i send you requirement listr of teacher , this is my ppt topic "Clinical and laboratory diagnosis of fungal diseases affecting the skin and its appendages" i want make ppt on this include image related to data at every slide , and kindly make ppt sccording to requirement list
Asking for Preferences
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Finding Sources
Finding Sources
Searching Images
tinea capitis dermatophyte ringworm scalp clinical
Searching Images
candidiasis skin nails oral thrush clinical photo
Searching Images
KOH mount microscopy fungal hyphae skin scraping laboratory diagnosis
Searching Images
Sabouraud dextrose agar fungal culture dermatophyte colony
Searching Images
onychomycosis nail fungal infection tinea unguium
Searching Images
Wood lamp ultraviolet fluorescence tinea capitis green
Running Command
Great. I’m ready to build it, but one important mismatch first:
You wrote:
- Topic: Clinical and laboratory diagnosis of fungal diseases affecting the skin and its appendages
- Student info: Course: Medical Immunology
Your teacher requirement says the discipline must be:
Microbiology, Virology and Immunology.
I can proceed in either way:
- Keep exactly what you gave: Medical Immunology
- Use requirement-compliant course title: Microbiology, Virology and Immunology
Reply with 1 or 2, and I will generate the final PPTX file with images on every content slide and share the download link.
1
Writing File
Running Command
Running Command
Running Command
Running Command
Running Command
give me content per slide and images i will copy paste by my self
Here is the complete content for every slide, with suggested images you can download and paste in.
📋 PRESENTATION DETAILS (for every slide header/footer)
- Title: Clinical and Laboratory Diagnosis of Fungal Diseases Affecting the Skin and Its Appendages
- Course: Medical Immunology
- Student: Zeel Kathiriya
- Group: 11 | Semester: 4
- Instructor: Duyshenkulova Marina
🖼️ SLIDE 1 — TITLE SLIDE
Title (large, bold):
Clinical and Laboratory Diagnosis of Fungal Diseases Affecting the Skin and Its Appendages
Subtitle block:
Course: Medical Immunology Presented by: Zeel Kathiriya Group: 11 | Semester: 4 Instructor: Duyshenkulova Marina
Image to use:
 (Place on right half of slide, slightly faded/transparent as background)
(Place on right half of slide, slightly faded/transparent as background)

🖼️ SLIDE 2 — TABLE OF CONTENTS
Title: Table of Contents
Content (numbered list):
- Aim & Objectives
- Overview of Fungal Diseases of Skin & Appendages
- Dermatophytosis (Tinea Infections)
- Candidiasis of Skin & Nails
- Clinical Diagnosis Methods
- Laboratory Diagnosis — Direct Microscopy (KOH Mount)
- Laboratory Diagnosis — Culture & Identification
- Molecular & Serological Methods
- Antifungal Treatment Overview
- Conclusion
- References
Image to use:
 (Small image, right side or corner)
(Small image, right side or corner)

🖼️ SLIDE 3 — AIM & OBJECTIVES
Title: Aim & Objectives
AIM:
To understand the clinical features and laboratory methods used for the diagnosis of superficial and cutaneous fungal diseases affecting the skin and its appendages.
OBJECTIVES:
- Classify fungal diseases by affected structure (epidermis, hair, nails)
- Describe clinical presentations of tinea and candidiasis
- Explain direct microscopy (KOH mount) and its interpretation
- Describe fungal culture on Sabouraud Dextrose Agar (SDA)
- Outline molecular and serological diagnostic methods
- Identify Wood lamp and dermoscopy as clinical adjuncts
- Summarise antifungal treatment principles
Image to use:
 (Right side of slide)
(Right side of slide)

🖼️ SLIDE 4 — OVERVIEW: CLASSIFICATION
Title: Overview — Fungal Diseases of Skin & Appendages
Three columns / boxes:
| Superficial Mycoses | Cutaneous Mycoses | Subcutaneous Mycoses |
|---|---|---|
| Affect only outermost keratin layer | Invade stratum corneum, hair & nails | Below epidermis / dermis |
| No significant host immune response | Caused by dermatophytes | Inoculated by trauma |
| e.g. Pityriasis versicolor (Malassezia) | Trichophyton, Microsporum, Epidermophyton | e.g. Sporotrichosis, Mycetoma |
| Tinea nigra, White/Black piedra | Cause tinea infections | Chromoblastomycosis |
Key pathogens table:
| Genus | Source | Common infections |
|---|---|---|
| Trichophyton | Anthropophilic / Zoophilic | Tinea pedis, unguium, capitis, corporis |
| Microsporum | Zoophilic / Geophilic | Tinea capitis, corporis |
| Epidermophyton | Anthropophilic | Tinea cruris, pedis (not hair) |
| Candida | Endogenous | Cutaneous candidiasis, paronychia |
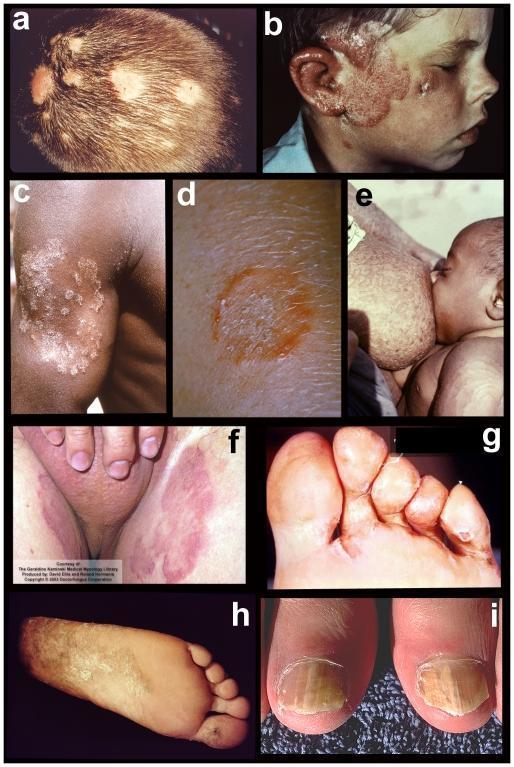
| Malassezia | Commensal | Pityriasis versicolor |
Image to use:
 (Bottom of slide or right side — shows a-i composite of all tinea sites)
(Bottom of slide or right side — shows a-i composite of all tinea sites)

