Barbiturates in Anaesthesia MD
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
I now have all the content needed for a comprehensive MD-level answer. Let me compile the complete response.
Barbiturates in Anaesthesia
1. Historical Background
Barbiturates were first synthesized in 1903 by Fischer and von Mering. The first barbiturate used for induction of anaesthesia was diethylbarbituric acid (barbital), but it was the introduction of hexobarbital in 1927 that made barbiturate induction popular. Thiopental was synthesized in 1932 by Volwiler and Tabern and first used clinically by John Lundy and Ralph Waters in 1934 — for decades it remained the gold standard intravenous induction agent. Methohexital was introduced clinically in 1957 by V.K. Stoelting.
- Morgan and Mikhail's Clinical Anesthesiology, 7e
- Miller's Anesthesia, 10e
2. Chemistry & Formulation
Barbiturates are derivatives of barbituric acid (2,4,6-trioxohexahydropyrimidine), formed by condensation of malonic acid and urea. The nucleus itself is hypnotically inactive; hypnotic activity arises from substitution at position 5 (aryl or alkyl groups) and at positions 1 and 2.
Two major classes:
- Thiobarbiturates — sulfur at position 2: thiopental, thiamylal
- Oxybarbiturates — oxygen at position 2: methohexital
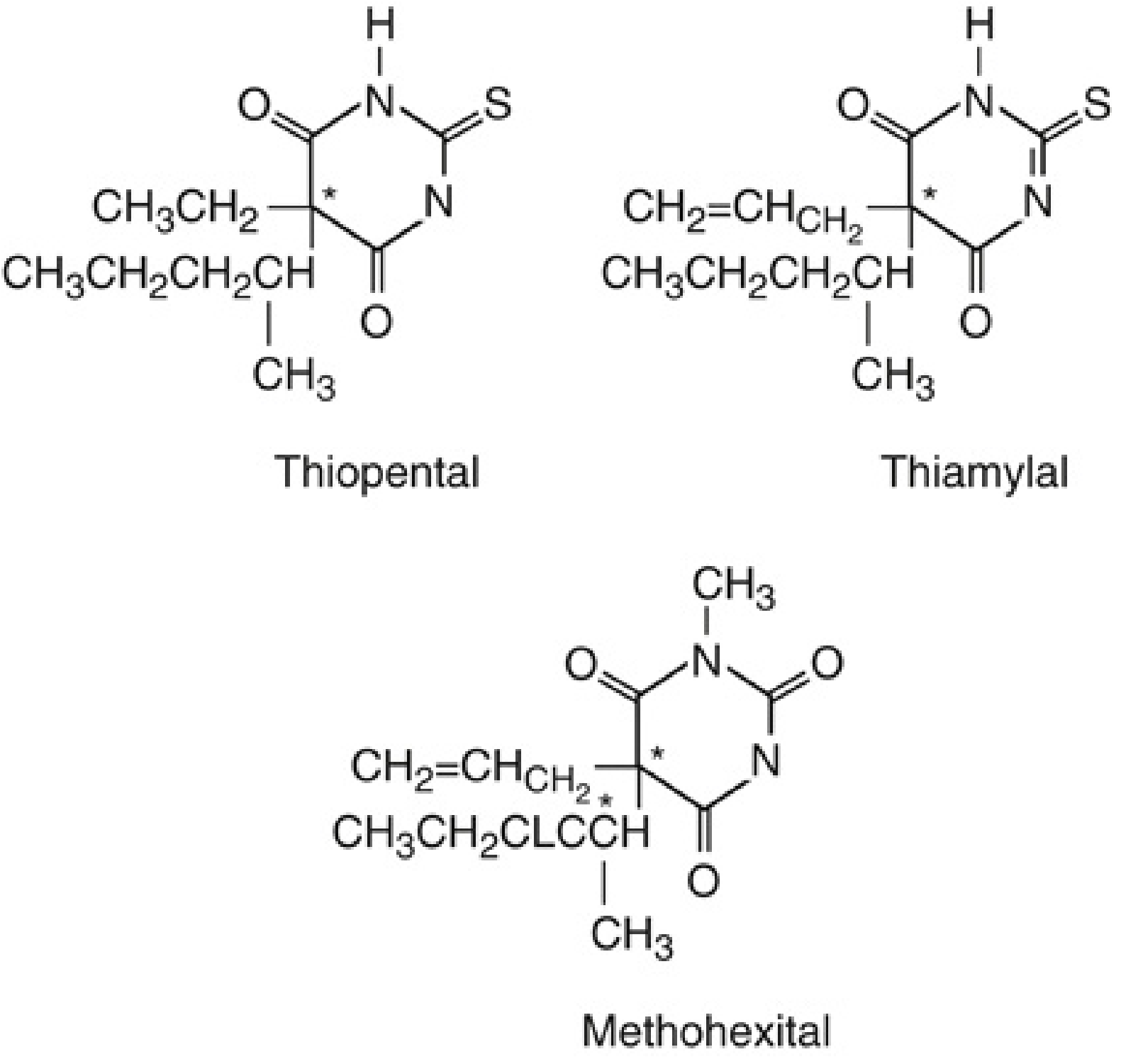
Structure-Activity Relationships:
- Longer alkyl chain at C5 → increased hypnotic potency but also toxicity
- Phenyl group at C5 → anticonvulsant activity (phenobarbital)
- Sulfur at C2 (thiobarbiturates) → increased lipid solubility and faster CNS onset
- N-methylation at position 1 → increased CNS excitability (methohexital's proconvulsant property)
- Branching at C5 → increased potency and shorter duration
Formulation:
-
Supplied as sodium salts mixed with 6% anhydrous sodium carbonate, reconstituted in water, 5% dextrose, or normal saline
