Gluconeogenesis
Reading File
Finding Sources
Searching PubMed
gluconeogenesis regulation metabolism
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
Here is a comprehensive overview of gluconeogenesis.
Gluconeogenesis
Overview
Gluconeogenesis is the de novo synthesis of glucose from non-carbohydrate precursors. It is not simply a reversal of glycolysis — the overall equilibrium of glycolysis strongly favors pyruvate formation, so gluconeogenesis requires four unique enzymatic steps to bypass the three irreversible reactions of glycolysis.
Primary site: Liver (~90% after an overnight fast). During prolonged starvation (≥48 hours), the kidneys contribute ~40% of total glucose production. The small intestine can also participate.
Physiologic role: Maintains blood glucose for obligate glucose consumers — brain, erythrocytes (no mitochondria), kidney medulla, lens and cornea of the eye, testes, and exercising muscle. Hepatic glycogen stores are depleted within <24 hours of fasting, making gluconeogenesis essential beyond that point.
Substrates (Gluconeogenic Precursors)
| Precursor | Source | Entry point |
|---|---|---|
| Lactate | Exercising muscle, RBCs | → Pyruvate → OAA → PEP (Cori cycle) |
| Glycerol | TAG hydrolysis in adipose | → DHAP (glycolytic intermediate) |
| Glucogenic amino acids | Muscle protein breakdown | → Pyruvate, OAA, α-ketoglutarate, etc. |
Key rule: All amino acids except leucine and lysine are glucogenic. Acetyl CoA cannot produce net glucose synthesis because the pyruvate dehydrogenase reaction is irreversible — hence ketogenic AAs and fatty acid carbons cannot contribute.
The Cori cycle is the archetypal interorgan relationship: lactate produced by anaerobic glycolysis in muscle/RBCs is carried to the liver, converted back to glucose, and returned to the circulation.
The Four Unique Reactions (Bypassing Glycolytic Irreversibility)
Seven of the 10 glycolytic reactions are reversible and shared with gluconeogenesis. The three irreversible glycolytic steps (pyruvate kinase, PFK-1, glucokinase/hexokinase) are bypassed by four gluconeogenic enzymes:
1. Pyruvate → Oxaloacetate (OAA)
Enzyme: Pyruvate carboxylase (PC)