🖼️ SLIDE 5 — DERMATOPHYTOSIS (TINEA INFECTIONS)
Title: Dermatophytosis — Tinea Infections
Causative Agents:
- Trichophyton spp. — most common; T. rubrum, T. tonsurans, T. violaceum
- Microsporum spp. — M. canis (zoophilic), M. gypseum (geophilic)
- Epidermophyton floccosum — affects skin and nails only (not hair)
Clinical Forms:
| Type | Site | Key Features |
|---|---|---|
| Tinea capitis | Scalp | Alopecia, scaling, black-dot sign; children |
| Tinea corporis | Body | Annular plaque, raised edge, central clearing |
| Tinea pedis | Feet | Interdigital maceration, scaling, vesicles |
| Tinea unguium | Nails | Discolouration, subungual hyperkeratosis, dystrophy |
| Tinea cruris | Groin | Pruritic erythematous patches, well-demarcated |
| Tinea faciei | Face | Erythematous scaly plaque, may resemble eczema |
| Tinea manuum | Hands | Diffuse scaling, hyperkeratosis of palm |
Image to use:
 (Right side — same composite showing a–i tinea sites)
(Right side — same composite showing a–i tinea sites)

🖼️ SLIDE 6 — CANDIDIASIS OF SKIN & NAILS
Title: Candidiasis of Skin & Nails
Causative Agent: Candida albicans (most common), C. tropicalis, C. parapsilosis, C. glabrata
Three Forms:
1. Cutaneous Candidiasis
- Skin folds (intertriginous areas): groins, axillae, submammary
- Erythematous macerated patches + satellite pustules at margins
- Risk factors: obesity, diabetes, excessive moisture, broad-spectrum antibiotics
2. Candida Onychomycosis / Paronychia
- Nail fold inflammation (paronychia) → nail plate invasion
- White/yellow discolouration, proximal subungual pattern
- Chronic mucocutaneous candidiasis (CMC) in immunodeficiency
3. Oral Candidiasis (Thrush)
- Thick white pseudomembranous plaques on tongue/buccal mucosa
- Underlying erythematous mucosa
- Seen in infants, immunocompromised, antibiotic users, denture wearers
Images to use:
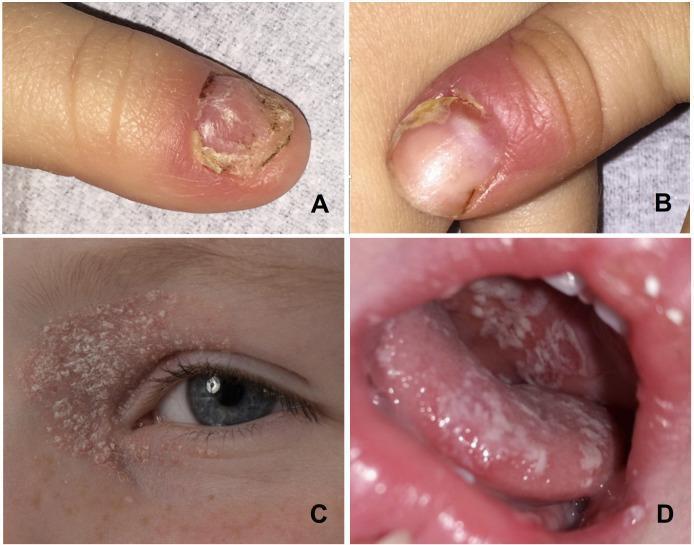
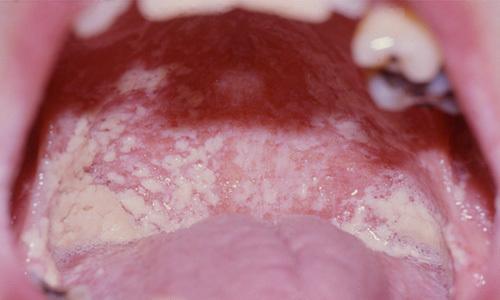
🖼️ SLIDE 7 — CLINICAL DIAGNOSIS METHODS
