Explain carbohydrates
Reading File
Finding Sources
Searching the Web
carbohydrates biochemistry classification structure function
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
I now have comprehensive information from multiple medical textbooks. Here is a thorough explanation of carbohydrates.
Carbohydrates
Overview
Carbohydrates are the most abundant organic molecules in nature. Their empiric formula for simple forms is (CH₂O)n (where n ≥ 3) — literally a "hydrate of carbon." They serve multiple critical roles: they are the primary dietary energy source, a storage form of energy, structural components (e.g., cellulose in plants, chitin in insects), and cell-surface molecules that mediate intercellular communication.
Oxidation of carbohydrates in the body yields approximately 4 kcal/g — the same as protein, but far less than fat (9 kcal/g). — Basic Medical Biochemistry, 6e
Classification
Carbohydrates are classified by size (degree of polymerization):
| Class | Units | Examples |
|---|---|---|
| Monosaccharides | 1 | Glucose, fructose, galactose |
| Disaccharides | 2 | Sucrose, lactose, maltose, trehalose |
| Oligosaccharides | 3–10 | Short sugar chains on glycoproteins |
| Polysaccharides | >10 (up to millions) | Starch, glycogen, cellulose |
— Lippincott Illustrated Reviews: Biochemistry, 8e
Monosaccharides
Monosaccharides are the simplest carbohydrates and the building blocks of all larger structures. They are classified by:
By Number of Carbons
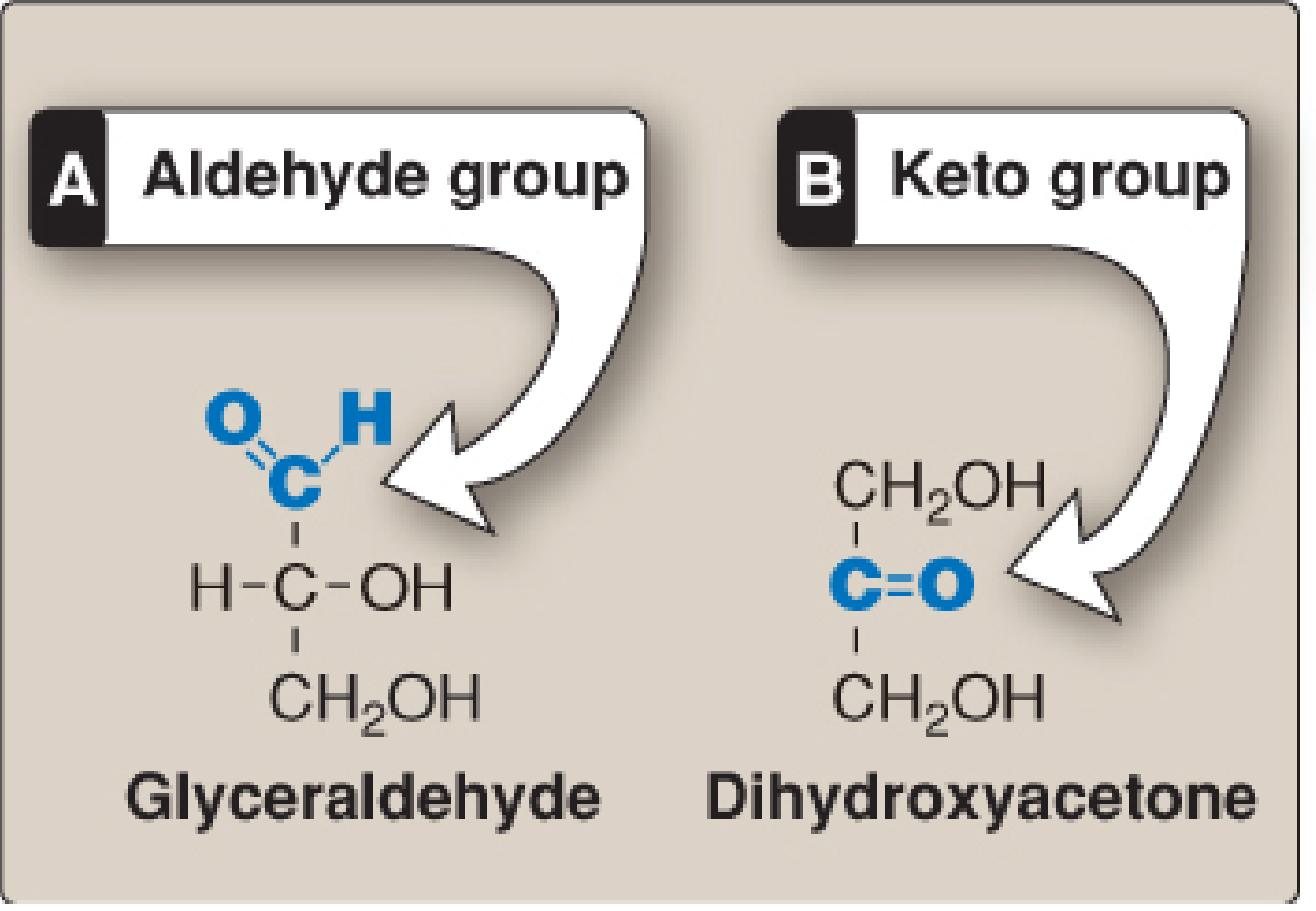
- Trioses (3C): glyceraldehyde, dihydroxyacetone
- Pentoses (5C): ribose (RNA backbone), deoxyribose (DNA backbone)
- Hexoses (6C): glucose, fructose, galactose — the most metabolically important
By Carbonyl Group Type
- Aldoses: contain an aldehyde group (e.g., glyceraldehyde, glucose)
- Ketoses: contain a ketone group (e.g., dihydroxyacetone, fructose)
Isomers and Epimers
Monosaccharides with the same chemical formula but different structures are isomers. Glucose, fructose, mannose, and galactose all share the formula C₆H₁₂O₆. When two sugars differ in configuration around only one carbon, they are epimers:
- Glucose and galactose are C-4 epimers (differ at carbon 4)
- Glucose and mannose are C-2 epimers
Ring Forms and Anomers
In solution, monosaccharides cyclize into ring structures. Glucose forms a pyranose (6-membered ring). The carbon at the ring junction (anomeric carbon) can be in two positions:
- α-glucose: -OH on anomeric carbon points down