-
Thiopental: 2.5% solution; thiamylal: 2% solution; methohexital: 1% solution
-
Solution is highly alkaline (pH 10–11)
-
Thiobarbiturates stable for 1 week refrigerated; methohexital stable for 6 weeks after reconstitution
-
Cannot be mixed with acidic solutions (atracurium, vecuronium, rocuronium, suxamethonium, alfentanil, sufentanil, midazolam) — precipitation occurs as the free acid
-
Miller's Anesthesia, 10e, p. 2492–2494
-
Goodman & Gilman's, p. 1127
3. Mechanism of Action
Barbiturates produce anaesthesia through two complementary mechanisms:
- Enhancement of inhibitory neurotransmission — potentiate GABA-A receptor–mediated Cl⁻ current (at a site distinct from benzodiazepines; at high concentrations they can directly activate the channel even without GABA)
- Inhibition of excitatory neurotransmission — suppress glutamate and acetylcholine receptor activity
They do not possess analgesic properties; some evidence suggests they may even lower the pain threshold (hyperalgesia).
- Katzung's Basic & Clinical Pharmacology, 16e
- Miller's Anesthesia, 10e
4. Pharmacokinetics
| Parameter | Thiopental | Methohexital |
|---|---|---|
| Protein binding | ~85% | ~73% |
| pKa | 7.6 | 7.9 |
| Onset (IV) | 15–30 s | ~30 s |
| Elimination half-life | Long (hours–days) | Shorter (faster clearance) |
| Primary elimination | Hepatic (oxidation, N-dealkylation, desulfuration) | Hepatic (greater plasma clearance) |
Recovery mechanism: After a single bolus, recovery depends on redistribution from brain to muscle and fat, not on hepatic metabolism — hence both agents have similar emergence profiles after a single dose.
After repeated bolus or infusion, recovery depends on elimination (metabolism), and thiopental accumulates markedly — its context-sensitive half-time increases substantially. Methohexital accumulates less because of its larger plasma clearance.