Title: Clinical Diagnosis Methods
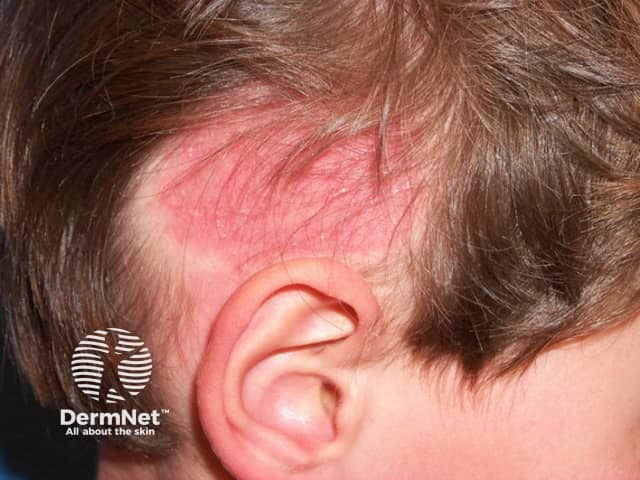
1. Wood Lamp Examination (UV 365 nm)
- Performed in a darkened room
- Microsporum spp. → apple-green / blue-green fluorescence of infected hair shafts
- Trichophyton spp. — usually non-fluorescent
- Quick, non-invasive, bedside screening tool
- Also useful for Pityriasis versicolor (yellow-green), Erythrasma (coral-red)
2. Dermoscopy (Onychoscopy)
- Comma / corkscrew hairs → tinea capitis
- Spiked pattern + longitudinal striae → onychomycosis
- Dermatoscopic "black-dot" pattern in endothrix tinea capitis
3. Clinical Pattern Recognition
- Annular plaque with central clearing → Tinea corporis
- Satellite pustules at lesion edges → Cutaneous candidiasis
- Nail dystrophy + subungual debris → Onychomycosis
- Fluorescence guides targeted sample collection for KOH & culture
Image to use:
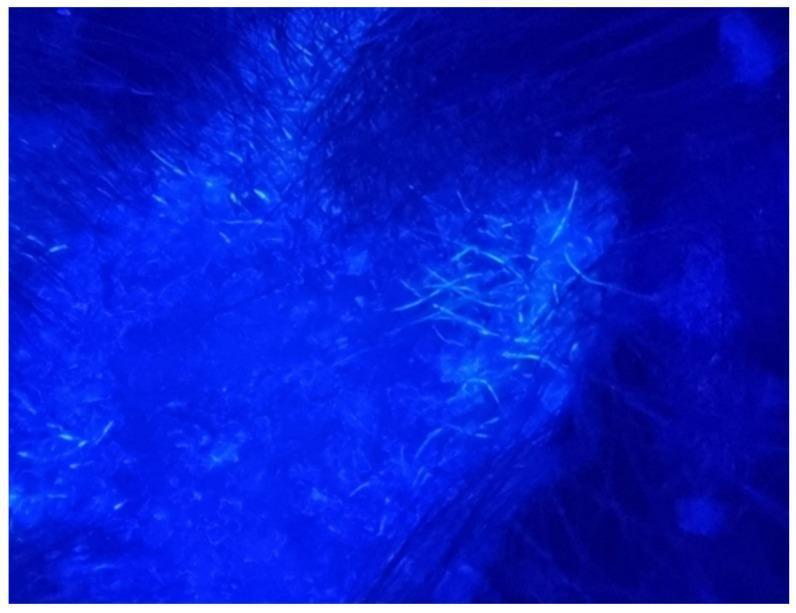 (Wood lamp — blue-green hair shaft fluorescence in Microsporum tinea capitis)
(Wood lamp — blue-green hair shaft fluorescence in Microsporum tinea capitis)

🖼️ SLIDE 8 — LAB DIAGNOSIS: DIRECT MICROSCOPY (KOH MOUNT)
Title: Laboratory Diagnosis — Direct Microscopy (KOH Mount)
Specimen Collection:
- Skin scrapings (from active border of lesion)
- Nail clippings + subungual debris
- Plucked hairs (include root/bulb)
Procedure:
- Place specimen on glass slide
- Add 10–20% KOH solution (± DMSO or Calcofluor white)
- Gently heat or wait 15–30 min to digest keratin
- Apply cover-slip; examine at 10× then 40×
Microscopic Findings:
| Organism | KOH Finding |
|---|---|
| Dermatophytes | Septate, branching hyphae crossing cell walls |
| Candida spp. | Pseudohyphae + budding yeast cells (blastospores) |
| Malassezia | "Spaghetti and meatballs" — short curved hyphae + round spores |
| Tinea capitis | Ectothrix: spores outside hair shaft / Endothrix: spores inside shaft |
Sensitivity: ~70–80% | Specificity: ~80–95%
Images to use:
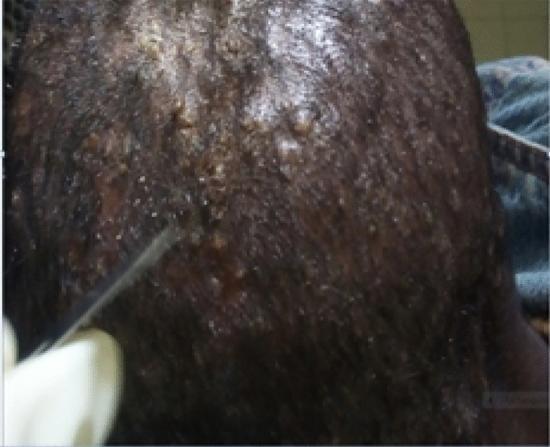

