Explain in detail biosynthesis and catabolism of catecholamine
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Loading Image
Loading Image
Loading Image
Reading File
Finding Sources
Loading Image
Reading File
Finding Sources
I now have comprehensive information from all sources. Let me compile the detailed answer.
Biosynthesis and Catabolism of Catecholamines
Catecholamines — dopamine, norepinephrine (NE), and epinephrine (Epi) — are biogenic amines sharing a catechol nucleus (a benzene ring with two adjacent hydroxyl groups) and a side-chain amine. They serve as both neurotransmitters and hormones.
Part I: Biosynthesis
Precursor and Sites of Synthesis
All three catecholamines are synthesized from the amino acid L-tyrosine, which is either obtained from the diet or synthesized in the liver from the essential amino acid phenylalanine via phenylalanine hydroxylase. The key biosynthetic sites are:
- Adrenal medulla chromaffin cells — produce predominantly epinephrine (~80%) with some norepinephrine
- Sympathetic postganglionic nerve terminals — produce norepinephrine in situ (~80% of total body NE)
- Central dopaminergic neurons (substantia nigra, VTA) — terminate synthesis at dopamine
- Adrenergic neurons of the CNS — synthesize epinephrine
Catecholamines cannot cross the blood–brain barrier, so they must be synthesized locally in each compartment. — Harper's Illustrated Biochemistry, 32nd Ed.
Step-by-Step Biosynthetic Pathway
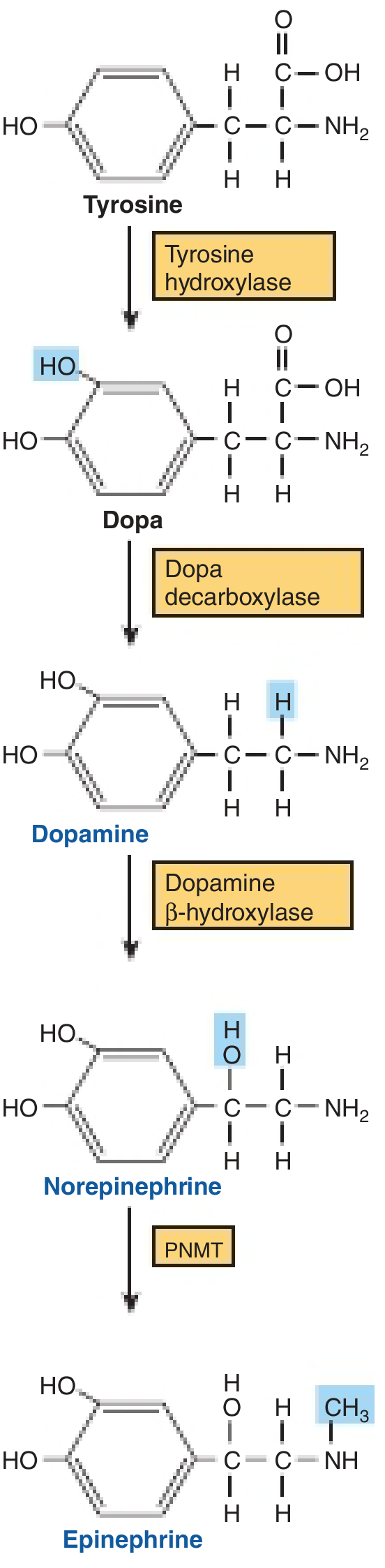
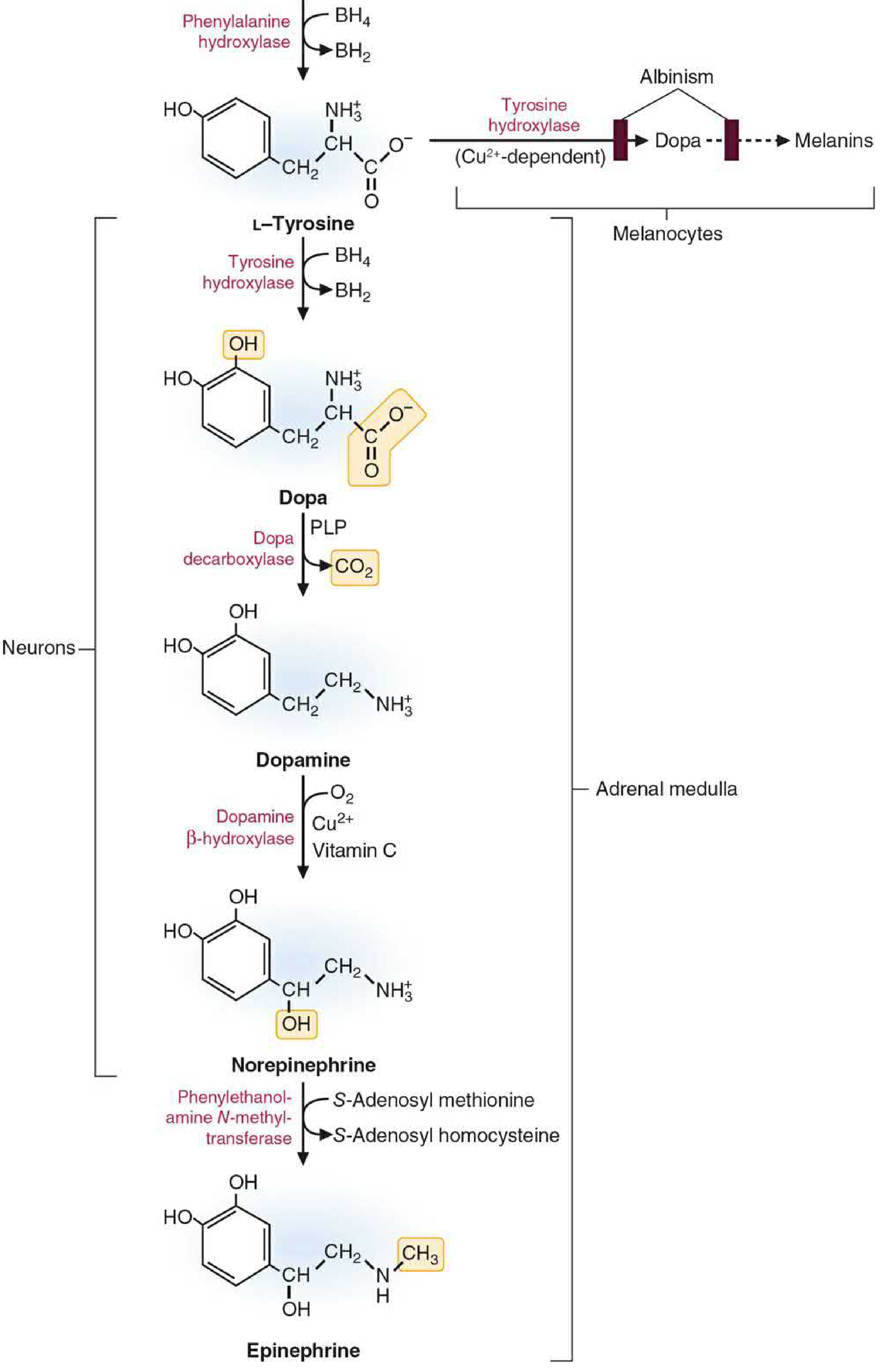
Step 1: L-Tyrosine → L-DOPA (Rate-limiting step)
| Parameter | Detail |
|---|---|
| Enzyme | Tyrosine hydroxylase (TH), encoded by TH gene |
| Reaction | Ring hydroxylation at the 3-position of tyrosine |
| Cofactor | Tetrahydrobiopterin (BH₄) as electron donor; molecular O₂ |
| Location | Cytosol of catecholamine-producing cells |
| Significance | Rate-limiting step for the entire pathway |
Tyrosine hydroxylase is found only in catecholamine-synthesizing tissues. It functions as a mixed-function oxidase (oxidoreductase). The BH₄ cofactor is oxidized to dihydrobiopterin (BH₂) and must be regenerated by dihydropteridine reductase.
