BD — all given together for 14 days. Highly effective when dual clarithromycin+metronidazole resistance is low.
Sequential and Hybrid variants exist but are generally less effective than concomitant therapy.
Resistance-Based Decision Algorithm (Maastricht V/Florence)
(High clarithromycin resistance area)
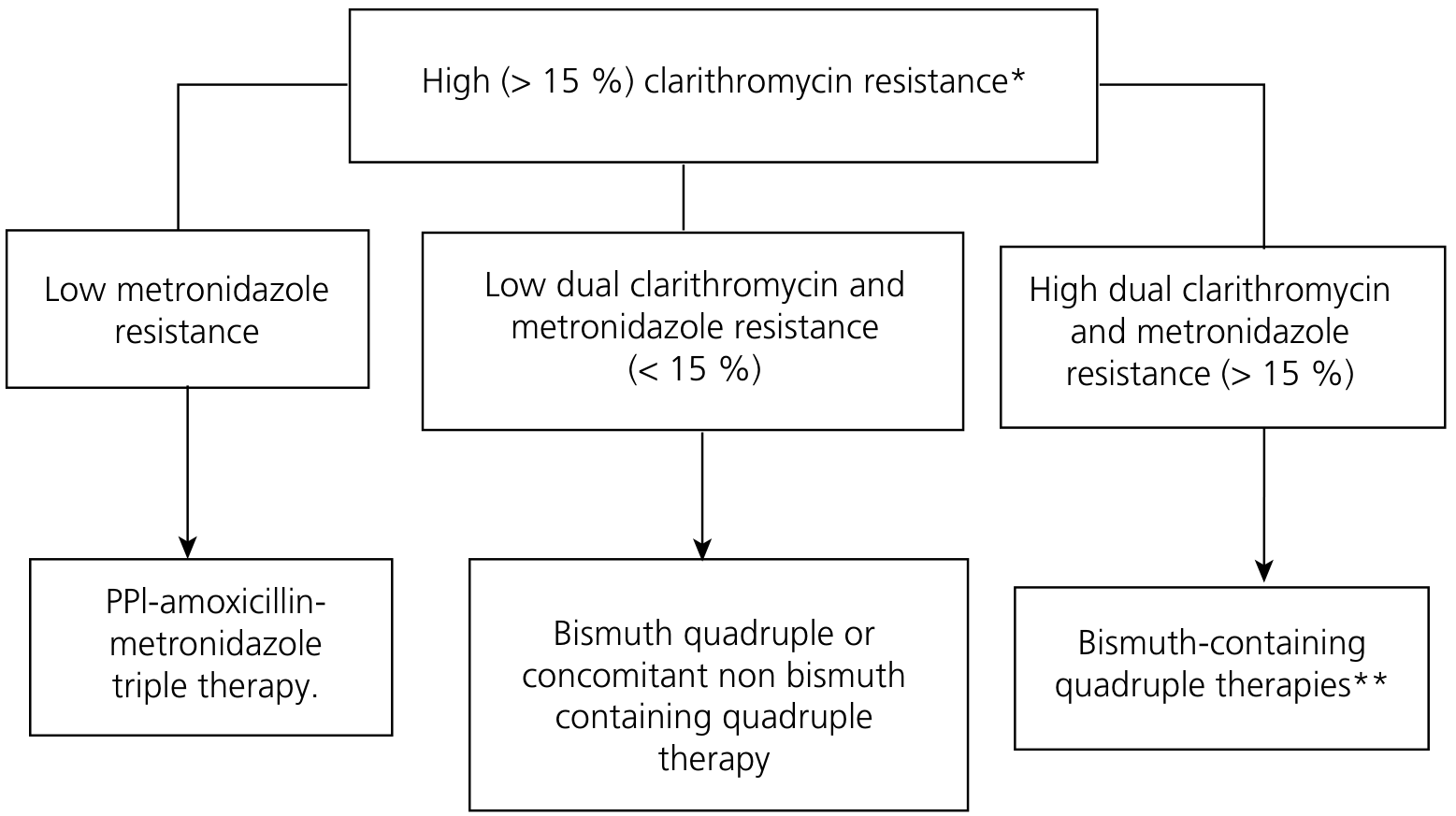
Second-Line / Rescue Therapy
If first-line fails, avoid any antibiotic used in the initial regimen (assume resistance):
| Regimen | Notes |
|---|---|
| Bismuth quadruple | If not used first-line |
| Levofloxacin triple | PPI + Levofloxacin 500 mg BD + Amoxicillin 1 g BD × 10–14 days; eradication ~74–86% as second-line |
| Rifabutin triple | PPI + Rifabutin + Amoxicillin; useful after multiple failures; limited by bone marrow toxicity |
| High-dose dual | PPI (high dose) + Amoxicillin 1 g TID–QID × 14 days |
Levofloxacin resistance is increasing (now 10–50% in many regions) — culture-guided therapy is ideal for repeat failures.
Vonoprazan-Based Regimens (Emerging)
Vonoprazan (potassium-competitive acid blocker, 20 mg BD) is more potent and less pH-dependent than PPIs. A 2024 meta-analysis (PMID 39533409) confirms high efficacy in dual/triple/quadruple vonoprazan regimens both as first-line and rescue therapy — now incorporated into some guidelines (Goldman-Cecil recommends vonoprazan as PPI alternative).
Confirming Eradication
- Test ≥4–6 weeks after completing therapy (and ≥2 weeks off PPI for breath test)
- Preferred: Urea breath test or validated stool antigen test (HpSA)
- Serology has no role in confirming eradication (remains positive post-treatment)
- Endoscopy-based tests are acceptable alternatives if endoscopy is planned and patient is off PPI
Patient Counselling & Adherence
Non-compliance is the primary cause of treatment failure and leads to acquired antibiotic resistance. Key points to cover:
- Side effects are common (~50%) but usually mild: metallic taste (metronidazole/clarithromycin), diarrhoea, blackened stools (bismuth), photosensitivity (tetracycline)
- Disulfiram-like reaction with alcohol + metronidazole
- Stop only for severe allergic reaction or severe watery diarrhoea (C. diff)
- Written dosing instructions improve adherence
Adjuvants
- Probiotics (e.g., Lactobacillus species) added to standard triple therapy hasten symptom resolution and modestly increase eradication rates
Key Antibiotic Resistance Summary
| Antibiotic | Resistance Rate | Clinical Impact |
|---|---|---|
| Clarithromycin | 10–50% (rising globally) | High — major determinant of triple therapy failure |
| Metronidazole | 20–80% globally | Partial — less impact on quadruple therapy |
| Levofloxacin | 10–50% | Significant — limits second-line use |
| Amoxicillin | Rare | Minimal |
| Tetracycline | Rare | Minimal |
Recent evidence update: A 2025 systematic review (PMID 39824392, Gastroenterology) confirms that H. pylori eradication significantly reduces gastric cancer risk; and a 2024 meta-analysis (PMID 39533409, Helicobacter) supports vonoprazan-based regimens as effective alternatives to PPI-based therapy.
— Yamada's Textbook of Gastroenterology, 7th ed. | Goldman-Cecil Medicine | Lippincott Pharmacology
This is a shared conversation. Sign in to Orris to start your own chat.