🖼️ SLIDE 9 — LAB DIAGNOSIS: CULTURE & IDENTIFICATION
Title: Laboratory Diagnosis — Culture & Identification
Sabouraud Dextrose Agar (SDA):
- Acidic pH 5.6 — suppresses bacteria; selective for fungi
- Low nutrients — mimics keratin environment
- Incubation: 25–28°C for 2–4 weeks
- Add Cycloheximide + Chloramphenicol to inhibit saprophytes and bacteria
- Dermatophyte Test Medium (DTM): colour change red = dermatophyte
Macroscopic Colony Identification:
| Species | Surface | Reverse Pigment |
|---|---|---|
| T. rubrum | White/powdery | Deep red-wine |
| T. tonsurans | Cream, flat, suede-like | Yellow-brown |
| M. canis | Fluffy, white-yellow | Bright yellow |
| M. gypseum | Cinnamon/tan, granular | Golden-yellow |
| E. floccosum | Khaki, suede-like | Yellowish |
Microscopic Identification (LPCB stain):
- Trichophyton — thin-walled pencil-shaped macroconidia; teardrop microconidia
- Microsporum — large, thick-walled, rough spindle macroconidia (4–15 cells)
- Epidermophyton — club-shaped, smooth, 2–4 cell macroconidia; NO microconidia
- Candida — Germ tube test in serum at 37°C: positive = C. albicans (2 h)
Image to use:
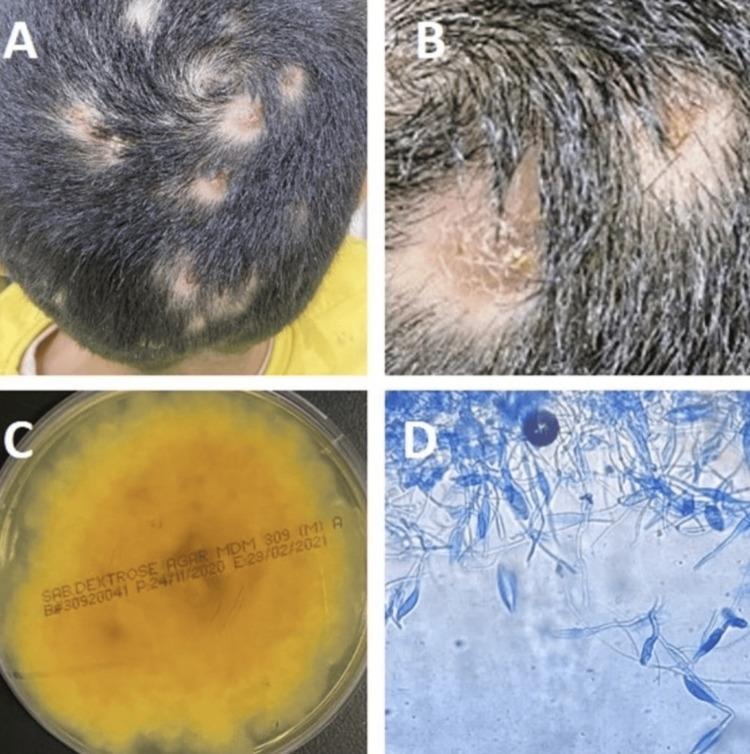 (Composite: scalp tinea capitis + SDA culture plate + LPCB macroconidia of M. gypseum)
(Composite: scalp tinea capitis + SDA culture plate + LPCB macroconidia of M. gypseum)