Regulation of tyrosine hydroxylase (short-term and long-term):
- Feedback inhibition: Free cytosolic catecholamines compete with BH₄ for the pteridine-binding site on TH, reducing activity when catecholamine levels are high
- Phosphorylation (activation): Nerve terminal depolarization activates PKA, PKC, and Ca²⁺/calmodulin-dependent kinases (CAM kinases), which phosphorylate TH at multiple serine residues; phosphorylated TH binds BH₄ more tightly, reducing sensitivity to end-product inhibition
- Long-term induction: Sustained sympathetic activity increases TH mRNA transcription via phosphorylation of CREB (cAMP response element-binding protein) → binding to CRE in the TH gene promoter → increased synthesis of TH and DBH, which are transported to nerve terminals — Basic Medical Biochemistry, 6th Ed.
Step 2: L-DOPA → Dopamine
| Parameter | Detail |
|---|---|
| Enzyme | DOPA decarboxylase (also called aromatic L-amino acid decarboxylase, AADC) |
| Reaction | Decarboxylation — removal of the carboxyl group from L-DOPA |
| Cofactor | Pyridoxal phosphate (PLP, vitamin B₆) |
| Location | Cytosol; widely distributed in tissues |
| Product | Dopamine (3,4-dihydroxyphenylethylamine) |
This enzyme has broad substrate specificity for aromatic amino acids. Dopaminergic neurons terminate synthesis here — they lack DBH (dopamine β-hydroxylase).
- Competitive inhibitor: α-methyldopa (used to treat hypertension)
- Supplying exogenous L-DOPA to the brain (as in Parkinson therapy) bypasses the rate-limiting TH step; L-DOPA crosses the blood-brain barrier whereas dopamine does not — Kaplan & Sadock's Comprehensive Textbook of Psychiatry
Step 3: Dopamine → Norepinephrine
| Parameter | Detail |
|---|---|
| Enzyme | Dopamine β-hydroxylase (DBH) |
| Reaction | β-hydroxylation of the dopamine side chain |
| Cofactors | Ascorbic acid (vitamin C) as electron donor; Cu²⁺ at active site; fumarate as modulator; molecular O₂ |
| Location | Inside secretory vesicles (particulate fraction) |
| Cell types | Noradrenergic neurons; adrenal chromaffin cells |
Because DBH is exclusively vesicular, dopamine must be transported into storage vesicles from the cytosol before β-hydroxylation occurs. This is mediated by vesicular monoamine transporters (VMATs) powered by an ATP-dependent H⁺ electrochemical gradient across the vesicle membrane. — Basic Medical Biochemistry, 6th Ed.; Tietz Textbook of Laboratory Medicine, 7th Ed.
Step 4: Norepinephrine → Epinephrine (Adrenal medulla only)
| Parameter | Detail |
|---|---|
| Enzyme | Phenylethanolamine N-methyltransferase (PNMT) |
| Reaction | N-methylation of the amine group |
| Methyl donor | S-adenosyl methionine (SAM) → S-adenosyl homocysteine (SAH) |
| Location | Cytosol of adrenal medullary chromaffin cells |
| Dependency | Requires vitamin B₁₂ and folate (for SAM synthesis); induced by glucocorticoids |
Since PNMT is cytosolic, norepinephrine must first leak out of storage vesicles into the cytoplasm for methylation, then the epinephrine formed is transported back into chromaffin granules for storage.
Critical point: PNMT induction requires high intra-adrenal glucocorticoid concentrations delivered via the intra-adrenal portal system (a concentration gradient ~100× greater than systemic arterial blood). This is why adrenal cortical function is essential for epinephrine synthesis. — Harper's Illustrated Biochemistry, 32nd Ed.
