Copd
Checking Date & Time
Reading File
Asking for Preferences
Finding Sources
Searching PubMed
"chronic obstructive pulmonary disease"[MeSH Terms] AND "management"[MeSH Terms]
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Loading Image
Finding Sources
Searching PubMed
"chronic obstructive pulmonary disease"[MeSH Terms] AND "treatment"[MeSH Terms]
Reading File
Reading File
Searching PubMed
"COPD" AND "GOLD guidelines"
I now have comprehensive content from multiple authoritative textbooks. Let me compile the full review.
Chronic Obstructive Pulmonary Disease (COPD) — Comprehensive Review
1. Definition
COPD is a progressive, largely irreversible airflow limitation caused by an abnormal inflammatory response of the lung to noxious particles or gases — most commonly cigarette smoke. It encompasses two main pathologic entities that frequently coexist:
- Emphysema — destruction of alveolar walls with permanent airspace enlargement distal to the terminal bronchiole
- Chronic bronchitis — clinically defined as productive cough for ≥3 consecutive months in ≥2 consecutive years, in the absence of another identifiable cause
COPD is the third most common cause of death in the United States and accounts for >$40 billion per year in direct and indirect healthcare costs. — Katzung's Basic and Clinical Pharmacology, 16th ed.
2. Risk Factors
| Factor | Notes |
|---|---|
| Cigarette smoking | Primary cause; responsible for ~90% of cases. Only 15–30% of habitual smokers develop overt COPD, but radiographic changes occur even in smokers with normal spirometry |
| α₁-Antitrypsin (AAT) deficiency | Genetic cause — panacinar emphysema, typically lower-lobe, often in non-smokers or young patients |
| Air pollution | Occupational dusts, sulfur dioxide, nitrogen dioxide, ozone, biomass fuels |
| HIV/AIDS | Associated with premature emphysema |
| Recurrent respiratory infections | Do not initiate COPD but sustain and worsen it |
3. Pathophysiology
3a. Emphysema
The central mechanism is the protease-antiprotease imbalance: cigarette smoke recruits inflammatory cells (especially neutrophils and macrophages) that release proteases (elastase, MMP) → destruction of elastic tissue in alveolar walls. Normally α₁-antitrypsin neutralizes these proteases; deficiency of AAT or overwhelming protease burden allows unchecked destruction.
Result: Loss of alveolar elastic recoil → airspace enlargement → loss of radial airway traction → airflow obstruction on expiration → air trapping and dynamic hyperinflation. During exercise, end-expiratory lung volume (EELV) fails to decline normally, causing dynamic hyperinflation, inspiratory reserve loss, and severe dyspnea. — Fishman's Pulmonary Diseases & Disorders