🖼️ SLIDE 10 — MOLECULAR & SEROLOGICAL METHODS
Title: Molecular & Serological Diagnostic Methods
1. PCR / Molecular Methods
- Pan-dermatophyte PCR directly from nail/hair specimens
- ITS (Internal Transcribed Spacer) region sequencing — gold standard for species ID
- Results in 24–48 hours (vs. 2–4 weeks for culture)
- Detects mixed infections and antifungal-resistant strains (e.g. T. indotineae)
- High sensitivity ~95%
2. MALDI-TOF Mass Spectrometry
- Protein fingerprint identification from cultured colonies
- Rapid (minutes per sample), high accuracy for Candida & dermatophytes
- Cost-effective for high-throughput laboratories
- Replaces traditional biochemical tests
3. Histopathology & Serology
- PAS (Periodic Acid–Schiff) stain on nail biopsy → hyphae stain magenta/red
- GMS (Grocott-Gomori Methenamine Silver) → fungal elements stain black
- Indicated when KOH and culture are negative/inconclusive
- Anti-Candida antibodies (serology) — useful in chronic mucocutaneous candidiasis
Comparison Table:
| Method | Speed | Sensitivity | Species ID | Cost |
|---|---|---|---|---|
| KOH Mount | Minutes | ~75% | No | Very Low |
| Culture (SDA) | 2–4 weeks | ~85% | Yes | Low |
| PCR / ITS | 24–48 h | ~95% | Yes | High |
| MALDI-TOF | Minutes | ~95% | Yes | Medium |
| PAS Biopsy | 1–2 days | ~87% | No | Medium |
Image to use:
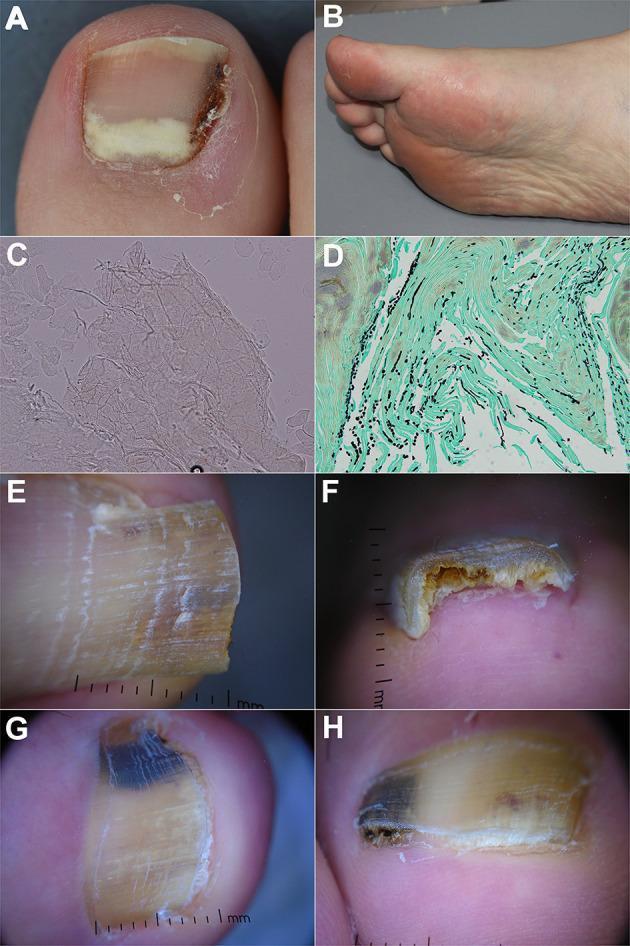 (Multi-panel: clinical nail, KOH hyphae, GMS stain, dermoscopy of onychomycosis)
(Multi-panel: clinical nail, KOH hyphae, GMS stain, dermoscopy of onychomycosis)

🖼️ SLIDE 11 — ANTIFUNGAL TREATMENT OVERVIEW
Title: Antifungal Treatment Overview
Topical Antifungals (mild/localised disease):
- Clotrimazole, Miconazole (azoles) — tinea corporis, cruris, pedis, candidiasis
- Terbinafine cream — tinea pedis, corporis (allylamine)
- Ciclopirox nail lacquer — mild onychomycosis
- Nystatin cream/powder — cutaneous candidiasis
Systemic Antifungals (extensive or nail/scalp disease):
- Terbinafine (oral) — drug of choice for dermatophyte onychomycosis & tinea capitis
- Itraconazole — pulse therapy for onychomycosis; Candida; non-dermatophyte molds
- Fluconazole — candidiasis, tinea capitis (alternative)
- Griseofulvin — first-line tinea capitis in children (older, less used now)
Treatment Duration:
| Site | Duration |
|---|---|
| Tinea corporis / cruris / pedis | 2–6 weeks |
| Tinea capitis | 6–8 weeks (systemic only) |
| Onychomycosis — fingernails | 6 weeks – 3 months |
| Onychomycosis — toenails | 3–6 months or longer |
| Cutaneous candidiasis | 2–4 weeks |
⚠️ Emerging resistance: T. indotineae (terbinafine-resistant) — increasing globally. Always confirm diagnosis before starting systemic therapy.
Image to use:
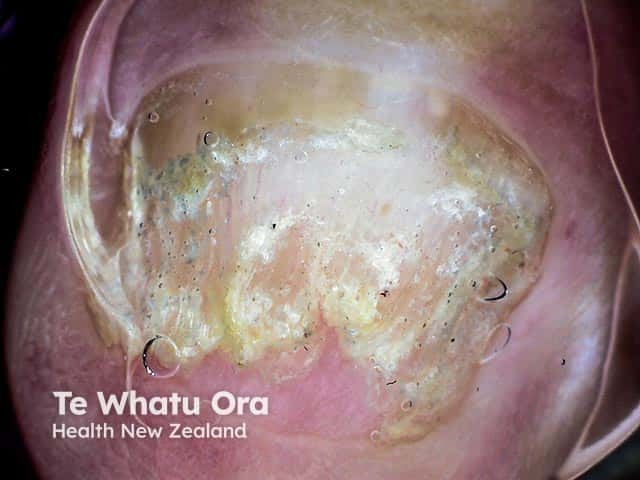 (Severe great toe onychomycosis — illustrates why systemic therapy is needed)
(Severe great toe onychomycosis — illustrates why systemic therapy is needed)

