Acute suppurative otitis media
Reading File
Finding Sources
Finding Sources
Searching PubMed
"acute otitis media" AND management
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Searching Images
acute otitis media tympanic membrane bulging erythema
Reading File
Reading File
Loading Image
Reading File
Reading File
Reading File
Reading File
I now have comprehensive data from multiple authoritative sources. Let me compile the full answer:
Acute Suppurative Otitis Media (AOM)
Acute suppurative (acute purulent) otitis media is an acute bacterial infection of the middle ear space characterized by vascular dilation, mucosal edema, exudation, bacterial proliferation, leukocyte infiltration, and pus formation behind the tympanic membrane.
Otoscopic Appearances
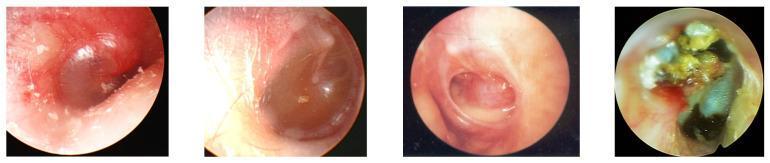
Epidemiology
- Most common bacterial infection of childhood and the third most common ED diagnosis in children under 15
- ~85% of all children experience at least one episode; 60% by age 3
- Peak incidence: 6–24 months of age; global incidence ~10.8 new episodes/100 people/year
Risk factors:
| Category | Factors |
|---|---|
| Environmental | Daycare attendance, tobacco smoke exposure, pacifier use, siblings, supine bottle feeding, lack of breastfeeding |
| Anatomic | Eustachian tube dysfunction, cleft palate, craniofacial syndromes (Treacher Collins), Down syndrome |
| Demographic | Male sex, indigenous populations (Native American, Inuit, Aboriginal Australian), low socioeconomic status, premature birth |
| Protective | Breastfeeding in infancy, pneumococcal conjugate vaccine |
Pathophysiology
AOM follows a viral URI → Eustachian tube dysfunction → middle ear bacterial superinfection sequence:
- Viral URI (RSV, rhinovirus, coronavirus, influenza, enterovirus, adenovirus, hMPV) causes nasopharyngeal and Eustachian tube inflammation and edema
- Eustachian tube obstruction impairs mucociliary clearance, drainage, and pressure equalization
- Fluid accumulates in the middle ear, providing a culture medium
- Nasopharyngeal bacteria reflux into the middle ear via the dysfunctional tube
- Bacterial proliferation → acute suppurative inflammation, pus formation, increased intratympanic pressure
Why children are more susceptible: The pediatric Eustachian tube is shorter (~13 mm vs. 35 mm in adults), wider, more floppy, and nearly horizontal (~10° vs. 45° in adults) — facilitating pathogen transmission from the nasopharynx to the middle ear. These anatomical differences resolve progressively with growth.
Biofilms: Early nasopharyngeal colonization with otopathogens, particularly S. pneumoniae, promotes biofilm formation in the middle ear, explaining recurrence and chronicity.
Microbiology
| Pathogen | Notes |
|---|---|
| Streptococcus pneumoniae | Historically most common (~30%); PCV7/PCV13 reduced vaccine-serotype disease; serotype 19A is highly multidrug-resistant; resistance via altered PBPs |
| Nontypeable Haemophilus influenzae | ~29%; now more prevalent post-PCV; 50% are β-lactamase positive |
| Moraxella catarrhalis | ~10%; 100% β-lactamase positive |
| Streptococcus pyogenes (Group A) | ~3.7% |
| Viruses | Isolated in up to 75% of AOM aspirates (RSV, rhinovirus, coronavirus, parainfluenza, enterovirus, adenovirus) |
PCV7 (2000) and PCV13 (2010) significantly reduced absolute S. pneumoniae burden and invasive complications, but increased the proportional share of nontypeable H. influenzae and M. catarrhalis.
Clinical Features
Symptoms:
- Rapid-onset otalgia (often <48 h onset) — hallmark symptom
- Fever (common but not universal; fever ≥40.5°C should prompt alternative diagnoses)
- Irritability, ear tugging/rubbing in infants
- Otorrhea (if TM has perforated — this confirms diagnosis)
- Conductive hearing loss, anorexia
Signs on otoscopy (pneumatic otoscopy is the key diagnostic tool):
- Moderate to severe bulging of the tympanic membrane (most specific)
- Erythema and opacification of the TM (erythema alone insufficient — crying/fever also cause it)
- Obscured landmarks (manubrium of malleus, cone of light)
- Reduced or absent TM mobility on pneumatic otoscopy (also seen in OME/perforation)
- Air-fluid level or purulent effusion behind the TM
- If TM has ruptured: purulent otorrhea in the canal
Diagnostic Criteria (AAP 2013)
AOM should be diagnosed in any of the following three clinical scenarios:
| Scenario | Criteria |
|---|---|
| 1 | Moderate to severe bulging of the TM |
| 2 | Mild bulging of TM + (acute ear pain <48 h OR intense erythema of TM) |
| 3 | New-onset otorrhea not due to otitis externa |
AOM should not be diagnosed in children without middle-ear effusion. Erythema alone is insufficient.
Tympanocentesis is the diagnostic gold standard but is not routinely performed outside of specialist settings; it is used when: culture is needed, treatment is failing, or complications are suspected.