- Location: Mitochondrial matrix
- Cofactor: Biotin (covalently bound to a lysine residue)
- Reaction: Pyruvate + HCO₃⁻ + ATP → OAA + ADP + Pᵢ
- Allosterically activated by acetyl CoA — this is a critical regulatory signal
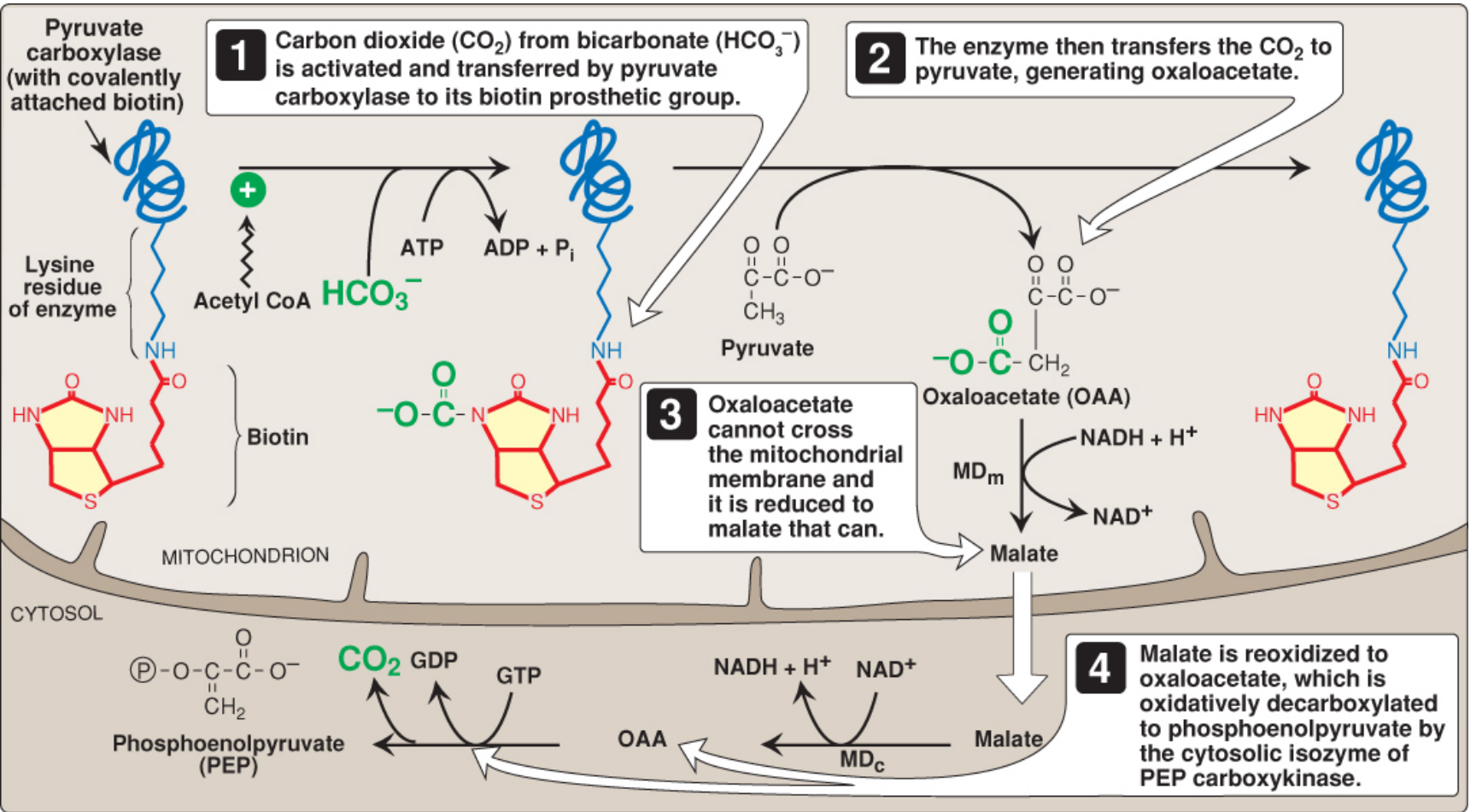
2. OAA → Phosphoenolpyruvate (PEP)
Enzyme: PEPCK (PEP carboxykinase)
- Location: Cytosol (primarily)
- Problem: OAA cannot cross the inner mitochondrial membrane directly
- Solution: OAA is reduced to malate by mitochondrial malate dehydrogenase (using NADH); malate exits to the cytosol; cytosolic malate dehydrogenase re-oxidizes it to OAA (generating cytosolic NADH needed for gluconeogenesis); PEPCK then decarboxylates and phosphorylates OAA using GTP → PEP + CO₂ + GDP
- This shuttle also transfers reducing equivalents (NADH) from mitochondria to cytosol
3. Fructose 1,6-bisphosphate → Fructose 6-phosphate
Enzyme: Fructose 1,6-bisphosphatase (FBPase-1)
- Simple hydrolysis: releases inorganic phosphate (no ATP generated, unlike glycolysis)
- Inhibited by AMP and fructose 2,6-bisphosphate
- Activated when fructose 2,6-bisphosphate levels fall (during fasting/glucagon signaling)
4. Glucose 6-phosphate → Glucose
Enzyme: Glucose 6-phosphatase
- Location: ER membrane (liver and kidney only — explains why these are the sole gluconeogenic organs releasing free glucose)
- Hydrolysis; free glucose exits the hepatocyte into the bloodstream
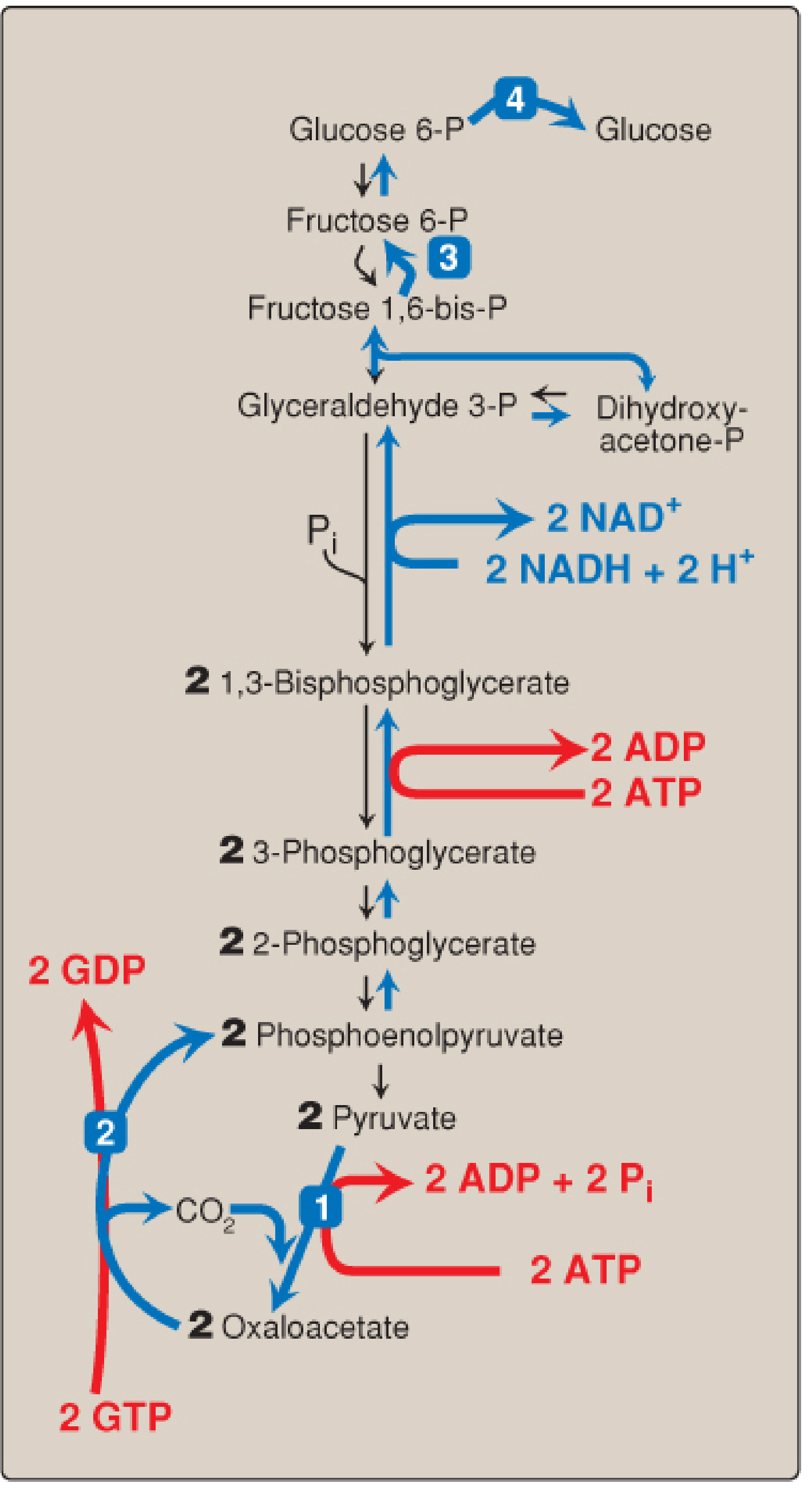
Energy Cost
Synthesizing one glucose from two pyruvate requires:
- 4 ATP (2 for PC, 2 for phosphoglycerate kinase reversal)
- 2 GTP (for PEPCK)
- 2 NADH
Compare to glycolysis, which yields a net of 2 ATP per glucose — gluconeogenesis is energetically expensive and is fueled primarily by fatty acid β-oxidation (which provides ATP, NADH, and acetyl CoA).
Regulation
Gluconeogenesis and glycolysis are reciprocally regulated — when one is active, the other is suppressed.
Hormonal Control
| Hormone | Effect | Mechanism |
|---|---|---|
| Glucagon (fasting) | ↑ Gluconeogenesis | ↓ Fructose 2,6-bisphosphate → FBPase-1 activated, PFK-1 inhibited; phosphorylates/inactivates hepatic pyruvate kinase; induces PEPCK transcription via cAMP–CREB |