🖼️ SLIDE 12 — CONCLUSION
Title: Conclusion
Key Takeaways (one per bullet — you can make these into cards):
- Fungal diseases of skin and appendages range from superficial to subcutaneous, classified by depth of infection.
- Dermatophytes (Trichophyton, Microsporum, Epidermophyton) are the principal agents of tinea infections affecting skin, hair and nails.
- Candida albicans causes cutaneous candidiasis, paronychia, and oral thrush — especially in immunocompromised patients.
- KOH direct microscopy is the fastest, most accessible first-line laboratory test, demonstrating hyphae or pseudohyphae.
- Culture on SDA remains the gold standard — enables species identification and antifungal susceptibility testing.
- Molecular tools (PCR, ITS sequencing, MALDI-TOF) provide rapid, species-level diagnosis in complex or resistant cases.
- Wood lamp and dermoscopy are valuable non-invasive clinical adjuncts to guide sample collection.
- Treatment ranges from topical azoles/allylamines for limited disease to prolonged systemic antifungals for nail and scalp involvement.
Image to use:
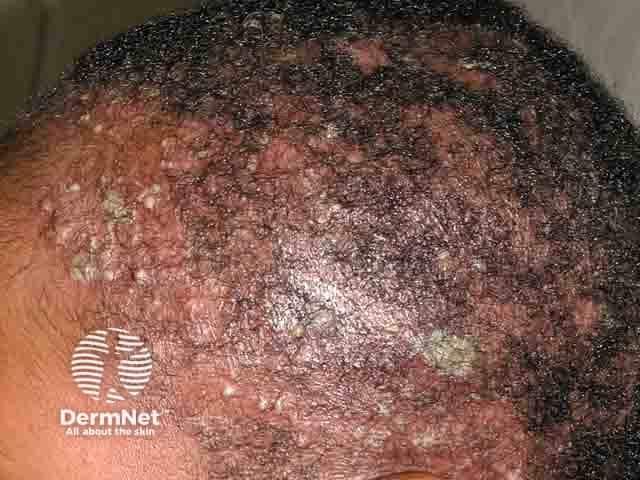 (Tinea capitis with pustules and patchy alopecia — good closing clinical reminder)
(Tinea capitis with pustules and patchy alopecia — good closing clinical reminder)

🖼️ SLIDE 13 — REFERENCES
Title: References
- Murray, P.R., Rosenthal, K.S., Pfaller, M.A. (2021). Medical Microbiology (9th ed.). Elsevier.
- Brooks, G.F., Carroll, K.C., Butel, J.S. et al. (2022). Jawetz, Melnick & Adelberg's Medical Microbiology (28th ed.). McGraw-Hill.
- Havlickova, B., Czaika, V.A., Friedrich, M. (2008). Epidemiological trends in skin mycoses worldwide. Mycoses, 51(Suppl 4): 2–15.
- Aly, R. (1994). Ecology and epidemiology of dermatophyte infections. J Am Acad Dermatol, 31(3): S21–S25.
- Ameen, M. (2010). Epidemiology of superficial fungal infections. Clin Dermatol, 28(2): 197–201.
- Nenoff, P. et al. (2014). Mycology — an update Part 1: Dermatomycoses. J Dtsch Dermatol Ges, 12(3): 188–210.
- Gupta, A.K., Cooper, E.A. (2008). Update in antifungal therapy of dermatophytosis. Mycopathologia, 166(5–6): 353–367.
- Rezusta, A. et al. (2011). MALDI-TOF MS identification of dermatophytes. Med Mycol, 50(5): 541–545.
(No image needed on this slide — keep it clean)
📌 QUICK SUMMARY: Images per slide
| Slide | Image to paste |
|---|---|
| 1 — Title | Composite tinea a–i |
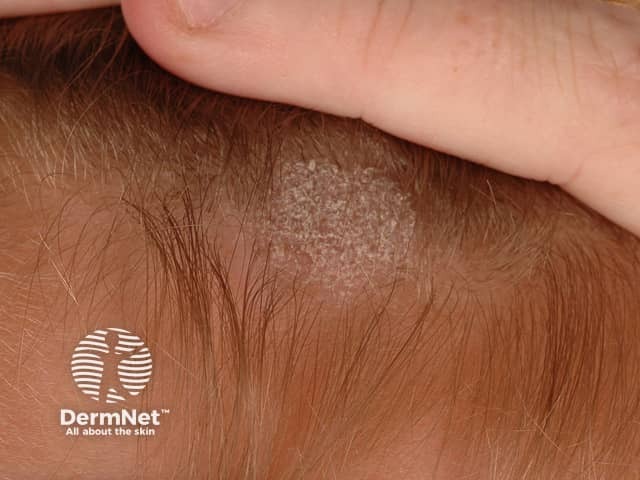
| 2 — TOC | Tinea capitis scalp lesion |
| 3 — Aim | Tinea capitis close-up |
| 4 — Overview | Composite tinea a–i |
| 5 — Tinea | Composite tinea a–i |
| 6 — Candida | CMC nails + oral thrush |
| 7 — Clinical Dx | Wood lamp fluorescence |
| 8 — KOH | Scraping procedure + KOH hyphae |
| 9 — Culture | SDA plate + LPCB microscopy |
| 10 — Molecular | KOH + GMS + dermoscopy panel |
| 11 — Treatment | Severe onychomycosis toe |
| 12 — Conclusion | Tinea capitis with alopecia |
| 13 — References | None |
All image URLs are clickable — right-click → Save image as to download them.
reduce content it is too much per slide if i use 18pt font
Here is the trimmed-down version — every slide fits comfortably at 18 pt font with room for an image.
SLIDE 1 — TITLE SLIDE
Title (large):
Clinical and Laboratory Diagnosis of Fungal Diseases Affecting the Skin and Its Appendages
Below title:
Course: Medical Immunology Presented by: Zeel Kathiriya Group: 11 | Semester: 4 Instructor: Duyshenkulova Marina
Image: Paste composite tinea photo on the right half