A small fraction of thiopental undergoes desulfuration to pentobarbital (a longer-acting hypnotic).
Protein binding: Conditions that reduce serum albumin (hepatic disease, burns, malnutrition, uremia, malignancy) increase free drug concentration and the hypnotic effect of a given dose.
- Katzung's Basic & Clinical Pharmacology, 16e, p. 710
- Miller's Anesthesia, 10e
5. Organ System Effects
5.1 Central Nervous System
- Dose-dependent CNS depression: sedation → hypnosis → general anaesthesia → burst suppression → isoelectric EEG
- No analgesia (may cause hyperalgesia)
- Potent cerebral vasoconstrictors → ↓ cerebral blood flow (CBF) → ↓ cerebral blood volume (CBV) → ↓ intracranial pressure (ICP)
- ↓ CMRO₂ (cerebral metabolic rate for O₂) in a dose-dependent manner up to EEG burst suppression
- Neuroprotection: effective against focal cerebral ischaemia (stroke, retraction, temporary clips during aneurysm surgery); not effective after global ischaemia (cardiac arrest)
- Anticonvulsant (except methohexital)
- Methohexital exception: activates epileptic foci → drug of choice for ECT anaesthesia and for identifying epileptic foci intraoperatively
5.2 Cardiovascular System
- ↓ Systemic blood pressure primarily from peripheral vasodilation (predominantly venodilation)
- Direct negative inotropic effect on the heart
- Reflex tachycardia (baroreceptor reflex is partially but less completely blunted compared to propofol)
- BP decrease is usually smaller than with propofol
- Thiopental maintains cardiac output better than equipotent doses of propofol
- Caution in hypovolaemia, haemorrhagic shock, cardiac disease
5.3 Respiratory System
- Dose-dependent respiratory depression — ↓ tidal volume, ↓ respiratory rate, ↓ hypercapnic and hypoxic ventilatory responses
- Apnoea is common after induction doses, especially with rapid injection
- Does NOT cause bronchodilation (unlike propofol and ketamine) — avoid in active bronchospasm
5.4 Hepatic/Renal
-
Porphyria: Barbiturates stimulate aminolevulinic acid (ALA) synthetase → ↑ porphyrin production → can precipitate an acute porphyric crisis → absolute contraindication in acute intermittent porphyria (AIP)
-
Katzung's, pp. 2403–2413
-
Miller's Anesthesia, 10e
6. Clinical Uses
| Indication | Agent | Dose |
|---|---|---|
| Induction of GA (adult) | Thiopental | 3–4 mg/kg IV |
| Induction of GA (adult) | Methohexital | 1–2 mg/kg IV |
| ECT anaesthesia | Methohexital (preferred) | 0.5–1 mg/kg IV |
| Maintenance of GA (infusion) | Methohexital | 50–150 mcg/kg/min |
| Paediatric premedication (rectal) | Methohexital | 25 mg/kg rectal (10% solution) |
| ICP reduction / cerebral protection | Thiopental | Titrated to burst suppression |
| Barbiturate coma (refractory ICP) | Thiopental/pentobarbital | Loading dose, then infusion |
Dose reductions required in: elderly, haemorrhagic shock, low cardiac output, lean body mass extremes, obesity, hypoalbuminaemia, severe anaemia, burns, uremia, malignancy, and with opioid/benzodiazepine premedication.