| Cortisol (stress/fasting) | ↑ Gluconeogenesis | Mobilizes amino acids from muscle; upregulates PEPCK gene |
| Insulin (fed state) | ↓ Gluconeogenesis | Suppresses PEPCK transcription; raises fructose 2,6-bisphosphate |
| ACTH (low carbohydrate) | ↑ Gluconeogenesis | Stimulates adrenal cortisol release |
Allosteric Control
| Effector | Target | Effect |
|---|---|---|
| Acetyl CoA (↑ with FA oxidation) | Pyruvate carboxylase | Activates → diverts pyruvate to OAA |
| Acetyl CoA | Pyruvate dehydrogenase kinase | Activates PDH kinase → inhibits PDH → less acetyl CoA from pyruvate |
| AMP | FBPase-1 | Inhibits gluconeogenesis (energy low = run glycolysis instead) |
| Fructose 2,6-bisphosphate | FBPase-1 / PFK-1 | Inhibits FBPase-1; activates PFK-1 (fed state) |
Substrate Availability
Increased amino acid flux from muscle (driven by low insulin, high cortisol) provides carbon skeletons. Fatty acid oxidation provides the ATP/NADH coenzymes needed to run the pathway.
Clinical Correlations
| Condition | Gluconeogenesis Link |
|---|---|
| Fasting hypoglycemia | Enzyme deficiencies (PC, PEPCK, FBPase-1, glucose 6-phosphatase) → failure to maintain glucose |
| Type 2 diabetes | Hepatic gluconeogenesis is constitutively elevated despite high insulin (hepatic insulin resistance → PEPCK/G6Pase not suppressed) |
| Metformin | Inhibits complex I of the mitochondrial ETC → raises AMP/ATP ratio → activates AMPK → suppresses gluconeogenesis (primarily by ↓ PEPCK and G6Pase expression) |
| Alcohol (ethanol) | Ethanol oxidation floods liver with NADH; excess NADH traps OAA as malate and pyruvate as lactate → gluconeogenic substrates depleted → fasting hypoglycemia |
| DKA | Glucagon excess and insulin deficiency → maximal gluconeogenesis + glycogenolysis → severe hyperglycemia |
| Biotin deficiency | Impairs PC → reduces gluconeogenesis from pyruvate |
Sources: Biochemistry, 8th ed. Lippincott Illustrated Reviews, pp. 353–368; Basic Medical Biochemistry: A Clinical Approach, 6e, pp. 1007–1015; Guyton and Hall Textbook of Medical Physiology, p. 840.
This is a shared conversation. Sign in to Orris to start your own chat.