SLIDE 2 — TABLE OF CONTENTS
Title: Table of Contents
- Aim & Objectives
- Overview & Classification
- Dermatophytosis — Tinea Infections
- Candidiasis of Skin & Nails
- Clinical Diagnosis Methods
- Laboratory Diagnosis — KOH Mount
- Laboratory Diagnosis — Culture
- Molecular & Serological Methods
- Antifungal Treatment
- Conclusion
- References
Image: Small tinea capitis photo, top-right corner


SLIDE 3 — AIM & OBJECTIVES
Title: Aim & Objectives
Aim:
To understand the clinical features and laboratory methods for diagnosing fungal diseases of the skin and its appendages.
Objectives:
- Classify fungal diseases by affected structure
- Describe clinical presentations of tinea & candidiasis
- Explain KOH microscopy and culture methods
- Outline molecular diagnostic tools
- Summarise antifungal treatment principles
Image: Right side — tinea capitis scalp


SLIDE 4 — OVERVIEW & CLASSIFICATION
Title: Classification of Fungal Skin Diseases
Three boxes (one per type):
Superficial Mycoses
- Affect outermost keratin only
- e.g. Pityriasis versicolor, Tinea nigra
Cutaneous Mycoses
- Invade skin, hair and nails
- Caused by dermatophytes (Trichophyton, Microsporum, Epidermophyton)
Subcutaneous Mycoses
- Enter via trauma
- e.g. Sporotrichosis, Mycetoma
Image: Composite tinea (all body sites) — bottom or right


SLIDE 5 — DERMATOPHYTOSIS
Title: Dermatophytosis — Tinea Infections
Causative Agents:
- Trichophyton spp. — T. rubrum, T. tonsurans (most common)
- Microsporum spp. — M. canis (zoophilic)
- Epidermophyton floccosum — skin & nails only
Clinical Forms (small table):
| Type | Site | Feature |
|---|---|---|
| Tinea capitis | Scalp | Alopecia, scaling |
| Tinea corporis | Body | Annular plaque, central clearing |
| Tinea pedis | Feet | Maceration, itching |
| Tinea unguium | Nails | Dystrophy, discolouration |
| Tinea cruris | Groin | Erythematous patches |
Image: Right side — composite tinea


SLIDE 6 — CANDIDIASIS
Title: Candidiasis of Skin & Nails
Causative Agent: Candida albicans (most common)
Forms:
Cutaneous Candidiasis
- Skin folds — erythema + satellite pustules
- Risk: diabetes, obesity, moisture
Candida Paronychia / Onychomycosis
- Nail fold swelling, nail discolouration
- Chronic mucocutaneous candidiasis (CMC) in immunodeficiency
Oral Candidiasis (Thrush)
- White curd-like plaques on tongue/mucosa
- Common in infants & immunocompromised patients
Images: Two side by side — nails and oral thrush


SLIDE 7 — CLINICAL DIAGNOSIS METHODS
Title: Clinical Diagnosis Methods
Wood Lamp (UV 365 nm)
- Microsporum spp. → apple-green fluorescence of hair shafts
- Trichophyton → usually non-fluorescent
- Quick, non-invasive, bedside screening
Dermoscopy
- Comma/corkscrew hairs → tinea capitis
- Spiked pattern → onychomycosis
Clinical Pattern Recognition
- Annular lesion + central clearing → tinea corporis
- Satellite pustules → candidiasis
- Nail dystrophy + debris → onychomycosis
Image: Wood lamp fluorescence — right side


