Takayasu arteritis follow-up investigations
Reading File
Finding Sources
Searching PubMed
"Takayasu arteritis" AND "follow-up" AND monitoring
Reading File
Finding Sources
Reading Article
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Searching the Web
Takayasu arteritis follow-up investigations monitoring guidelines 2024 2025
I now have comprehensive information from multiple authoritative sources. Let me compile the complete answer.
Takayasu Arteritis: Follow-up Investigations
Takayasu arteritis (TA) is a chronic relapsing granulomatous large-vessel vasculitis — remission is common but relapse occurs in up to 40–60% of patients, making structured follow-up essential. There is no single gold-standard test; monitoring requires an integrated approach combining clinical assessment, laboratory markers, and serial imaging.
1. Clinical Assessment
Every follow-up should include:
- Blood pressure (both arms) — detect new asymmetry (>10 mmHg difference) suggesting progressive subclavian or aortic disease
- Peripheral pulse examination — loss or reduction of previously palpable pulses indicates disease progression
- Auscultation for bruits — over carotid, subclavian, renal, and aortic territories
- Symptoms review — claudication (upper > lower limb), carotidynia, headache, visual symptoms, renovascular hypertension, chest/abdominal pain
The Disease Extent Index – Takayasu (DEI.Tak) score has been developed to quantify disease activity by clinical findings alone, though one study found it missed active disease (by physician assessment) in ~14% of patients with a score of 0 — emphasising its limitations as a standalone tool.
2. Laboratory Investigations
No specific biomarker reliably mirrors vascular inflammation in TA. Labs are used adjunctively:
| Test | Role | Limitations |
|---|---|---|
| ESR | Reflects systemic inflammation; serial trend useful | Does not correlate with PET activity or patient-reported outcomes in LVV |
| CRP | Modest correlation with disease activity; more sensitive than ESR for acute flares | Can be normal in active disease |
| FBC | Anaemia of chronic disease (mild), thrombocytosis in active disease | Non-specific |
| γ-globulins / immunoglobulins | Elevated in active disease | Non-specific |
| Renal function (eGFR, creatinine) + urinalysis | Detect renal artery stenosis–mediated ischaemic nephropathy | Structural, not inflammatory |
| Pentraxin-3 (PTX3) | More specific than CRP for vascular wall inflammation; studied but not yet in routine use | Not widely available |
| IL-6, MMP-3, MMP-9 | Proposed biomarkers; elevated in active TA | Not validated for clinical practice |
Key point: ESR and CRP are unreliable markers of vascular disease activity. Normal inflammatory markers do not exclude active arteritis, and imaging remains essential for relapse detection. — Brenner and Rector's The Kidney; Current Surgical Therapy 14e
3. Imaging Investigations