3b. Chronic Bronchitis
Mechanisms include:
- Mucus hypersecretion: submucosal gland hyperplasia + goblet cell metaplasia in small airways, driven by IL-13, histamine, and acquired CFTR dysfunction from smoking
- Airway inflammation: neutrophils, macrophages, lymphocytes → chronic bronchiolitis → small airway fibrosis
- Impaired mucociliary clearance: cigarette smoke damages ciliary structure and function; reduces airway surface liquid via CFTR inhibition/ENaC activation → mucus hyperconcentration → persistent infection, especially Haemophilus influenzae
The extent of small airway mucus occlusion correlates with degree of airflow obstruction and predicts longevity. — Fishman's
4. Types of Emphysema
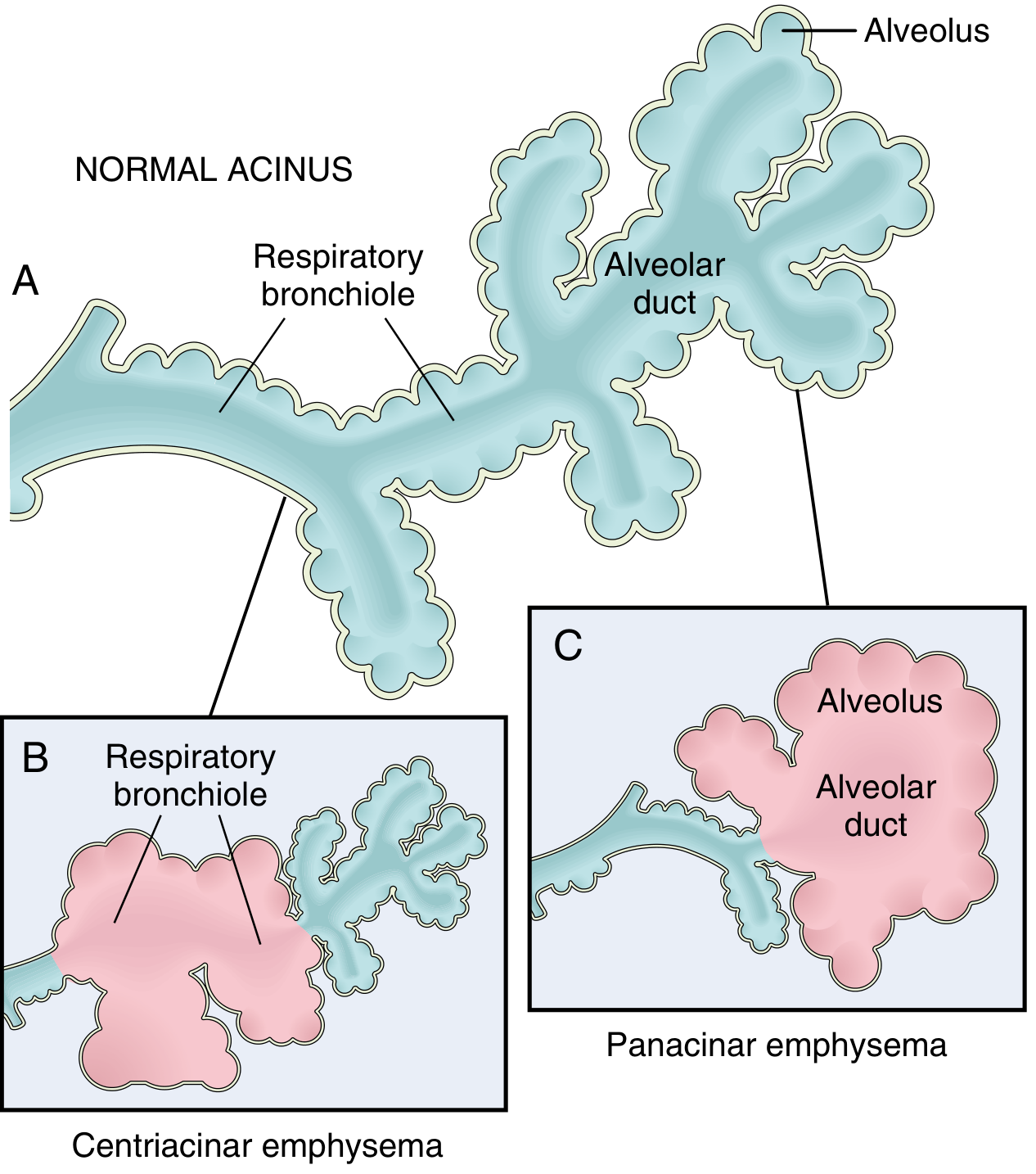
Clinically significant patterns of emphysema — Robbins, Cotran & Kumar Pathologic Basis of Disease
| Type | Distribution | Association | Notes |
|---|---|---|---|
| Centriacinar (centrilobular) | Respiratory bronchioles (central acinus); upper lobes | Cigarette smoking | >95% of clinically significant emphysema |
| Panacinar (panlobular) | Entire acinus uniformly; lower lobes/bases | α₁-Antitrypsin deficiency (± smoking) | Most severe at lung bases |
| Paraseptal (distal acinar) | Distal acinus; subpleural | Spontaneous pneumothorax in young adults | — |
| Irregular | Variable | Scar tissue (healed tuberculosis, etc.) | Usually asymptomatic |
5. Clinical Features
"Pink Puffer" vs "Blue Bloater"
| Feature | Emphysema dominant ("Pink Puffer") | Chronic Bronchitis dominant ("Blue Bloater") |
|---|---|---|
| Age | 50–75 | 40–45 |
| Dyspnea | Severe, early | Mild, late |
| Cough | Late; scanty sputum | Early; copious sputum |
| Cyanosis | Absent (well-oxygenated at rest) | Present |
| Cor pulmonale | Uncommon until end-stage | Common |
| Infections | Occasional | Common |
| Elastic recoil | Markedly reduced | Normal |
| Chest X-ray | Hyperinflation; normal heart | Prominent vessels; large heart |
| Blood gases | Relatively normal at rest | Hypoxemia + hypercapnia |
Most patients fall between these extremes. — Robbins, Cotran & Kumar
6. Diagnosis
Spirometry (Gold Standard)
- FEV₁/FVC < 0.70 post-bronchodilator = confirms obstructive pattern (unlike asthma, not fully reversible)
- Once obstruction is confirmed, FEV₁ % predicted classifies severity (GOLD staging)
- Lung volumes: elevated TLC (hyperinflation), elevated RV (air trapping)
- DLCO: reduced in emphysema; independent predictor of mortality — Fishman's
Physical Examination
Findings of hyperinflation (low diaphragm, hyperresonance, decreased breath sounds) are highly specific but only apparent in advanced disease. A thyroid-to-sternal notch distance < 4 cm in a smoker > 45 years is highly indicative.
