ASTHMA COPD
Asking for Preferences
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Searching PubMed
asthma COPD management guidelines
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Loading Image
Searching Images
COPD emphysema pathology diagram
I now have comprehensive information to deliver a thorough, well-structured overview. Let me compile everything.
Asthma & COPD: A Comprehensive Overview
1. Definitions
| Feature | Asthma | COPD |
|---|---|---|
| Core defect | Reversible airway hyperresponsiveness | Irreversible (or poorly reversible) airflow obstruction |
| Key subtypes | Atopic, non-atopic | Emphysema, chronic bronchitis (often coexist) |
| Progression | Usually NOT progressive | Progressive, especially with continued smoking |
| Typical age of onset | Often childhood/young adult | >40 years, typically |
2. Pathophysiology
Asthma
Airflow obstruction in asthma results from three concurrent mechanisms:
- Bronchial smooth muscle contraction (bronchoconstriction)
- Bronchial wall inflammation
- Increased mucus secretion
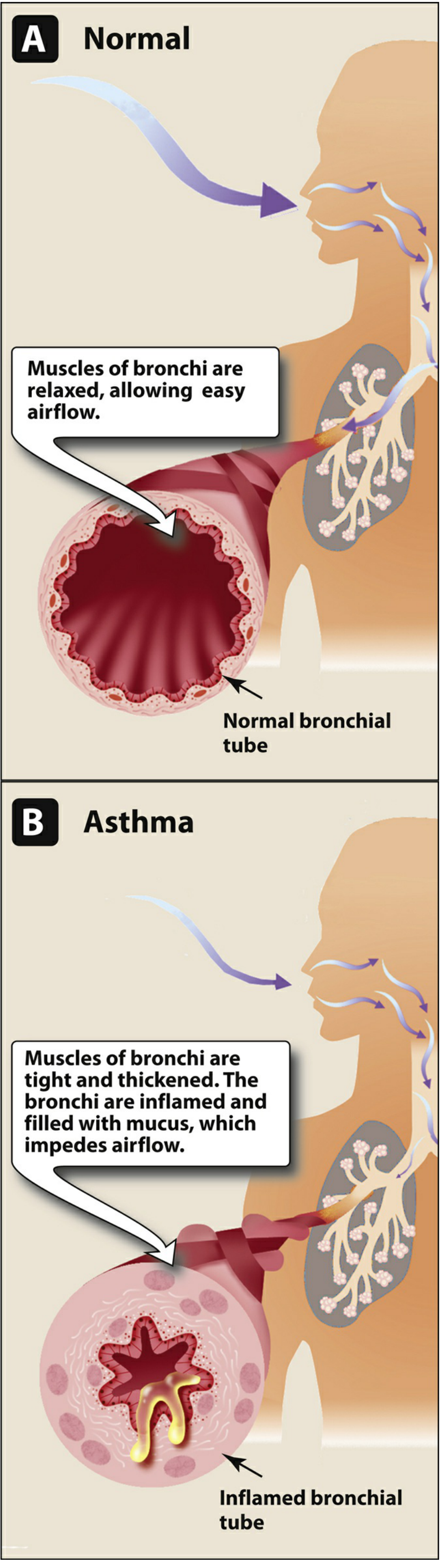
Atopic (allergic) asthma — a Th2/IgE-mediated reaction to environmental allergens:
- Early-phase reaction: mast cell degranulation → histamine, leukotrienes → immediate bronchoconstriction
- Late-phase reaction: eosinophil/lymphocyte infiltration driven by Th2 cytokines (IL-4, IL-5, IL-13)
Non-atopic asthma triggers: viral infections, cold air, exercise, pollutants.
Key inflammatory cells: eosinophils (virtually all subtypes) — eosinophil products such as major basic protein cause airway damage.
Airway remodeling: if untreated, subbasement membrane thickening + bronchial gland/smooth muscle hypertrophy can add an irreversible component.
— Robbins & Kumar Basic Pathology, Lippincott Pharmacology
COPD
COPD encompasses two often-coexisting entities:
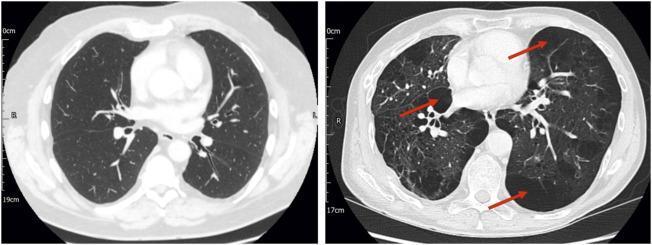
Emphysema
- Enlarged air spaces distal to terminal bronchioles from destruction of elastic support by proteases (especially from neutrophils)
- Loss of elastic recoil → expiratory collapse, air trapping, static and dynamic hyperinflation
- Subtypes: centriacinar (most common; smoking-related) vs. panacinar (α₁-antitrypsin deficiency)
- Clinical hallmarks: barrel chest, dyspnea, relatively preserved oxygenation at rest ("pink puffer")
Chronic Bronchitis
- Defined clinically: productive cough ≥3 consecutive months/year for ≥2 consecutive years
- Pathology: hyperplasia of mucus-secreting glands, goblet cell metaplasia, small airway inflammation (chronic bronchiolitis), bronchiolar wall fibrosis
- Mucus concentrations of MUC5AC are increased 10-fold in severe COPD
- Tends to develop hypoxemia and hypercapnia ("blue bloater")
Inflammatory profile comparison:
| Asthma | COPD | |
|---|---|---|
| Dominant cells | Eosinophils, mast cells, Th2 lymphocytes | Neutrophils, macrophages, CD8+ T cells |
| Cytokines | IL-4, IL-5, IL-13 | IL-8, TNF-α, IL-1β |
| Response to ICS | Excellent | Moderate (especially if eosinophilic) |
— Katzung's Basic and Clinical Pharmacology, 16th Ed.