Storage and Release
Catecholamines in the cytosol are maintained at low concentrations; the bulk is sequestered in chromaffin granules/secretory vesicles where they are co-stored with ATP, chromogranins, and neuropeptides. Vesicular monoamine transporters (VMAT1 in adrenal; VMAT2 in neurons) drive active uptake. Disruption of the H⁺ gradient (ischemia, anoxia, cyanide poisoning, reserpine) causes massive efflux from vesicles into the cytoplasm, where MAO degrades them.
Exocytotic release is triggered by membrane depolarization and Ca²⁺ influx, mediated by docking protein complexes (SNAREs) at the cell surface. — Tietz Textbook of Laboratory Medicine, 7th Ed.
Part II: Catabolism (Inactivation and Degradation)
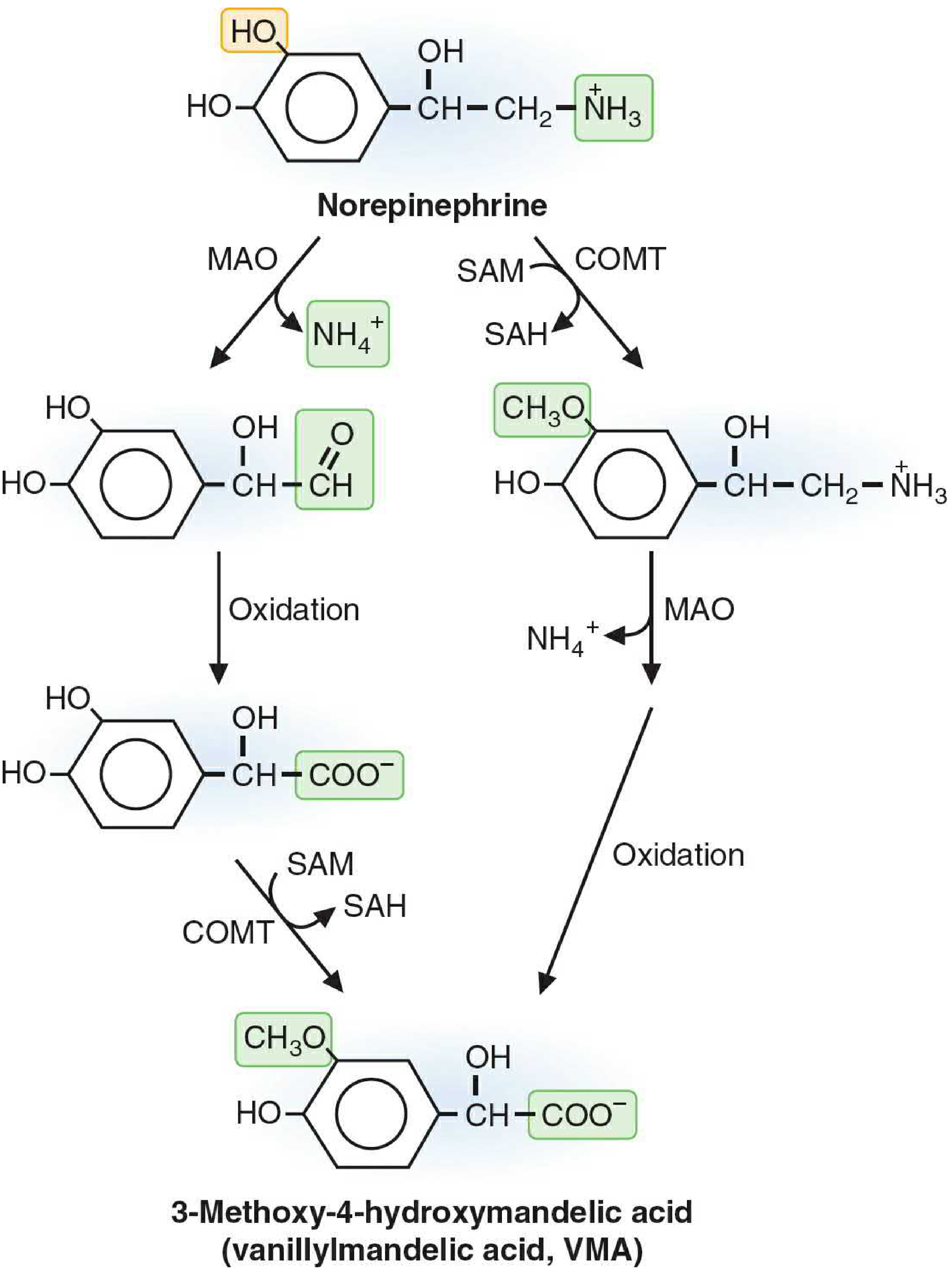
Mechanisms of Termination of Action
Catecholamine signaling is terminated by three mechanisms:
- Reuptake into the presynaptic terminal (primary, most efficient) — via norepinephrine transporter (NET) or dopamine transporter (DAT)
- Diffusion away from the synapse
- Enzymatic degradation — by MAO and/or COMT
Two Key Degradative Enzymes
Monoamine Oxidase (MAO)
| Parameter | Detail |
|---|---|
| Location | Outer mitochondrial membrane of many cell types |
| Reaction | Oxidative deamination: converts the amine group-bearing carbon to an aldehyde, releasing NH₄⁺ |
| Isoforms | MAO-A: preferentially deaminates NE, epinephrine, serotonin; MAO-B: broad substrate specificity for phenylethylamines |
| Function | Inactivates cytosolic catecholamines not protected in vesicles; prevents accumulation of dietary biogenic amines (e.g., tyramine) |
| Inhibitors | MAO inhibitors (MAOIs) — antidepressants; risk of tyramine reaction ("cheese effect") when on MAOIs |
MAO in the presynaptic terminal degrades catecholamines that leak from vesicles. MAO in the liver and gut protects against dietary amines such as tyramine (found in aged cheeses), which normally stimulates NE release. If MAO is inhibited pharmacologically, ingested tyramine is not inactivated and can precipitate hypertensive crisis.
