Nitrogen balance and limiting amino acid
Reading File
Finding Sources
Finding Sources
Searching the Web
nitrogen balance limiting amino acid biochemistry nutrition
Reading File
Reading File
Reading File
Finding Sources
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Now I have comprehensive content from multiple authoritative textbooks. Let me compile a thorough answer.
Nitrogen Balance & Limiting Amino Acids
1. Nitrogen Balance — Definition
Nitrogen is the fundamental component of amino acids; ~16% of protein mass is nitrogen, meaning 1 g nitrogen ≈ 6.25 g protein. Nitrogen balance is the difference between nitrogen intake and nitrogen output over 24 hours:
N balance = Protein intake (g) / 6.25 − (UUN + 4 g)
Where UUN = urinary urea nitrogen, and 4 g accounts for non-urea urinary losses plus skin and fecal losses.
Nitrogen is excreted primarily as:
- Urea (main form in urine)
- Creatinine, NH₄⁺, uric acid (minor urinary forms)
- Undigested protein, shed intestinal epithelial cells (fecal)
- Small amounts via sweat, skin, hair
2. States of Nitrogen Balance
| State | Nitrogen Relationship | Physiologic Meaning |
|---|---|---|
| Nitrogen equilibrium | Intake = Output | Normal healthy adult |
| Positive N balance | Intake > Output | Net protein anabolism |
| Negative N balance | Intake < Output | Net protein catabolism |
Positive Nitrogen Balance
- Growing children and adolescents
- Pregnancy
- Recovery from illness, emaciating disease, or trauma
- Reflects net protein synthesis exceeding breakdown
Negative Nitrogen Balance
- Inadequate total dietary protein
- Lack of one or more essential amino acids (EAAs) — even with adequate total protein, if a single EAA is absent, protein synthesis halts and unused amino acids are deaminated and excreted as nitrogen
- Physiologic stress: trauma, burns, surgery, infections, cancer, hyperthyroidism, starvation
- During these catabolic states, cytokines and glucocorticoids drive increased tissue protein breakdown; up to 6–7% of total body protein may be lost over 10 days following major trauma
Note: A high protein intake does NOT produce positive nitrogen balance in healthy adults — it increases both synthesis and catabolism proportionally, maintaining equilibrium at a higher turnover rate (with increased diet-induced thermogenesis).
3. Protein Requirements from N Balance Studies
- Average daily requirement: 0.66 g protein/kg body weight (reference intake ~0.825 g/kg/day allowing for individual variation), roughly 55 g/day
- RDA (US): 0.8 g/kg/day for adults
- Sedentary 70-kg adult: ~56 g/day
- Athletes/regular vigorous exercise: ~1 g/kg/day
- Pregnancy/lactation: +30 g/day above baseline
- Infants: 2 g/kg/day (for growth)
- Kidney disease: may require restriction
- Burns: require increased intake
4. The Requirement Is for Specific Amino Acids, Not Just Total Protein
Essential (Indispensable) Amino Acids — the 9 that cannot be synthesized in humans:
| Histidine | Isoleucine |
| Leucine | Lysine |
| Methionine | Phenylalanine |
| Threonine | Tryptophan |
| Valine |
Conditionally essential (synthesized only from essential precursors):
- Cysteine ← from methionine
- Tyrosine ← from phenylalanine
Truly dispensable (synthesized from common metabolic intermediates): alanine, aspartate, glutamate.
5. Limiting Amino Acid — The "Barrel Stave" Concept
A limiting amino acid is the essential amino acid present in the lowest amount relative to the body's requirement — it limits the rate of protein synthesis regardless of how much total protein is consumed.
The analogy: imagine a barrel where each stave represents one amino acid. The barrel can only be filled to the level of the shortest stave — the limiting amino acid.
- If any one EAA is absent or deficient, protein synthesis stops at that point, and the remaining amino acids are deaminated and their nitrogen excreted → negative nitrogen balance results.
- The first-limiting amino acid is the EAA that becomes deficient first.
Common Limiting Amino Acids by Food Source
| Food | First-Limiting AA | Second-Limiting AA |
|---|---|---|
| Wheat / cereals / grains | Lysine | Threonine |
| Legumes (beans) | Methionine | — |
| Corn (maize) | Lysine + Tryptophan | — |
| Rice | Lysine | Threonine |
| Animal proteins | Generally none (complete) | — |
Lysine is the most commonly first-limiting amino acid in cereal-based diets worldwide.
6. Protein Quality & Scoring Systems
Biological Value (BV)
Measures the proportion of absorbed nitrogen actually retained for use. Higher BV = more complete amino acid profile. Animal proteins (egg, milk, meat, fish) → complete proteins; plant proteins → incomplete.
