Explain
ketone body synthesis pathway ketogenesis diagram
Synthesis of Ketone Bodies (Ketogenesis)
Where and Why It Happens
- Fatty acids undergo β-oxidation
- Pyruvate is oxidized (from glucose)
- Certain amino acids (leucine, lysine, etc.) are catabolized
The 4-Step Pathway
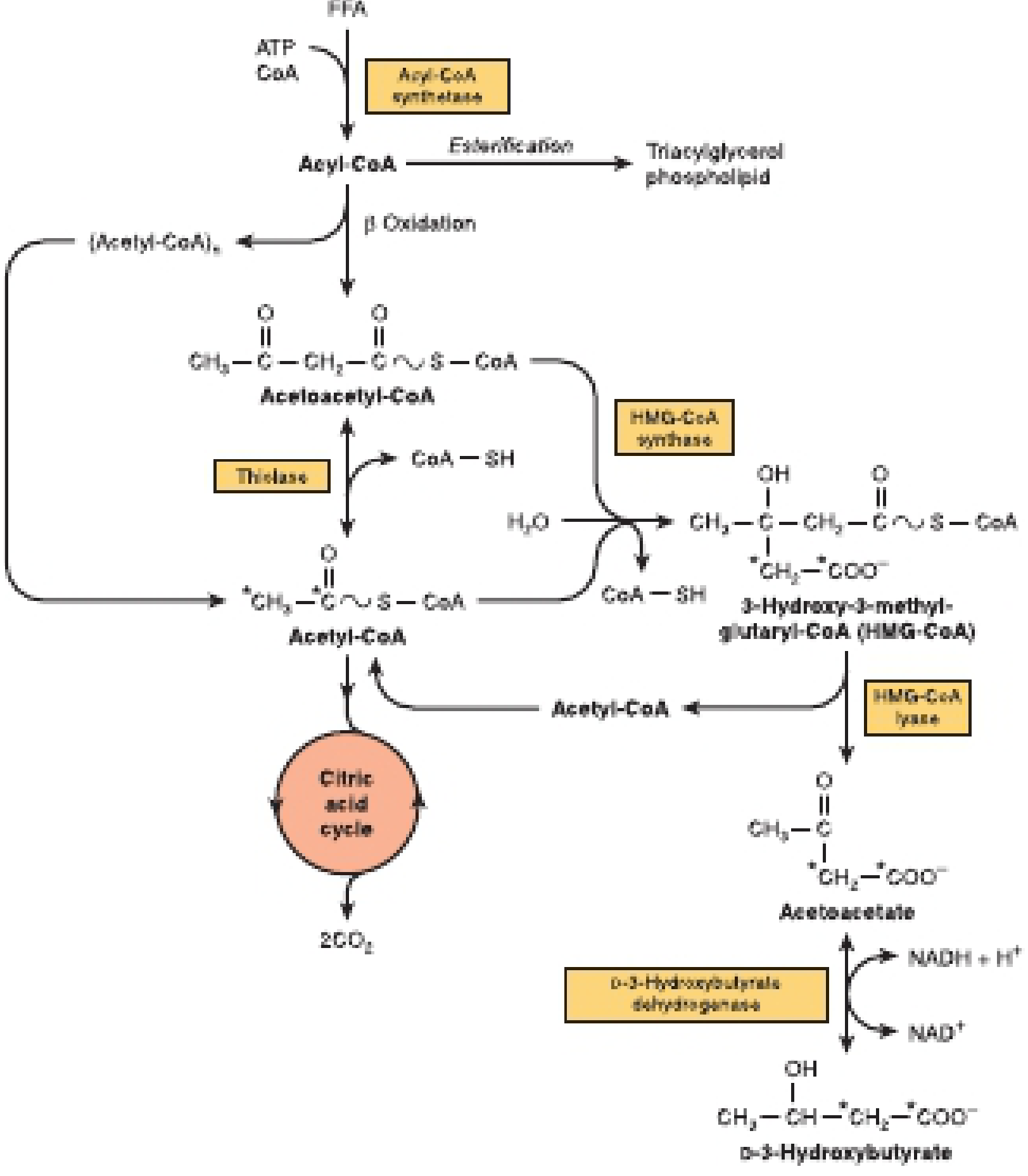
2 Acetyl-CoA → Acetoacetyl-CoA + CoA-SH
Acetoacetyl-CoA + Acetyl-CoA + H₂O → HMG-CoA + CoA-SH
HMG-CoA → Acetoacetate + Acetyl-CoA
| Route | Product | Mechanism |
|---|---|---|
| Spontaneous decarboxylation | Acetone | Non-enzymatic; CO₂ is lost; acetone is volatile and exhaled via lungs (causes "fruity breath" in ketoacidosis) |
| Enzymatic reduction | β-Hydroxybutyrate (3-hydroxybutyrate) | Catalyzed by D-3-hydroxybutyrate dehydrogenase using NADH; this is the major ketone body in the blood |
The Three Ketone Bodies at a Glance
| Ketone Body | Notes |
|---|---|
| Acetoacetate | First formed; can be used directly as fuel |
| β-Hydroxybutyrate | Most abundant in blood; major fuel for the brain during starvation |