- Miller's Anesthesia, 10e, p. 2509–2511
7. Contraindications
| Contraindication | Reason |
|---|---|
| Acute intermittent porphyria | Stimulates ALA synthetase → porphyric crisis |
| Known hypersensitivity | Anaphylaxis / anaphylactoid reactions |
| Absence of resuscitation facilities | Apnoea risk |
| Severe cardiovascular compromise | Vasodilation + negative inotropy |
| No IV access | Administration requires IV access |
- Miller's Anesthesia, 10e
8. Adverse Effects & Complications
| Effect | Details |
|---|---|
| Apnoea | Common after induction dose; dose/rate dependent |
| Cardiovascular depression | Hypotension, especially in hypovolaemia |
| Laryngospasm | More likely with airway stimulation under light barbiturate anaesthesia (compared to propofol) |
| Pain on injection | Mild; less than propofol |
| Intra-arterial injection | Can cause intense vasospasm, endarteritis, gangrene — requires immediate treatment (dilute, papaverine, sympathetic block) |
| Subcutaneous extravasation | Highly alkaline → tissue necrosis |
| Anaphylaxis | Rare; mediated by direct histamine release |
| Excitatory phenomena | Hiccup, tremor, myoclonic movements (esp. methohexital) |
| Prolonged recovery | After infusion/repeated doses due to accumulation |
| Adrenal suppression | NOT a barbiturate issue (etomidate) |
9. Comparison: Thiopental vs. Methohexital vs. Propofol
| Property | Thiopental | Methohexital | Propofol |
|---|---|---|---|
| Class | Thiobarbiturate | Oxybarbiturate | Phenol |
| Induction dose | 3–4 mg/kg | 1–2 mg/kg | 1.5–2.5 mg/kg |
| Onset | 15–30 s | ~30 s | ~30 s |
| Anticonvulsant | Yes | No (proconvulsant) | Yes |
| ECT | No | First choice | Used but shortens seizure |
| BP effect | ↓ (moderate) | ↓ (moderate) | ↓ (greater) |
| Recovery after infusion | Prolonged | Faster | Fastest |
| Anti-emetic | No | No | Yes |
| Pain on injection | Mild | Mild | Common |
| Availability (US) | Limited/discontinued | Available | Widely available |
- Miller's Anesthesia, 10e
- Katzung's, p. 2390
10. Current Status
Barbiturates have been largely replaced by propofol as the agent of choice for IV induction of anaesthesia worldwide. This is due to propofol's superior pharmacokinetic profile (faster, more complete recovery), anti-emetic properties, and better recovery characteristics.
Thiopental's availability has been further restricted by legal controversies around its use in lethal injection (US executions), leading to a shortage and withdrawal from the US market.
Methohexital remains in active clinical use primarily for ECT anaesthesia and as a rectal premedication in paediatric patients.
Thiopental retains a role in:
-
Rapid sequence induction where propofol is contraindicated
-
Raised ICP management / barbiturate coma
-
Neuroprotection during temporary vascular occlusion in neurosurgery
-
Status epilepticus refractory to other agents
-
Miller's Anesthesia, 10e; Katzung's Basic & Clinical Pharmacology, 16e; Morgan & Mikhail's Clinical Anesthesiology, 7e; Goodman & Gilman's Pharmacological Basis of Therapeutics
Make a ppt
Reading File
Reading File
Reading File
Running Command
Running Command
Writing File
Running Command
Running Command
Your presentation is ready!
Barbiturates in Anaesthesia
Presentation · PPTX
12 slides with a deep navy + teal + amber color scheme:
| Slide | Content |
|---|---|
| 1 | Title slide |
| 2 | Outline |
| 3 | Historical Background (timeline) |
| 4 | Chemistry & SAR + Formulation table |
| 5 | Mechanism of Action (GABA-A + excitatory suppression) |
| 6 | Pharmacokinetics (comparison table) |
| 7 | CNS Effects (dose-response ladder + effects) |
| 8 | CVS & Respiratory Effects |
| 9 | Clinical Uses & Dosing (full table) |
| 10 | Contraindications, Adverse Effects & Injection Complications |
| 11 | Thiopental vs Methohexital vs Propofol comparison |
| 12 | Current Status & Summary |
Viva question in MD practicals on Barbiturates
Here is a comprehensive set of MD Anaesthesia viva questions on Barbiturates, organized by difficulty and theme — exactly as they come up in practical/clinical viva sessions.