3. Spirometry
Both conditions cause obstructive pattern on spirometry:
- ↓ FEV₁
- Normal or near-normal FVC
- ↓ FEV₁/FVC ratio (<0.70 post-bronchodilator = diagnostic criterion for COPD per GOLD)
Key distinction: In asthma, obstruction is reversible (≥12% and ≥200 mL improvement in FEV₁ after bronchodilator). In COPD, obstruction is not fully reversible.
4. Treatment
Asthma (GINA Guidelines)
| Symptom Frequency | Preferred Controller | Reliever |
|---|---|---|
| <2×/month | Low-dose ICS-formoterol as needed | — |
| >2×/month, <daily | Low-dose ICS-formoterol as needed | — |
| Most days / weekly nocturnal | Low-dose maintenance ICS-formoterol | Low-dose ICS-formoterol PRN |
| Daily symptoms / low lung function | Medium-dose maintenance ICS-formoterol | Low-dose ICS-formoterol PRN |
Key drug classes:
- Inhaled corticosteroids (ICS) — cornerstone; inhibit phospholipase A2 → ↓ arachidonic acid release → anti-inflammatory. Low oral bioavailability is critical (fluticasone ~1%, mometasone <1%)
- SABA (e.g., albuterol/salbutamol) — acute relief
- LABA (e.g., formoterol, salmeterol) — always combined with ICS in asthma
- Biologic agents (for severe/refractory disease): dupilumab (anti-IL-4Rα), mepolizumab (anti-IL-5), omalizumab (anti-IgE)
COPD (GOLD Guidelines)
- Stop smoking — single most important intervention
- SABA (albuterol) or short-acting anticholinergic (ipratropium) — acute symptom relief
- LAMA (tiotropium, umeclidinium) — preferred long-acting maintenance; reduces exacerbations and dynamic hyperinflation
- LABA (salmeterol, formoterol, indacaterol) — alternative/additive to LAMA
- ICS — reserved for: severe obstruction, frequent exacerbations, high blood eosinophil count (≥300 cells/μL favors ICS response); ICS increases risk of bacterial pneumonia in COPD
- Roflumilast (PDE4 inhibitor) — reduces exacerbation frequency in severe COPD with chronic bronchitis phenotype
- Antibiotics during exacerbations (β-lactams, doxycycline, azithromycin — targeting H. influenzae)
— Katzung's Basic and Clinical Pharmacology, 16th Ed.
5. Asthma-COPD Overlap (ACO)
Not a distinct syndrome but a clinically useful concept when patients exhibit features of both conditions simultaneously.
Key points from Harrison's (2025):
- Smoking can blunt the response to ICS in asthmatics
- Dupilumab has been approved for COPD patients with recurrent exacerbations and elevated eosinophils
- Earlier initiation of anticholinergics may be warranted in patients with both conditions
- Blood eosinophil count is now the preferred biomarker for guiding ICS use in COPD (replacing the older asthma-COPD overlap framework)
— Harrison's Principles of Internal Medicine, 22nd Ed. (2025)
6. Key Distinguishing Features at a Glance
| Feature | Asthma | COPD |
|---|---|---|
| Age of onset | Often young | >40 years |
| Smoking history | Not required | Major risk factor |
| Airflow obstruction | Reversible | Irreversible (or minimally reversible) |
| Main inflammation | Eosinophilic / Th2 | Neutrophilic |
| ICS response | Excellent | Moderate (eosinophil-dependent) |
| Progression | Non-progressive | Progressive |
| FEV₁ post-BD | Normalizes | Does not normalize |
| Exacerbation triggers | Allergens, exercise, URIs | URIs, air pollution, bacteria |
| Cor pulmonale | Rare | Common in advanced disease |
Sources: Robbins & Kumar Basic Pathology; Lippincott Illustrated Reviews: Pharmacology; Katzung's Basic and Clinical Pharmacology 16e; Fishman's Pulmonary Diseases and Disorders; Murray & Nadel's Textbook of Respiratory Medicine; Harrison's Principles of Internal Medicine 22e (2025)
This is a shared conversation. Sign in to Orris to start your own chat.