| Acetone | Waste product; exhaled; responsible for fruity breath |
Why the Liver Cannot Use Its Own Ketone Bodies
Clinical Relevance
- In diabetic ketoacidosis (DKA), insulin deficiency causes uncontrolled lipolysis → massive Acetyl-CoA overflow → runaway ketogenesis → dangerous accumulation of ketone bodies in blood (ketonemia) and urine (ketonuria), dropping blood pH.
- In starvation/fasting, ketogenesis is a physiological adaptation to spare glucose for the brain.
- The fruity/acetone smell in a patient's breath is a clinical sign of severe ketonemia.
What do we mean by reversal of beta oxidation I don't understand
What is β-Oxidation? (The Normal Direction)
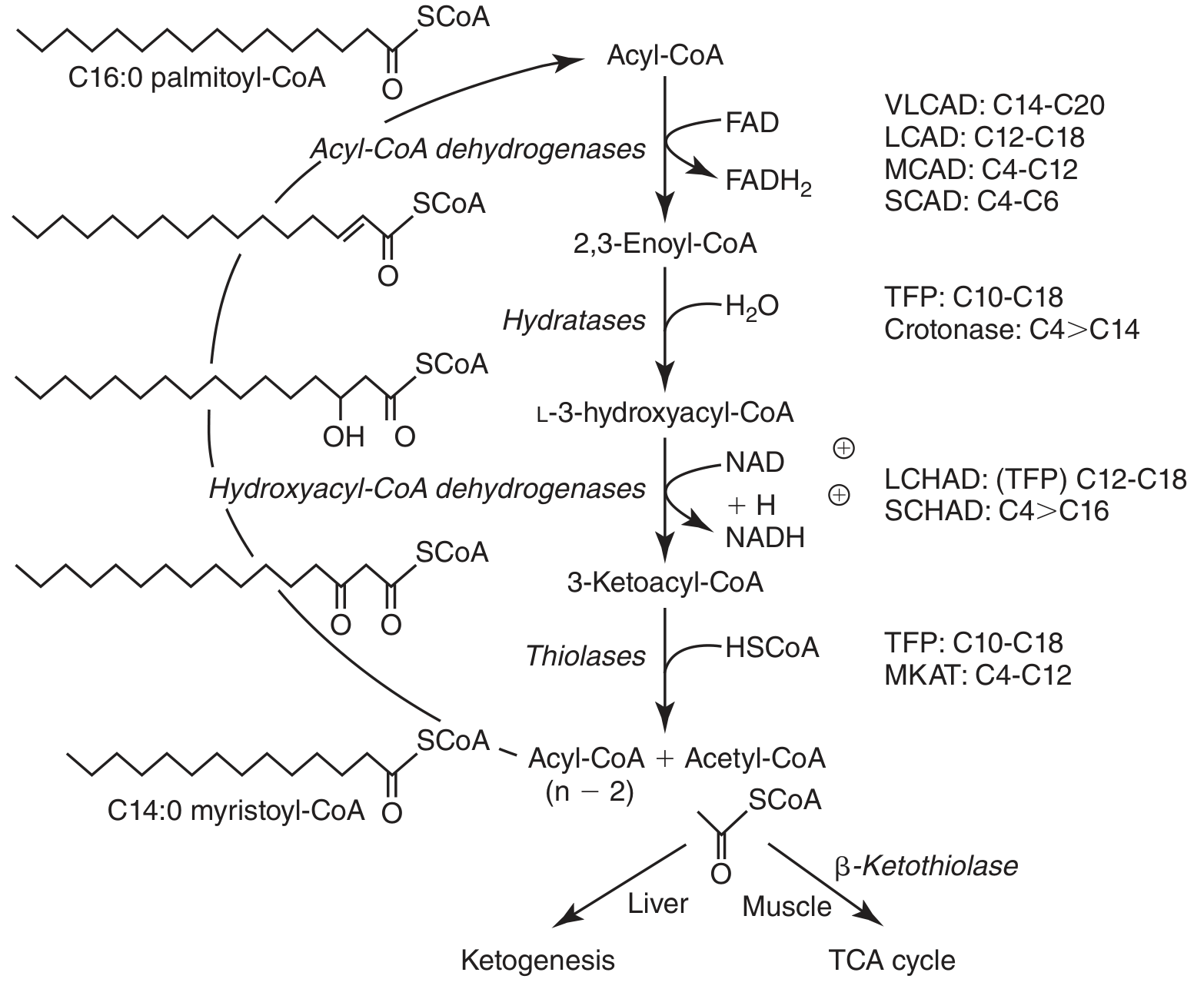
| Step | Reaction | Enzyme |
|---|---|---|
| 1 | Acyl-CoA → Enoyl-CoA (add double bond) | Acyl-CoA dehydrogenase (uses FAD) |
| 2 | Enoyl-CoA → 3-Hydroxyacyl-CoA (add water) | Enoyl-CoA hydratase |
| 3 | 3-Hydroxyacyl-CoA → 3-Ketoacyl-CoA (oxidize) | Hydroxyacyl-CoA dehydrogenase (uses NAD⁺) |
| 4 | 3-Ketoacyl-CoA → Acetyl-CoA + shorter Acyl-CoA | Thiolase (splits the chain) |
Now — What Does "Reversal of Thiolase" Mean in Ketogenesis?
2 Acetyl-CoA → Acetoacetyl-CoA + CoA-SH
Acetoacetyl-CoA + CoA-SH → 2 Acetyl-CoA (this is the normal, forward direction)
A Simple Analogy
- In β-oxidation (forward): the scissors cut a chain apart → 2 pieces
- In ketogenesis (reverse): the scissors work backward, acting like glue → joining 2 pieces together
Why Can the Reaction Run Backwards?
- Concentration of substrates and products — if Acetyl-CoA accumulates massively (e.g., during starvation or uncontrolled diabetes), the reaction is pushed toward condensation
- Energy state of the cell
Summary in One Sentence
"Reversal of the thiolase reaction" simply means that instead of using thiolase to break acetoacetyl-CoA apart (as in β-oxidation), the liver uses it to build acetoacetyl-CoA by joining two acetyl-CoA molecules together — the very first step of ketogenesis.