MD Anaesthesia Viva — Barbiturates
🔬 CHEMISTRY & PHARMACOLOGY (Basic Science Questions)
Q1. What is the basic nucleus of barbiturates?
Barbituric acid — formed by condensation of malonic acid and urea (2,4,6-trioxohexahydropyrimidine). The nucleus itself is hypnotically inactive.
Q2. What substitutions at which positions confer hypnotic activity?
- Position 5 — aryl or alkyl substitution → hypnotic/sedative effect
- Position 2 — oxygen → oxybarbiturate; sulfur → thiobarbiturate (↑ lipid solubility, faster onset)
- Position 1 (N-methylation) — e.g. methohexital → ↑ CNS excitability, proconvulsant
- C5 phenyl group → anticonvulsant activity (e.g. phenobarbital)
Q3. How are barbiturates classified? Give anaesthetic examples.
| Class | C2 substitution | Examples |
|---|---|---|
| Thiobarbiturates | Sulfur | Thiopental, Thiamylal |
| Oxybarbiturates | Oxygen | Methohexital, Phenobarbital |
Q4. Why are barbiturates formulated as sodium salts? What is the pH of the solution?
Through keto-enol tautomerism, the C2 oxygen/sulfur becomes reactive in enol form, allowing formation of water-soluble sodium salts. Reconstituted in water with 6% anhydrous sodium carbonate → pH 10–11 (highly alkaline).
Q5. Why can't you mix thiopental with rocuronium or suxamethonium in the same syringe?
Rocuronium, suxamethonium, midazolam, alfentanil, and atracurium are all acidic solutions. Mixing with highly alkaline thiopental (pH 10–11) causes precipitation of the barbiturate as the free acid → can occlude the IV line, especially critical during RSI.
Q6. What is the shelf life of reconstituted thiopental and methohexital?
- Thiopental (thiobarbiturate): 1 week refrigerated
- Methohexital (oxybarbiturate): 6 weeks refrigerated
⚗️ MECHANISM OF ACTION
Q7. What is the mechanism of action of barbiturates?
Two mechanisms:
- Enhance inhibitory transmission — potentiate GABA-A receptor Cl⁻ channel (prolong channel opening duration); at high doses directly activate GABA-A even without GABA
- Inhibit excitatory transmission — suppress AMPA glutamate receptors and nicotinic ACh receptors
Key point: Barbiturate binding site is distinct from the benzodiazepine site on the GABA-A receptor.
Q8. How does the barbiturate site on GABA-A differ from the benzodiazepine site?
| Feature | Barbiturates | Benzodiazepines |
|---|---|---|
| Binding site | β subunit | α–γ interface |
| Effect | Prolong Cl⁻ channel duration | ↑ frequency of channel opening |
| Direct activation | Yes (high dose) | No |
| Reversal agent | None | Flumazenil |
Q9. Do barbiturates have analgesic properties?
No. They are purely hypnotic. They may actually lower the pain threshold (hyperalgesia). Additional analgesia from opioids or volatile agents is always required during surgery.
🔄 PHARMACOKINETICS
Q10. Explain the mechanism of termination of action of a single bolus of thiopental.
After a single bolus, recovery is due to redistribution — not hepatic metabolism. Thiopental is highly lipid-soluble and rapidly redistributes from the highly perfused brain → muscle (vessel-rich group) → fat. The brain concentration falls below the threshold for anaesthesia within minutes.Context: The elimination half-life is hours–days, but this is irrelevant after a single dose. This is why thiopental gives "rapid emergence" from a single induction dose despite slow metabolism.
Q11. Why is thiopental NOT suitable for infusion/TIVA?
After repeated boluses or infusion, the peripheral compartments (especially fat) become saturated. Recovery then depends on elimination (hepatic metabolism) rather than redistribution. The context-sensitive half-time increases markedly → prolonged sedation/hangover. Methohexital and propofol accumulate far less.