PDCAAS (Protein Digestibility-Corrected Amino Acid Score)
- Standard adopted by most governments
- Based on EAA profile corrected for protein digestibility
- Maximum score = 1.0 (casein, whey, egg, soy)
DIAAS (Digestible Indispensable Amino Acid Score)
- Proposed by FAO in 2013 to replace PDCAAS
- Uses true ileal digestibility of individual EAAs (more accurate) vs. total tract crude protein digestibility used in PDCAAS
- More precisely reflects actual bioavailability
7. Complementary Proteins — Practical Application of Limiting AA Concept
Two incomplete plant proteins can be combined to compensate for each other's deficiencies:
- Wheat (lysine-deficient, methionine-rich) + Kidney beans (methionine-poor, lysine-rich) → combined BV approaches complete protein

This is the nutritional basis for traditional food pairings (rice + lentils, corn tortillas + beans).
8. Clinical Significance
| Condition | N Balance | Mechanism |
|---|---|---|
| Kwashiorkor | Negative | Protein deficiency with adequate calories |
| Marasmus | Negative | Total calorie + protein deficiency |
| Post-surgery/trauma | Negative | Cytokine/glucocorticoid-driven catabolism |
| Burns | Markedly negative | Threonine/cysteine consumed in acute-phase protein synthesis |
| Prolonged bed rest | Negative | Muscle atrophy, reduced anabolic stimulus |
| Renal failure | Managed by restriction | Urea retention worsened by excess protein |
| Growth/pregnancy | Positive | Net protein accretion required |
1 g nitrogen = 6.25 g protein = ~30 g hydrated lean body mass — so negative nitrogen balance manifests clinically as measurable loss of muscle mass, a diagnostic criterion for malnutrition.
Sources: Lippincott Illustrated Reviews: Biochemistry 8e, p. 1022–1025 | Harper's Illustrated Biochemistry 32e, p. 544–546 | Basic Medical Biochemistry: A Clinical Approach 6e | Yamada's Textbook of Gastroenterology 7e
HCL synthesis and functions of hcl
Reading File
Finding Sources
Finding Sources
Searching the Web
HCl gastric acid synthesis parietal cell mechanism functions physiology
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
I now have comprehensive information from Guyton & Hall, Histology (Wheater's/Ross), and Harrison's. Let me compile the full answer.
Gastric HCl: Synthesis and Functions
1. Site of Production
HCl is produced exclusively by parietal cells (also called oxyntic cells) of the oxyntic (fundic) glands in the body and fundus of the stomach. The parietal cell:
- Has an extensive intracellular canalicular system — deep branching invaginations of the apical membrane
- Is packed with mitochondria (providing ATP for active transport)
- HCl is formed at the villus-like projections inside these canaliculi and conducted outward into the gastric lumen
Concentration: ~160 mM HCl → gastric juice pH as low as 0.8 (millions of times more acidic than blood)
2. Mechanism of HCl Synthesis
The synthesis involves three coordinated ion transport steps:
Step 1 — H⁺ Generation via Carbonic Anhydrase
CO₂ (from blood or cell metabolism) + H₂O → H₂CO₃ → H⁺ + HCO₃⁻
- Catalyzed by carbonic anhydrase in the parietal cell cytoplasm
- CO₂ diffuses across the basolateral membrane from blood capillaries
- This is the source of H⁺ ions
Step 2 — H⁺ Secretion via H⁺/K⁺-ATPase (Proton Pump)
- H⁺/K⁺-ATPase on the apical canalicular membrane actively pumps H⁺ into the canalicular lumen in exchange for K⁺ (entering the cell)
- This is the primary driving force — against a massive concentration gradient
- K⁺ recycled back into the lumen via K⁺ channels, keeping the pump running
Step 3 — Cl⁻ Secretion & HCO₃⁻ Exchange
- Cl⁻ is transported into the canalicular lumen via Cl⁻ channels
- On the basolateral membrane, HCO₃⁻ generated in Step 1 is exchanged for plasma Cl⁻ via a HCO₃⁻/Cl⁻ antiporter → this is the "alkaline tide" (post-meal rise in blood pH)
- Na⁺/K⁺-ATPase on the basolateral membrane maintains low intracellular Na⁺ and drives the overall electrochemical gradient
Final Result
H⁺ + Cl⁻ → HCl in the canalicular lumen → secreted into the gastric gland lumen
Water follows by osmosis into the canaliculus.