Imaging is the cornerstone of follow-up. EULAR 2023 and ACR/VF 2021 guidelines both emphasise that imaging should not be used routinely at every visit, but is indicated when relapse is suspected, inflammatory markers are unreliable, or for periodic structural damage assessment.
3a. MRI / MR Angiography (MRA) — Preferred modality for TA
- MRI is the preferred imaging modality per EULAR 2023 recommendations for TA
- Demonstrates wall oedema (active inflammation), wall thickening, gadolinium enhancement, and structural changes (stenosis/aneurysm)
- MRA provides luminography — identifies stenoses, occlusions, aneurysms, and collaterals
- No ionising radiation — preferred for serial follow-up studies (especially in young women)
- Limitation: Wall changes do not always correlate with disease activity; gadolinium avoidance needed in renal impairment
3b. CT Angiography (CTA)
- High-resolution assessment of vessel wall thickening, calcification, stenosis, aneurysms
- Preferred when MRI is contraindicated or unavailable
- Limitation: Radiation exposure — a concern given repeated imaging in young patients
- May demonstrate mural thickening with enhancement during active inflammation
3c. ¹⁸F-FDG PET/CT or PET/MRI
- Detects metabolically active vascular inflammation even before structural changes appear
- Particularly useful to differentiate active ("hot") from burned-out ("cold") disease
- Used for evaluating suspected relapse and assessing response to therapy
- Limitation: Not validated as a reliable measure of disease activity in follow-up; FDG uptake may reflect chronic remodelling; not widely available; radiation exposure
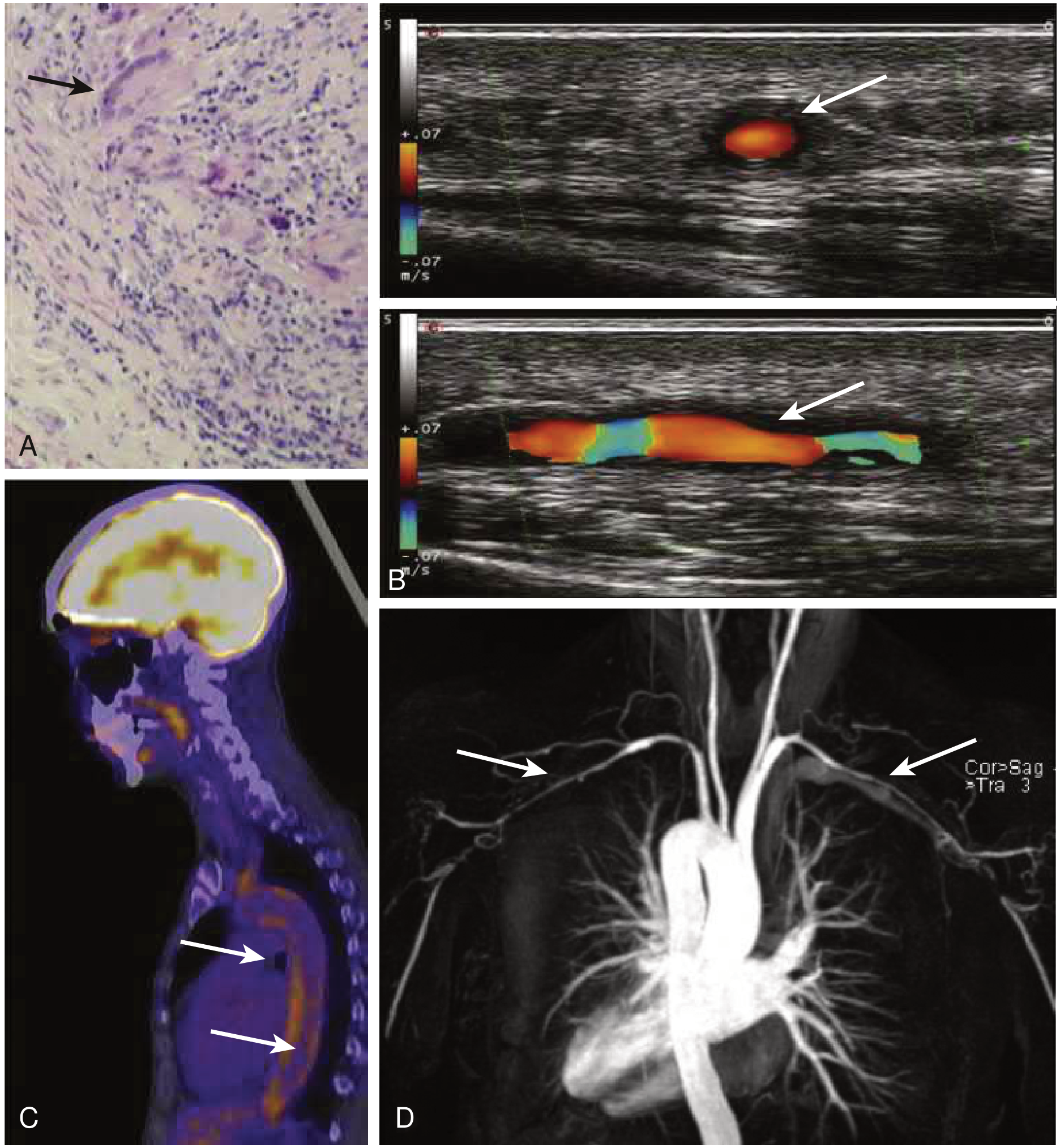
3d. Vascular Ultrasound (Duplex/Colour Doppler)
- Assesses carotid, subclavian, axillary, and renal arteries for wall thickening, flow velocity (stenosis), and intima-media thickness
- Useful at bedside and in serial monitoring — no radiation
- Detects the "macaroni sign" (circumferential wall thickening) in large vessels
- Limited to accessible vessels; operator-dependent; cannot image thoracic aorta directly
3e. Conventional (Catheter) Angiography
- Previously the gold standard; now largely replaced by CTA/MRA
- Provides luminography only — no vessel wall information
- Reserved for pre-intervention planning (angioplasty, stenting, bypass)
- Noninvasive imaging is preferred per ACR/VF 2021 and EULAR guidelines
Summary: Imaging Choice by Purpose
| Purpose | Preferred Modality |
|---|---|
| Routine structural monitoring (long-term) | MRA or CTA |
| Detecting active inflammation / suspected relapse | MRI with contrast or FDG-PET/CT |
| Monitoring accessible vessel territories (carotid, subclavian) | Duplex ultrasound |
| Pre-interventional planning | CTA or catheter angiography |
| Serial follow-up (radiation-sparing) | MRA |
4. Echocardiography
- Assess for aortic root dilation and aortic regurgitation (AR) — occurs in ~18% of TA patients due to ascending aortic inflammation
- AR requiring valve replacement affects approximately 15% of patients
- Also evaluates left ventricular function, coronary artery origins (coronary arteritis), pulmonary arterial pressure (pulmonary artery involvement in up to 50%)