Q12. What is context-sensitive half-time? How does it apply to barbiturates?
The time for plasma concentration to fall by 50% after terminating an infusion, as a function of infusion duration. Thiopental's context-sensitive half-time increases dramatically with infusion duration because its large fat reservoir slowly releases drug back into plasma. This makes it unsuitable for maintenance infusions.
Q13. What happens to thiopental in patients with hypoalbuminaemia?
Thiopental is ~85% protein-bound (primarily to albumin). In hypoalbuminaemia (liver disease, burns, malnutrition, nephrotic syndrome), free drug fraction increases → greater CNS effect from the same dose → reduce the induction dose.
Q14. What is the product of thiopental's desulfuration?
Pentobarbital — a longer-acting barbiturate hypnotic. This accounts for a small fraction of thiopental's metabolism but contributes to prolonged sedation after large doses.
🫀 ORGAN SYSTEM EFFECTS
Q15. What are the CNS effects of thiopental in order of increasing dose?
Sedation → Hypnosis → General Anaesthesia → Burst Suppression → Isoelectric EEGAt each level: ↓ CBF, ↓ CBV, ↓ ICP, ↓ CMRO₂ (all dose-dependent)
Q16. How do barbiturates reduce ICP?
- Potent cerebral vasoconstrictors → ↓ cerebral blood volume (CBV) → ↓ ICP
- ↓ CMRO₂ → reduced metabolic demand → cerebral vasoconstriction (flow-metabolism coupling)
- Effect is dose-dependent and maximal at EEG burst suppression
Q17. Do barbiturates protect the brain from ischemia?
- Focal cerebral ischaemia (stroke, surgical retraction, temporary clips during aneurysm surgery) — YES, barbiturates are neuroprotective
- Global cerebral ischaemia (cardiac arrest) — NO, barbiturates do not reduce injury
The distinction is because focal ischaemia has penumbral tissue where reducing CMRO₂ helps; global ischaemia has no zone of partial perfusion.
Q18. What are the cardiovascular effects of thiopental?
- ↓ BP — primarily from peripheral vasodilation (venodilation)
- Direct negative inotropy (↓ cardiac contractility)
- Reflex tachycardia (partial baroreceptor blunting)
- BP decrease is less than propofol; cardiac output is better maintained than propofol
- Caution in hypovolaemia, haemorrhagic shock, cardiac disease
Q19. Why does thiopental cause tachycardia?
Two reasons:
- Reflex sympathetic activation in response to hypotension (baroreflex — though partially blunted)
- Direct vagolytic effect of thiopental (mild)
Q20. What are the respiratory effects?
- Dose-dependent ↓ tidal volume and respiratory rate
- Apnoea common after induction dose (rate- and dose-dependent)
- ↓ hypercapnic and hypoxic ventilatory drives
- NO bronchodilation — unlike propofol and ketamine
- Risk of laryngospasm under light depth with airway stimulation
💊 CLINICAL USE
Q21. What is the induction dose of thiopental? What factors reduce it?
Standard: 3–4 mg/kg IV (ED50 ~2.2–2.7 mg/kg)Dose is reduced in:
- Elderly patients
- Haemorrhagic shock / ↓ cardiac output
- Hypoalbuminaemia (burns, liver disease, malnutrition, uraemia)
- Opioid or benzodiazepine premedication
- Severe anaemia, malignancy, obesity, extremes of lean body mass
Q22. Why is methohexital the drug of choice for ECT?
- It is a proconvulsant — activates epileptic foci (N-methylation at position 1)
- Produces longer, better-quality seizures during ECT compared to thiopental or propofol
- Propofol shortens seizure duration; thiopental is anticonvulsant and reduces seizure quality
- Faster recovery due to higher plasma clearance
- Note: propofol can still be used if methohexital is unavailable, but seizure monitoring is important
Q23. What is the rectal dose of methohexital in paediatric patients?