- β-glucose: -OH on anomeric carbon points up
These interconvert spontaneously in solution through a process called mutarotation. This distinction is biologically critical — α(1→4) bonds link glucose in starch and glycogen; β(1→4) bonds link glucose in cellulose, which humans cannot digest because we lack β-endoglucosidase.
Disaccharides
Formed when two monosaccharides are linked by a glycosidic bond (a covalent bond between the anomeric carbon of one sugar and a hydroxyl group of another).
| Disaccharide | Monomers | Bond | Source |
|---|---|---|---|
| Sucrose | Glucose + Fructose | α-1,2 | Table sugar, plants |
| Lactose | Galactose + Glucose | β-1,4 | Milk |
| Maltose | Glucose + Glucose | α-1,4 | Starch digestion |
| Trehalose | Glucose + Glucose | α-1,α-1 | Fungi, insects |
— Basic Medical Biochemistry, 6e
Polysaccharides
Polysaccharides are formed by extensive polymerization of monosaccharide units. The major ones in human biology:
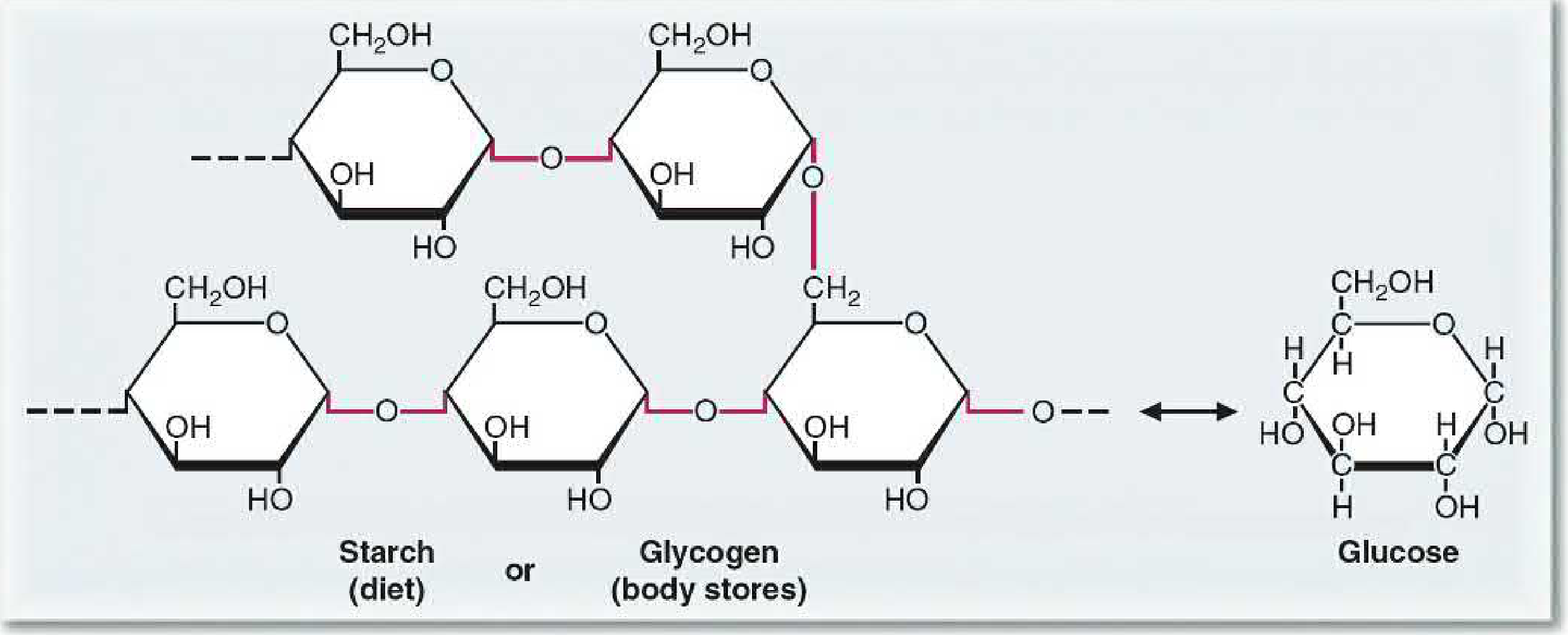
Starch (dietary plant carbohydrate)
- Composed of two forms:
- Amylose: linear chain of glucose via α(1→4) bonds
- Amylopectin: branched, with additional α(1→6) branch points
- The major dietary carbohydrate source in most of the world
Glycogen (animal energy storage)
- The storage form of glucose in humans, concentrated in liver and skeletal muscle
- Structurally similar to amylopectin but more highly branched
- Formation: glycogenesis; breakdown: glycogenolysis
Cellulose (structural, plant)
- Linear glucose polymer with β(1→4) bonds — humans cannot digest this (dietary fiber)
- Provides structural rigidity to plant cell walls
Chitin (structural, insect/fungi)
- Polymer of N-acetyl-D-glucosamine via β(1→4) linkages
- Forms the exoskeleton of insects and crustaceans
Digestion and Absorption
Since only monosaccharides can be absorbed by intestinal epithelial cells, all dietary carbohydrates must first be hydrolyzed. — Costanzo Physiology, 7e
Step-by-step digestion:
-
Mouth: Salivary α-amylase begins hydrolyzing random α(1→4) bonds in starch → produces dextrins, maltose, maltotriose. (Plays limited overall role since it is inactivated by gastric acid.)
-
Stomach: Amylase activity halts due to low pH.
-
Small intestine:
- Pancreatic α-amylase resumes starch digestion → α-limit dextrins, maltose, maltotriose
- Brush-border enzymes of the intestinal epithelium complete digestion:
- α-dextrinase → glucose
- Maltase → glucose
- Sucrase → glucose + fructose