Note: Finger clubbing is rare in COPD — if present, consider bronchiectasis, asbestosis, or lung cancer.
Imaging
- Chest X-ray: flat diaphragm, hyperlucency, increased AP diameter
- HRCT: gold standard for emphysema quantification and early detection; identifies small airway disease
When to Test for α₁-Antitrypsin Deficiency
- Emphysema onset < 45 years
- Emphysema in a non-smoker or lower-lobe predominant emphysema
- Family history of early-onset emphysema or cirrhosis
- Bronchiectasis without identifiable cause
7. Classification — GOLD Staging
GOLD Airflow Limitation Severity (post-bronchodilator FEV₁, all require FEV₁/FVC < 0.70)
| GOLD Grade | Severity | FEV₁ % Predicted |
|---|---|---|
| GOLD 1 | Mild | ≥ 80% |
| GOLD 2 | Moderate | 50–79% |
| GOLD 3 | Severe | 30–49% |
| GOLD 4 | Very Severe | < 30% |
GOLD ABCD Assessment Tool
Beyond spirometry, the GOLD framework also categorizes patients by symptom burden (mMRC dyspnea scale or CAT score) and exacerbation history to guide therapy:
| Group | Symptoms | Exacerbation History |
|---|---|---|
| A | Few | 0–1 (not hospitalized) |
| B | Many | 0–1 (not hospitalized) |
| C | Few | ≥2, or ≥1 hospitalization |
| D | Many | ≥2, or ≥1 hospitalization |
Source: Rosen's Emergency Medicine; GOLD 2020 Guidelines
8. Management of Stable COPD
Goals
Prevent progression, relieve symptoms, improve exercise capacity and quality of life, prevent/treat exacerbations, and improve survival. — Fishman's
Non-Pharmacological
| Intervention | Notes |
|---|---|
| Smoking cessation | Single most effective intervention to slow FEV₁ decline; even in advanced COPD |
| Pulmonary rehabilitation | Improves exercise tolerance, dyspnea, and depression; reduces exacerbations |
| Vaccinations | Influenza and pneumococcal (reduce exacerbations and mortality) |
| Long-term O₂ | Indicated when PaO₂ ≤ 55 mmHg (or ≤ 59 mmHg with cor pulmonale/polycythemia); shown to improve survival |
| NIV/CPAP | For overlap syndrome (COPD + OSA); consider nocturnal NIV in chronic hypercapnia |
| Pulmonary rehabilitation | Reduces hospitalizations and depression |
| Surgery | Lung volume reduction surgery (LVRS) for selected emphysema patients; lung transplantation for end-stage disease; bullectomy where indicated |
Pharmacotherapy (Stable COPD)
Short-acting agents (rescue)
- SABA (salbutamol/albuterol): first-line for acute symptom relief
- SAMA (ipratropium): anticholinergic; can be combined with SABA
Long-acting agents (maintenance)
- LABA (salmeterol, formoterol, indacaterol): reduces dyspnea and exacerbations
- LAMA (tiotropium, umeclidinium): long-acting anticholinergic; often preferred as first maintenance agent; can combine with LABA
Inhaled corticosteroids (ICS)
- Less central than in asthma; associated with increased risk of bacterial pneumonia
- Recommended for: severe obstruction (GOLD 3–4), frequent exacerbations, blood eosinophil counts suggesting ICS benefit (eosinophil-guided therapy), or clear coexisting asthma
- Current GOLD-based guidelines use blood eosinophil levels to guide ICS use rather than an asthma-COPD overlap diagnosis
Roflumilast (PDE4 inhibitor)
- Oral, selective phosphodiesterase-4 inhibitor
- Improves pulmonary function and reduces exacerbation frequency
- Approved for COPD; particularly for chronic bronchitis phenotype with frequent exacerbations — Katzung's
Theophylline
- A recent large RCT of low-dose theophylline showed no benefit on exacerbation frequency; no longer recommended as standard therapy
9. COPD Exacerbations
Definition
Increased dyspnea, often with increased cough, sputum production/purulence, wheezing, or chest tightness — in the absence of an alternative explanation. — Washington Manual
Causes
- Viral infections (rhinovirus most common), bacterial infections, and air pollution cause most exacerbations