Severity Classification
Severe AOM: moderate-to-severe otalgia, otalgia lasting ≥48 h, or temperature >102.2°F (39°C)
Treatment
1. Pain Control (mandatory regardless of antibiotic decision)
- Oral: Acetaminophen or ibuprofen (NSAIDs) — first-line for mild-moderate pain
- Topical: Benzocaine drops — brief additional relief only
- Myringotomy (with or without tympanostomy tube): relieves intratympanic pressure immediately; useful for severe pain, failed antibiotic treatment, or need for culture
2. "Watch and Wait" (Observation Option)
Up to 80% of AOM resolves without antibiotics. Observation for 48–72 h is appropriate in:
| Age | Criteria for Observation |
|---|---|
| 6 months–2 years | Unilateral, nonsevere AOM in otherwise healthy children |
| ≥2 years | Nonsevere symptoms (mild otalgia, afebrile or low-grade fever) |
Requires compliant caregivers with ready access to healthcare. Observation does not increase rates of mastoiditis.
3. Antibiotic Indications
Antibiotics are indicated when:
| Age | Indication |
|---|---|
| <6 months | All cases |
| 6 months–2 years | Bilateral AOM; any otorrhea |
| ≥6 months | Otorrhea |
| Any age | Severe otalgia, otalgia ≥48 h, or fever >102.2°F |
| >2 years | Symptoms not improving within 48–72 h of observation |
Benefit/harm context: Antibiotics decrease pain by day 2–3 (NNT = 20); but cause vomiting, diarrhea, or rash in 1 in 14 patients (NNH = 14). Prevention of mastoiditis requires treating ~5,000 patients per case prevented.
4. Antibiotic Regimens
First-line:
- Amoxicillin high-dose: 90 mg/kg/day divided TID (max 3 g/day) — achieves middle ear concentrations overcoming intermediate-resistant pneumococcal strains (MIC 0.1–1 μg/mL)
When amoxicillin should NOT be used:
- Amoxicillin received in the past 30 days
- Concurrent purulent conjunctivitis (suggests H. influenzae — "otitis-conjunctivitis syndrome")
- Penicillin allergy
Second-line / amoxicillin failure after 48–72 h:
- Amoxicillin-clavulanate (covers β-lactamase-producing H. influenzae and M. catarrhalis)
- Oral cephalosporins: cefdinir, cefuroxime, cefpodoxime
- IM ceftriaxone for 3 days (for persistent failure, vomiting, or penicillin allergy)
Duration:
| Age | Duration |
|---|---|
| <6 years | 10 days |
| ≥6 years (mild-moderate) | 5–7 days |
Highly resistant pneumococci (MIC >2 μg/mL) will not respond to amoxicillin.
Adults with AOM: Amoxicillin is the drug of choice; also treat with decongestants and analgesics regardless of bilaterality.
Complications
Acute (Local/Extracranial)
| Complication | Notes |
|---|---|
| TM perforation | Most common acute complication; usually heals spontaneously |
| Acute mastoiditis | Rare; postauricular tenderness, mass, or protrusion of ear — coalescent form requires mastoidectomy |
| Facial nerve palsy | Canal fallopian dehiscence → bacterial spread |
| Labyrinthitis | Sensorineural hearing loss, vertigo |
| Petrous apicitis | Gradenigo's triad: retro-orbital pain, abducens palsy, otorrhea |
| Bezold's abscess | Pus tracks along SCM/digastric through tip of mastoid |
Intracranial (Rare but Life-Threatening)
| Complication |
|---|
| Meningitis |
| Epidural/subdural abscess |
| Brain (intraparenchymal) abscess |
| Lateral sinus (sigmoid sinus) thrombosis |
| Otitic hydrocephalus |
Infection spreads via direct extension through bone or hematogenously.
Chronic Sequelae
- Chronic OME (middle ear effusion persisting >3 months): present in 60–70% of children after AOM; most resolve within 3 months
- Recurrent AOM (≥3 episodes in 6 months, or ≥4 in 1 year) → indication for tympanostomy tubes
- Conductive hearing loss (from persistent effusion)
- Acquired sensorineural hearing loss (from inner ear inflammation in recurrent/chronic disease)
- Cholesteatoma (keratin accumulation in middle ear; can destroy ossicles, cause CN palsies, labyrinthitis, intracranial spread)
Recurrent AOM — Prevention
- Tympanostomy tubes: Modest benefit (~1.5 fewer AOM episodes per child in first 6 months); indicated for recurrent AOM or persistent OME
- Antibiotic prophylaxis: No longer recommended (resistance concern; effect lost after discontinuation)
- Pneumococcal conjugate vaccine (PCV13/Prevnar 13): Reduces overall AOM burden and invasive complications
- Breastfeeding in infancy is protective
Sources
- Cummings Otolaryngology Head and Neck Surgery, blocks 31, 44
- Harrison's Principles of Internal Medicine, 22nd Ed. (2025), block 4
- K.J. Lee's Essential Otolaryngology, 11th Ed., block 2
- Tintinalli's Emergency Medicine, block 10
- Red Book 2021 (AAP), block 7
- Berek & Novak's Gynecology (sinusitis comparative context)
Recent evidence note: A 2025 Cochrane review (PMID 41307300) found decongestants and antihistamines do not improve AOM outcomes in children and are not recommended. A 2025 Cochrane review (PMID 40484402) confirmed topical antibiotics are preferred over systemic for chronic suppurative OM — relevant to post-AOM perforation management.
This is a shared conversation. Sign in to Orris to start your own chat.