- Lactase → glucose + galactose
- Trehalase → 2 glucose
The three final products of carbohydrate digestion are: glucose, galactose, and fructose.
Absorption mechanism:
- Glucose and galactose: absorbed via secondary active transport on the Na⁺-glucose cotransporter (SGLT1) on the apical membrane (driven by the Na⁺ gradient maintained by Na⁺-K⁺ ATPase); exit via GLUT2 on the basolateral membrane
- Fructose: absorbed by facilitated diffusion on both membranes (no energy-requiring step)
— Costanzo Physiology, 7e
Metabolism
After absorption, glucose is the primary circulating sugar. Normal fasting plasma glucose is 70–110 mg/dL (3.9–6.1 mmol/L). Upon entering cells, glucose is phosphorylated to glucose-6-phosphate by hexokinase (or glucokinase in the liver, which is induced by insulin).
Key metabolic pathways:
| Pathway | Direction | Product |
|---|---|---|
| Glycolysis (Embden–Meyerhof) | Glucose → pyruvate/lactate | ATP (anaerobic or aerobic) |
| Hexose monophosphate shunt | Glucose-6-P → pentoses | NADPH, ribose-5-phosphate |
| Glycogenesis | Glucose → glycogen | Energy storage |
| Glycogenolysis | Glycogen → glucose | Energy release |
| Gluconeogenesis | Non-glucose substrates → glucose | Maintains blood glucose |
Pyruvate is converted to acetyl-CoA, which enters the citric acid cycle (Krebs cycle) to generate ATP. Importantly, the conversion of pyruvate → acetyl-CoA is irreversible, meaning fats cannot be converted to carbohydrates via this route (except from glycerol). — Ganong's Review of Medical Physiology, 26e
Other Biological Roles
Beyond energy metabolism, carbohydrates serve structural and signaling functions:
- Glycoproteins: sugars attached to proteins via N-glycosidic bonds (to -NH₂) or O-glycosidic bonds (to -OH) — aid in cellular targeting, receptor signaling, and immune recognition
- Glycolipids: sugar moieties on lipid structures in cell membranes
- Nucleic acids: ribose (RNA) and deoxyribose (DNA) are pentose sugars forming the backbone of genetic material
- Inositol 1,4,5-trisphosphate (IP₃): a hexose-derived molecule that acts as an intracellular second messenger
Summary
Carbohydrates span a huge range of complexity — from simple glucose powering every cell to complex glycoproteins governing cell-cell recognition. Their central role in energy metabolism, combined with their structural and signaling functions, makes them indispensable to virtually all life processes.
This is a shared conversation. Sign in to Orris to start your own chat.