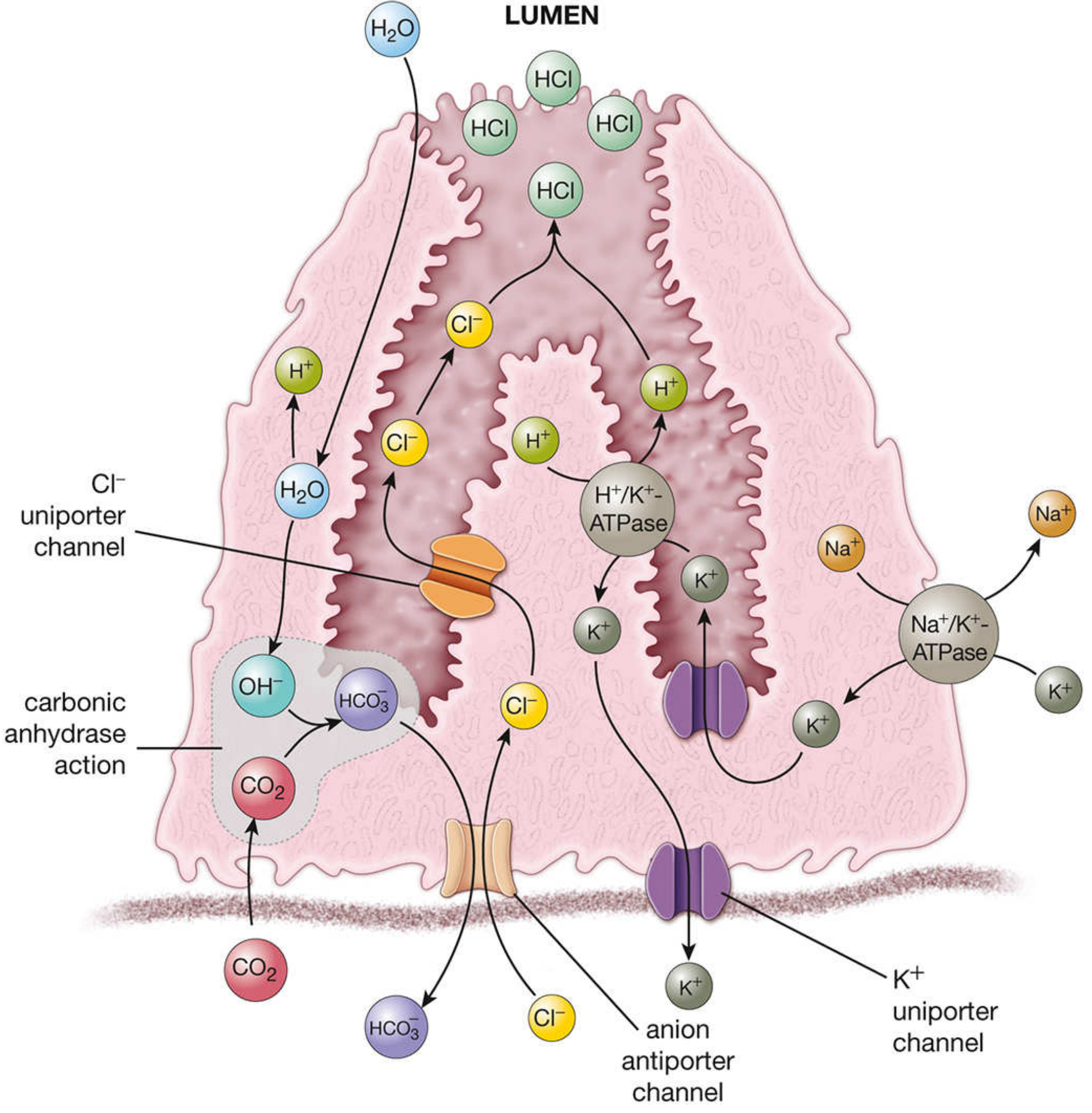
Fig: Parietal cell HCl synthesis — carbonic anhydrase generates H⁺; H⁺/K⁺-ATPase pumps H⁺ into canaliculus; Cl⁻ channels complete HCl formation; basolateral HCO₃⁻/Cl⁻ exchange creates the "alkaline tide."