- Bacteria: H. influenzae, S. pneumoniae, Moraxella catarrhalis (and gram-negative rods including Pseudomonas in patients with risk factors)
- Differential: pneumothorax, pneumonia, pleural effusion, CHF, PE, ACS
Severity Classification
- Mild: worsening symptoms only — home management
- Moderate: requires antibiotics and/or systemic corticosteroids
- Severe: requires ED visit or hospitalization
Indications for Hospital Admission
- Significant increase in symptom severity
- Severe underlying COPD
- Significant comorbidities
- Failure to respond to initial treatment
- Diagnostic uncertainty / insufficient home support
Indications for ICU Admission
- Need for mechanical ventilation
- Hemodynamic instability
- Severe dyspnea not responding to therapy
- Altered mental status
- Persistent/worsening hypoxemia, hypercapnia, or respiratory acidosis despite O₂ and NIV — Washington Manual; Fishman's
Treatment of Exacerbations
Bronchodilators
- SABAs (albuterol 2.5 mg nebulized q1–4h) are first-line
- Add ipratropium if inadequate response
Corticosteroids
- Prednisone 40 mg/day × 5 days (outpatient); 30–60 mg/day × 5–10 days (inpatient)
- Shortens duration of symptoms and reduces treatment failure
Antibiotics (indicated when increased sputum purulence or change in sputum character suggests bacterial infection)
| Patient Risk | Common Pathogens | Antibiotic |
|---|---|---|
| No risk factors | H. influenzae, S. pneumoniae, M. catarrhalis | Macrolide, 2nd/3rd-gen cephalosporin, doxycycline, TMP-SMX |
| Risk factors present (age >65, FEV₁ <50%, >3 exacerb/yr, recent antibiotics, cardiac comorbidity) | Above + gram-negatives incl. Pseudomonas | Antipseudomonal fluoroquinolone or β-lactam |
Treat for 3–7 days. Use CRP to guide antibiotic decisions when available. — Washington Manual; Fishman's
Oxygen
- Controlled supplementation at the lowest flow needed to reverse hypoxemia — to avoid worsening hypercapnia
- Target SpO₂ 88–92% in COPD
Non-invasive ventilation (NIV/BiPAP)
- Reduces need for intubation and improves survival in hypercapnic respiratory failure
- Preferred over invasive ventilation unless contraindicated
10. Complications
| Complication | Notes |
|---|---|
| Pulmonary hypertension / Cor pulmonale | COPD is the most common cause of chronic lung disease–related pulmonary hypertension (>80% of cases); severe PH (mPAP >35–40 mmHg) is uncommon — Murray & Nadel's |
| Respiratory failure | Type I (hypoxemic) or Type II (hypercapnic) |
| Polycythemia | Secondary to chronic hypoxemia |
| Pneumothorax | Rupture of subpleural blebs (especially in emphysema) |
| Lung cancer | Markedly increased risk (shared risk factor: smoking; but COPD is an independent risk) |
| Overlap syndrome (COPD + OSA) | Severe pulmonary hypertension and daytime hypercapnia |
| Depression and anxiety | Common; associated with poorer prognosis and more exacerbations |
11. Prognosis
COPD is progressive; no currently proven treatment reverses disease. The BODE index (BMI, airflow Obstruction, Dyspnea, Exercise capacity) predicts mortality better than FEV₁ alone. Long-standing disease, especially with a bronchitic component, commonly leads to pulmonary hypertension, cor pulmonale, and death from heart failure. Smoking cessation remains the only intervention proven to slow FEV₁ decline.
Sources: Robbins, Cotran & Kumar — Pathologic Basis of Disease | Robbins & Kumar Basic Pathology | Fishman's Pulmonary Diseases and Disorders, 2-Volume Set | Murray & Nadel's Textbook of Respiratory Medicine | Katzung's Basic and Clinical Pharmacology, 16th ed. | The Washington Manual of Medical Therapeutics | Rosen's Emergency Medicine | GOLD 2020/2021 Guidelines
Make a pdf of this
Reading File
Running Command
Writing File
You've run out of free credits. Sign up for a free account to continue using Orris.
This is a shared conversation. Sign in to Orris to start your own chat.