But acetoacetyl coa isn't present in beta oxidation
β-Oxidation of a 4-Carbon Fatty Acid (Butyryl-CoA)
Butyryl-CoA (4 carbons)
↓ Step 1: Acyl-CoA dehydrogenase
Crotonyl-CoA (4 carbons, with double bond)
↓ Step 2: Enoyl-CoA hydratase
3-Hydroxybutyryl-CoA
↓ Step 3: Hydroxyacyl-CoA dehydrogenase
3-Ketobutyryl-CoA = ACETOACETYL-CoA ✓
↓ Step 4: THIOLASE + CoA-SH
Acetyl-CoA + Acetyl-CoA
Now Compare: Ketogenesis Does the Opposite
| β-Oxidation (last step) | Ketogenesis (first step) | |
|---|---|---|
| Substrate | Acetoacetyl-CoA + CoA-SH | 2 × Acetyl-CoA |
| Product | 2 × Acetyl-CoA | Acetoacetyl-CoA + CoA-SH |
| Enzyme | Thiolase | Thiolase |
| Direction | Cleavage (forward) | Condensation (reverse) |
Why This Makes Sense Physiologically
- β-Oxidation produces a huge flood of Acetyl-CoA
- The TCA cycle gets saturated — it cannot accept all the Acetyl-CoA
- Acetoacetyl-CoA (the intermediate before the final thiolase step) accumulates
- Instead of being cleaved, it gets diverted → HMG-CoA → acetoacetate → ketone bodies
I don't get bring the steps out let me see of beta oxidation and ketogensis don't add the physiological stuff
β-Oxidation (one cycle, using a 4-carbon fatty acid)
Butyryl-CoA (4C)
│
│ Step 1 — Acyl-CoA dehydrogenase [removes 2H, uses FAD → FADH₂]
▼
Crotonyl-CoA (4C, double bond between C2–C3)
│
│ Step 2 — Enoyl-CoA hydratase [adds H₂O]
▼
L-3-Hydroxybutyryl-CoA (4C, OH group on C3)
│
│ Step 3 — Hydroxyacyl-CoA dehydrogenase [removes 2H, uses NAD⁺ → NADH]
▼
3-Ketobutyryl-CoA = Acetoacetyl-CoA (4C, ketone on C3)
│
│ Step 4 — Thiolase [adds CoA-SH, CLEAVES the bond]
▼
Acetyl-CoA + Acetyl-CoA
Ketogenesis
Acetyl-CoA + Acetyl-CoA
│
│ Step 1 — Thiolase [releases CoA-SH, JOINS the two]
▼
Acetoacetyl-CoA (4C)
│
│ Step 2 — HMG-CoA synthase [adds another Acetyl-CoA + H₂O, releases CoA-SH]
▼
HMG-CoA (6C)
│
│ Step 3 — HMG-CoA lyase [cleaves, releases Acetyl-CoA]
▼
Acetoacetate
│
├─ Step 4a — D-3-Hydroxybutyrate dehydrogenase [adds 2H, uses NADH]
│ ▼
│ β-Hydroxybutyrate
│
└─ Step 4b — Spontaneous decarboxylation [loses CO₂]
▼
Acetone
The Connection
β-Oxidation: Acetoacetyl-CoA → [Thiolase] → 2 Acetyl-CoA (splits)
Ketogenesis: 2 Acetyl-CoA → [Thiolase] → Acetoacetyl-CoA (joins)
Pls in written form
β-Oxidation Steps
Ketogenesis Steps
The Connection Between the Two
Pls do tell how nadh and fadh2 in formed and fix this acetoacetyl where it is supposed to be like make a note on beta oxidation something can say cuz I am a 200lvl student MBBS having test next week and oral exam
Beta-Oxidation of Fatty Acids — Complete Study Notes (200L MBBS)
What is Beta-Oxidation?
Before the Cycle Starts — Activation
The Four Steps of One Cycle
What Happens at the Very Last Cycle
Energy Yield Per Cycle
- 1 FADH₂ (from Step 1) → gives approximately 1.5 ATP in the electron transport chain
- 1 NADH (from Step 3) → gives approximately 2.5 ATP in the electron transport chain
- 1 Acetyl-CoA → enters TCA cycle and gives approximately 10 ATP
How FADH₂ and NADH are Formed — Simple Explanation
- NAD⁺ + 2H → NADH + H⁺ — NAD⁺ picks up 2 electrons and 1 proton; it becomes NADH. The other proton is released into solution.
- FAD + 2H → FADH₂ — FAD picks up both hydrogens (both protons and both electrons) and becomes FADH₂.
Quick Summary Table for Revision
| Step | Enzyme | What Happens | Coenzyme Reduced |
|---|---|---|---|
| 1 | Acyl-CoA dehydrogenase | Dehydrogenation — double bond formed | FAD → FADH₂ |
| 2 | Enoyl-CoA hydratase | Hydration — water added | None |
| 3 | Hydroxyacyl-CoA dehydrogenase | Dehydrogenation — alcohol → ketone | NAD⁺ → NADH |
| 4 | Thiolase | Thiolysis — chain cleaved, acetyl-CoA released | None |
One-Line Oral Exam Answer
"Beta-oxidation is a four-step repeating cycle in the mitochondrial matrix that breaks fatty acids into acetyl-CoA, producing one FADH₂ and one NADH per cycle. The final cycle produces acetoacetyl-CoA, which in the liver is the entry point for ketogenesis rather than being cleaved to acetyl-CoA."