SLIDE 8 — KOH DIRECT MICROSCOPY
Title: Laboratory Diagnosis — KOH Mount
Specimens: Skin scrapings, nail clippings, plucked hairs
Procedure:
- Add 10–20% KOH to specimen on slide
- Heat gently / wait 15–30 min
- Examine at 10× then 40×
Findings:
| Organism | Finding |
|---|---|
| Dermatophytes | Septate branching hyphae |
| Candida | Pseudohyphae + budding yeast |
| Malassezia | Short hyphae + round spores ("spaghetti & meatballs") |
Sensitivity ~75% | Specificity ~85–95%
Images: Two side by side — scraping procedure + hyphae under microscope


SLIDE 9 — CULTURE & IDENTIFICATION
Title: Laboratory Diagnosis — Culture on SDA
Sabouraud Dextrose Agar (SDA):
- Acidic pH 5.6 — selective for fungi
- Incubate 25–28°C for 2–4 weeks
- Add cycloheximide + chloramphenicol to suppress contaminants
Colony Identification:
| Species | Colony appearance | Reverse pigment |
|---|---|---|
| T. rubrum | White, powdery | Red-wine |
| M. canis | Fluffy, yellow-white | Bright yellow |
| M. gypseum | Tan, granular | Golden-yellow |
Microscopy (LPCB stain):
- Microsporum — large spindle-shaped rough macroconidia
- Trichophyton — thin pencil-shaped macroconidia
- Candida — germ tube test (+) in serum = C. albicans
Image: SDA culture plate + LPCB microscopy


SLIDE 10 — MOLECULAR & SEROLOGICAL METHODS
Title: Molecular & Serological Methods
PCR / ITS Sequencing
- Gold standard for species identification
- Results in 24–48 h vs. 2–4 weeks for culture
- Detects mixed infections & resistant strains
MALDI-TOF Mass Spectrometry
- Rapid protein fingerprint from cultured colonies
- High accuracy for Candida & dermatophytes
Histopathology
- PAS stain → hyphae stain magenta/red
- GMS stain → fungal elements stain black
- Used when KOH and culture are negative
Comparison:
| Method | Speed | Sensitivity |
|---|---|---|
| KOH Mount | Minutes | ~75% |
| Culture | 2–4 weeks | ~85% |
| PCR/ITS | 24–48 h | ~95% |
| MALDI-TOF | Minutes | ~95% |
Image: Multi-panel — KOH, GMS, dermoscopy of onychomycosis


SLIDE 11 — ANTIFUNGAL TREATMENT
Title: Antifungal Treatment Overview
Topical (mild/localised):
- Clotrimazole / Miconazole — tinea corporis, cruris, pedis
- Terbinafine cream — tinea pedis, corporis
- Nystatin — cutaneous candidiasis
- Ciclopirox lacquer — mild onychomycosis
Systemic (nail, scalp, extensive disease):
- Terbinafine oral — onychomycosis, tinea capitis
- Itraconazole — Candida, non-dermatophyte molds
- Fluconazole — candidiasis, tinea capitis
- Griseofulvin — tinea capitis in children
Duration:
- Skin (tinea) — 2–6 weeks
- Tinea capitis — 6–8 weeks
- Onychomycosis — 3–6 months
Image: Severe onychomycosis


SLIDE 12 — CONCLUSION
Title: Conclusion
- Fungal skin diseases range from superficial (keratin only) to subcutaneous (deep tissue).
- Dermatophytes cause tinea infections of skin, hair and nails; Candida causes candidiasis.
- KOH mount — fastest, cheapest first-line test; identifies hyphae/pseudohyphae.
- SDA culture — gold standard; enables species ID and susceptibility testing.
- Molecular methods (PCR, MALDI-TOF) give rapid species-level results.
- Wood lamp and dermoscopy are useful non-invasive clinical adjuncts.
- Treatment ranges from topical antifungals to prolonged systemic therapy depending on site and severity.
Image: Tinea capitis with alopecia and scaling


SLIDE 13 — REFERENCES
Title: References
- Murray, Rosenthal, Pfaller. Medical Microbiology, 9th ed. Elsevier, 2021.
- Brooks et al. Jawetz, Melnick & Adelberg's Medical Microbiology, 28th ed. McGraw-Hill, 2022.
- Havlickova et al. Epidemiological trends in skin mycoses. Mycoses, 2008; 51(Suppl 4): 2–15.
- Aly R. Ecology and epidemiology of dermatophyte infections. J Am Acad Dermatol, 1994; 31(3): S21–S25.
- Ameen M. Epidemiology of superficial fungal infections. Clin Dermatol, 2010; 28(2): 197–201.
- Nenoff et al. Mycology — an update Part 1. J Dtsch Dermatol Ges, 2014; 12(3): 188–210.
- Gupta AK, Cooper EA. Antifungal therapy of dermatophytosis. Mycopathologia, 2008; 166: 353–367.
- Rezusta et al. MALDI-TOF MS for dermatophytes. Med Mycol, 2011; 50(5): 541–545.
(No image needed — keep this slide clean)
💡 Layout tip per slide
Split each slide into left 60% text / right 40% image. With 18 pt body font and 24 pt headings, the above content fits on each slide without overflow.
This is a shared conversation. Sign in to Orris to start your own chat.