Catechol-O-Methyltransferase (COMT)
| Parameter | Detail |
|---|---|
| Location | Cytosol of many cells, including erythrocytes, liver, kidney, gut (extraneuronal) |
| Reaction | Transfers methyl group from SAM to a hydroxyl group on the catechol ring (usually the 3-OH, i.e., meta position) |
| Cofactor | SAM (indirectly dependent on vitamin B₁₂ and folate) |
| Substrates | Acts on catecholamines and their degradation products that diffuse away from synapses; broad substrate specificity |
| Inhibitors | Entacapone, tolcapone (used in Parkinson disease to prolong L-DOPA effect) |
Catabolic Pathways and End Products
MAO and COMT can act in either order, generating multiple intermediates. The key end products differ by catecholamine:
Norepinephrine and Epinephrine Catabolism
The two routes converge on vanillylmandelic acid (VMA) — also called 3-methoxy-4-hydroxymandelic acid:
Route A (MAO first → COMT second):
NE → 3,4-Dihydroxymandelic aldehyde → 3,4-Dihydroxymandelic acid (DHMA) → VMA (via COMT)
Route B (COMT first → MAO second):
NE → Normetanephrine (NMN, via COMT) → 3-Methoxy-4-hydroxyphenylglycoaldehyde → VMA (via MAO + oxidation)
Additional metabolites include:
- 3-Methoxy-4-hydroxyphenylglycol (MHPG) — a major metabolite of NE in the CNS; sulfate-conjugated (MHPG-SO₄) in mesenteric organs
- Metanephrine — O-methylated product of epinephrine (via COMT); used clinically as a pheochromocytoma marker
- Normetanephrine — O-methylated product of NE
Dopamine Catabolism
Dopamine → 3,4-Dihydroxyphenylacetic acid (DOPAC, via MAO) → Homovanillic acid (HVA) (via COMT)
HVA and 3-methoxytyramine are the main metabolic end products of dopamine; elevated HVA in urine/CSF is used in diagnostic workup of neuroblastoma and dopaminergic system disorders.