What I don't get is it that the last beta ketoacyle produced is the shortened acetoacetyl coa the four carbon or what
Tracing the Chain — Using a 6-Carbon Fatty Acid
- Releases acetyl-CoA (2 carbons) ✓
- Leaves behind butyryl-CoA (4 carbons) — this goes back to Step 1
- Releases acetyl-CoA (2 carbons) ✓
- Releases another acetyl-CoA (2 carbons) ✓ — because there is nothing left, both pieces are 2 carbons
So to Directly Answer Your Question
- In Cycle 1 on a 6-carbon chain → 6-carbon 3-ketoacyl-CoA
- In Cycle 2 on the remaining 4-carbon chain → 4-carbon 3-ketoacyl-CoA = acetoacetyl-CoA
The One Sentence Answer
Every cycle of beta-oxidation produces a 3-ketoacyl-CoA. When you are on the last cycle and the chain is only 4 carbons long, that 3-ketoacyl-CoA is acetoacetyl-CoA — same intermediate, just a 4-carbon version with its own name.
Now arrange all this into an exam format use standard English like reduced but add explanation for me talk about carbons Atp and circle 1 and two one more thing beta oxidation is the oxidation of the fatty acid at the beta carbon to release Atp
Beta-Oxidation of Fatty Acids
Definition
Activation (Before the Cycle)
The Four Steps of Beta-Oxidation
Step 1 — Oxidation at the Alpha–Beta Carbon Bond
How FADH₂ is formed: FAD accepts both hydrogen atoms (each carrying one electron). FAD + 2H → FADH₂. This is a low-energy oxidation so FAD, not NAD⁺, is the acceptor here.
Step 2 — Hydration
Step 3 — Oxidation at the Beta Carbon
How NADH is formed: NAD⁺ accepts 2 electrons and 1 proton. NAD⁺ + 2H → NADH + H⁺. This is a higher-energy oxidation (alcohol → ketone) so NAD⁺, which has greater energy capacity, is used here rather than FAD.
Step 4 — Thiolysis (Chain Cleavage)
- One molecule of acetyl-CoA (2 carbons) — goes to TCA cycle
- One molecule of fatty acyl-CoA shortened by 2 carbons — re-enters Step 1
Cycle 1 and Cycle 2 — Following the Carbons
Cycle 1
- Acetyl-CoA (2 carbons) → enters TCA cycle
- Butyryl-CoA (4 carbons) → re-enters Step 1 for Cycle 2
Cycle 2
- Acetyl-CoA (2 carbons)
- Acetyl-CoA (2 carbons)
The Acetoacetyl-CoA Note
- In muscle and most tissues — thiolase cleaves it normally into two acetyl-CoA molecules, which enter the TCA cycle.
- In the liver — when acetyl-CoA is in excess, acetoacetyl-CoA is not cleaved. Instead, it is diverted into ketogenesis. In ketogenesis, thiolase runs in the reverse direction, joining two acetyl-CoA molecules to form acetoacetyl-CoA instead of splitting it. This is what the phrase "reversal of the thiolase reaction" means.
Energy Yield Per Cycle
| Product | ATP Yield |
|---|---|
| 1 FADH₂ | 1.5 ATP (via electron transport chain) |
| 1 NADH | 2.5 ATP (via electron transport chain) |
| 1 Acetyl-CoA | ~10 ATP (via TCA cycle) |
| Total per cycle | ~14 ATP |
One-Line Exam Definition
Beta-oxidation is the sequential oxidation of a fatty acid at the beta carbon, producing one FADH₂, one NADH, and one acetyl-CoA per cycle, with the final cycle yielding acetoacetyl-CoA, which is cleaved into two acetyl-CoA molecules or diverted to ketogenesis in the liver.