- Annual or biennial echocardiography is recommended in patients with aortic involvement
5. Blood Pressure Monitoring
- Serial bilateral arm BP measurement at every visit
- Ambulatory blood pressure monitoring (ABPM) for accurate assessment where subclavian/axillary disease affects peripheral readings
- Renal artery stenosis is a common cause of renovascular hypertension — renal duplex Doppler or CTA/MRA of renal arteries if BP is difficult to control
6. Ophthalmologic Review
- Indicated in patients with carotid/vertebral involvement or symptoms
- Fundoscopy for hypertensive retinopathy, retinal vein/artery occlusion, optic atrophy
- Visual field assessment if visual symptoms occur
7. ACR/VF 2021 Recommendations for Monitoring (TA in Remission)
Per the ACR/VF 2021 Guideline:
- Strong recommendation: Long-term clinical monitoring over no monitoring in patients in apparent remission
- Inflammatory markers (ESR/CRP) to assist monitoring, acknowledging limitations
- Noninvasive imaging over catheter angiography
- In remission with asymptomatic elevation of inflammatory markers: increase monitoring frequency rather than automatically escalating immunosuppression
- In remission with new vascular inflammation on imaging (new stenosis or wall thickening): conditionally recommend treating with immunosuppression
8. EULAR 2023 Imaging Recommendations (Relevant to TA Follow-up)
Per Dejaco et al., Ann Rheum Dis 2024 [PMID: 37550004]:
- MRI is the preferred imaging modality for TA (both diagnosis and follow-up)
- FDG-PET, CT, or ultrasound are acceptable alternatives
- Imaging not routinely recommended for follow-up — use when relapse is suspected or inflammatory markers are unreliable
- MR-angiography, CT-angiography, or ultrasound for long-term structural damage monitoring at sites of prior vascular inflammation
Summary Table: Follow-up Investigations in TA
| Domain | Investigation | Frequency |
|---|---|---|
| Clinical | BP (bilateral), pulses, bruits, symptoms | Every visit |
| Inflammatory markers | ESR, CRP, FBC | Every 1–3 months (active); every 3–6 months (remission) |
| Renal function | eGFR, urine protein:creatinine | Every 3–6 months |
| Structural imaging | MRA or CTA (whole aorta + branches) | Annually or at relapse; MRA preferred |
| Functional imaging | FDG-PET/CT | When relapse suspected and labs are unhelpful |
| Cardiac | Echocardiography | Annually if aortic root/valve involved |
| Ophthalmology | Fundoscopy, visual fields | If symptomatic or carotid disease present |
| Vascular lab | Duplex ultrasound (accessible vessels) | At follow-up visits for targeted territories |
Sources: Harrison's Principles of Internal Medicine 22e (2025) · Braunwald's Heart Disease · Washington Manual of Medical Therapeutics · Current Surgical Therapy 14e · Comprehensive Clinical Nephrology 7e · Grainger & Allison's Diagnostic Radiology · EULAR 2023 Imaging Recommendations [PMID: 37550004] · ACR/VF 2021 Guidelines
This is a shared conversation. Sign in to Orris to start your own chat.