Urinary Excretion — Reference Values
| Metabolite | Urinary Excretion (approx.) | % of Total |
|---|---|---|
| VMA (vanillylmandelic acid) | ~20 μmol/d (4000 μg/d) | 59% |
| MHPG (free + conjugated) | ~11 μmol/d (2000 μg/d) | 32% |
| Normetanephrine (free + conjugated) | ~0.55 μmol/d | 1.6% |
| Metanephrine (free + conjugated) | ~0.33 μmol/d | 1% |
| Free NE | ~0.18 μmol/d | 0.5% |
| Free epinephrine | ~0.03 μmol/d | 0.1% |
VMA is the predominant urinary end product (~59% of total catecholamine metabolites), produced mainly in the liver. These metabolites and their sulfate conjugates are eliminated by urinary excretion; slow circulatory clearance means plasma concentrations of metabolites are high relative to the parent amines. — Tietz Textbook of Laboratory Medicine, 7th Ed.
Regional Metabolism
The Tietz textbook describes tissue-specific patterns:
- Sympathetic nerves: Most NE is metabolized intraneuronally by MAO → DHPG (dihydroxyphenylglycol)
- Mesenteric organs: Major site of sulfate conjugation (MHPG-SO₄, NMN-SO₄) by SULT1A3
- Liver: Primary site of VMA production
- Adrenal medulla: Continuous basal production of metanephrine and normetanephrine from intracellular catecholamine metabolism (not just during secretory bursts)
Summary Table: Biosynthetic Enzymes
| Step | Substrate | Enzyme | Product | Cofactor | Location |
|---|---|---|---|---|---|
| 1 (rate-limiting) | L-Tyrosine | Tyrosine hydroxylase | L-DOPA | BH₄, O₂ | Cytosol |
| 2 | L-DOPA | AADC (DOPA decarboxylase) | Dopamine | PLP (B₆) | Cytosol |
| 3 | Dopamine | Dopamine β-hydroxylase | Norepinephrine | Ascorbate, Cu²⁺, O₂ | Storage vesicles |
| 4 | Norepinephrine | PNMT | Epinephrine | SAM (→ SAH) | Cytosol (adrenal medulla) |
Summary Table: Catabolic Enzymes
| Enzyme | Location | Reaction | Key Products |
|---|---|---|---|
| MAO-A | Outer mitochondrial membrane | Oxidative deamination | Aldehydes → acids (VMA, DOPAC) |
| MAO-B | Outer mitochondrial membrane | Oxidative deamination | Broad phenylethylamine substrates |
| COMT | Cytosol, extraneuronal | O-methylation (meta-OH) using SAM | Metanephrine, NMN, VMA, HVA |
Clinical Correlations
| Condition | Mechanism | Relevance |
|---|---|---|
| Parkinson disease | Deficient dopamine synthesis in substantia nigra | Treat with L-DOPA (crosses BBB); carbidopa inhibits peripheral AADC |
| Pheochromocytoma | Catecholamine-secreting adrenal medullary tumor | Elevated plasma metanephrines/normetanephrines and urinary VMA/HVA are diagnostic |
| Hypertension | α-methyldopa competitively inhibits DOPA decarboxylase | Reduces catecholamine synthesis |
| MAO inhibitor therapy | Block MAO-A → ↑ NE, serotonin, dopamine | Antidepressant; cheese reaction risk with tyramine |
| Neuroblastoma | Dopamine-secreting tumor | ↑ urinary HVA and VMA |
| Albinism | Defective tyrosinase in melanocytes (distinct from TH in adrenal) | Absent melanin; catecholamine synthesis normal |
Sources:
- Harper's Illustrated Biochemistry, 32nd Ed. — Ch. 41
- Basic Medical Biochemistry: A Clinical Approach, 6th Ed. — Ch. 46
- Tietz Textbook of Laboratory Medicine, 7th Ed. — Ch. 53
- Kaplan & Sadock's Comprehensive Textbook of Psychiatry — Ch. 1.7
- Ganong's Review of Medical Physiology, 26th Ed.
This is a shared conversation. Sign in to Orris to start your own chat.