3. Stimulation of HCl Secretion
Parietal cells have three receptor types that stimulate HCl secretion:
| Receptor | Ligand | Source |
|---|---|---|
| Gastrin receptor (CCK-B) | Gastrin (G-34, G-17) | G cells of antrum |
| H₂ receptor | Histamine | ECL cells of oxyntic glands |
| M₃ muscarinic receptor | Acetylcholine | Vagal/enteric nerves |
The ECL Cell Axis (Primary Pathway)
- G cells (antrum) → secrete gastrin (stimulated by dietary proteins, stomach distension, vagal input)
- Gastrin → stimulates ECL (enterochromaffin-like) cells in the oxyntic glands
- ECL cells → release histamine directly onto parietal cells
- Histamine → activates H₂ receptors → ↑cAMP → stimulates H⁺/K⁺-ATPase
Three Phases of Gastric Secretion
| Phase | Trigger | Mechanism |
|---|---|---|
| Cephalic (~30%) | Sight, smell, taste, thought of food | Vagus nerve → ACh → parietal cells directly + via G cells |
| Gastric (~60%) | Food in stomach, stomach distension, protein | Gastrin release from G cells; local ENS reflexes |
| Intestinal (~10%) | Chyme in duodenum | Intestinal gastrin; absorbed AAs stimulate further secretion |
Inhibition of HCl Secretion
- Somatostatin from D cells (↑ when antral pH falls below 3) → inhibits G cells and parietal cells directly
- Enterogastric reflex: duodenal distension, acidic chyme, fat, or hyperosmolarity → inhibits gastric secretion
- Secretin (from S cells) and CCK (from I cells) → inhibit gastric emptying and secretion
4. Functions of HCl
(i) Protein Digestion — Pepsin Activation
- HCl denatures dietary proteins, unfolding them and making peptide bonds accessible
- HCl converts pepsinogen (inactive, MW ~42,500) → pepsin (active, MW ~35,000) by autocatalytic cleavage
- Pepsin has optimal proteolytic activity at pH 1.8–3.5; inactivated above pH 5
- HCl is thus as necessary as pepsin for gastric protein digestion
(ii) Antimicrobial Barrier
- The highly acidic environment (pH ~1–2) kills most ingested pathogens
- Acts as a defense against enteric pathogens including Salmonella, Giardia lamblia, helminths
- Patients with achlorhydria (loss of acid secretion — from surgery, PPIs, H₂ blockers, chronic gastritis) have significantly increased risk of enteric colonization and infections
(iii) Iron Absorption
- HCl maintains iron in its soluble ferrous (Fe²⁺) form, which is the absorbable form in the duodenum
- Achlorhydria impairs iron absorption → iron-deficiency anemia
(iv) Calcium & Mineral Absorption
- Acid environment promotes dissolution of calcium salts, facilitating Ca²⁺ absorption in the proximal small intestine
(v) Intrinsic Factor Secretion
- Intrinsic factor (IF) is a 44-kDa glycoprotein secreted by parietal cells alongside HCl
- IF binds vitamin B₁₂ in the stomach/duodenum → IF–B₁₂ complex is absorbed in the terminal ileum
- Destruction of parietal cells (e.g., chronic gastritis, autoimmune) → loss of both acid AND intrinsic factor → pernicious anemia (megaloblastic) due to B₁₂ deficiency
(vi) Regulation of Gastric Emptying
- Acid pH of gastric contents entering the duodenum triggers secretin and CCK release, which regulate the rate of gastric emptying
(vii) Stimulus for Pancreatic Secretion
- Acid chyme in the duodenum stimulates S cells → secrete secretin → stimulates pancreatic bicarbonate secretion (to neutralize acid)
- Also stimulates CCK → pancreatic enzyme secretion and bile release
5. Clinical Correlates
| Condition | Mechanism | Consequence |
|---|---|---|
| Achlorhydria | Parietal cell destruction (autoimmune gastritis, post-gastrectomy) | ↓ Protein digestion, ↓ Fe²⁺ absorption, ↓ IF → pernicious anemia, ↑ enteric infections |
| Peptic ulcer disease | H. pylori disrupts mucosal barrier; excess acid | Mucosal erosion by HCl + pepsin |
| Zollinger–Ellison syndrome | Gastrinoma (duodenal/pancreatic tumor) → unregulated gastrin → continuous parietal cell stimulation | Severe hypersecretion of HCl → refractory gastric and duodenal ulcers, diarrhea, steatorrhea |
| PPI use (omeprazole, etc.) | Irreversible block of H⁺/K⁺-ATPase | ↓ Acid → risk of C. diff, pneumonia, hypomagnesemia, B₁₂ deficiency with long-term use |
| H₂ blockers (ranitidine, etc.) | Block histamine H₂ receptors on parietal cells | Moderate acid reduction |
Sources: Guyton & Hall Textbook of Medical Physiology, Ch. 65 | Histology: A Text and Atlas (Ross & Pawlina), Ch. 17 | Harrison's Principles of Internal Medicine 22e | Lippincott Illustrated Reviews: Biochemistry 8e
This is a shared conversation. Sign in to Orris to start your own chat.