25 mg/kg rectally as a 10% solution through a 14F catheter inserted 7 cm into the rectum. Sleep onset is rapid; mean peak plasma levels occur within 14 minutes.
Q24. What is barbiturate coma? When is it used?
High-dose barbiturate (thiopental or pentobarbital) infusion titrated to EEG burst suppression to maximally reduce CMRO₂ and ICP.Indications: Refractory raised ICP (severe TBI, subarachnoid haemorrhage) not responding to other measures.Monitoring: Continuous EEG (aim for burst suppression pattern), haemodynamic monitoring (vasopressors often required).
⚠️ CONTRAINDICATIONS & COMPLICATIONS
Q25. What is the most important absolute contraindication to barbiturates?
Acute Intermittent Porphyria (AIP)Barbiturates stimulate aminolevulinic acid (ALA) synthetase (the rate-limiting enzyme in haem synthesis) → ↑ porphyrin production → acute porphyric crisis → abdominal pain, neurological manifestations, cardiovascular instability, potentially fatal.Safe alternatives for induction in porphyria: Propofol or ketamine
Q26. What happens if thiopental is injected intra-arterially?
Mechanism: Crystallisation of thiopental as free acid in the acidic arterial blood → microcrystal embolism → intense vasospasm → endarteritis obliterans → thrombosis → distal gangreneManagement:
- Do NOT remove the needle/cannula (use it for treatment)
- Dilute with normal saline
- Inject papaverine (vasodilator) through the same cannula
- Sympathetic block (stellate ganglion block or brachial plexus block) to relieve vasospasm
- Systemic anticoagulation (heparin)
- Warm soaks, analgesia
Q27. What happens with subcutaneous extravasation of thiopental?
Highly alkaline solution (pH 10–11) → chemical cellulitis and tissue necrosisManagement: Hyaluronidase infiltration (to disperse the drug), warm soaks, elevation, analgesia
Q28. What are the excitatory phenomena seen with methohexital and why?
- Hiccup, tremor, myoclonic movements
- Due to N-methylation at position 1 → subcortical excitation (disinhibition of inhibitory circuits)
- More common with methohexital than thiopental
- Can be reduced by opioid premedication
🔍 HIGHER-ORDER / TRICKY VIVA QUESTIONS
Q29. Thiopental is highly lipid-soluble — so why does it NOT have a long duration of action after a single bolus?
Because initial recovery is determined by redistribution, not by elimination. High lipid solubility means thiopental rapidly distributes from brain (highly perfused, rapid equilibration) to muscle, then slowly to fat. The brain concentration falls below the anaesthetic threshold within minutes, even though the drug is still present in the body and the elimination half-life is long. This is the "redistribution principle."
Q30. If thiopental has a long half-life and propofol a short one — why does propofol give better recovery from TIVA?
Propofol has a very high plasma clearance (20–30 mL/kg/min, exceeds hepatic blood flow — extrahepatic metabolism). Its context-sensitive half-time remains short even after prolonged infusions. Thiopental, despite hepatic metabolism, has a large volume of distribution and low clearance → prolonged context-sensitive half-time → hangover effect.
Q31. A patient with known porphyria requires emergency anaesthesia — what do you use?
- Induction: Propofol (safe in porphyria) or ketamine
- Maintenance: Propofol TIVA or volatile agents (isoflurane, sevoflurane — generally considered safe)
- Avoid: All barbiturates, etomidate (questionable), some opioids
- Regional anaesthesia preferred if feasible
Q32. Why does thiopental not cause nausea/vomiting postoperatively?
It has no antiemetic properties (unlike propofol) — but also no direct emetogenic effect. PONV rates are intermediate between propofol (antiemetic) and volatile agents (emetogenic). Thiopental's PONV profile is neutral.
Q33. Compare the EEG effects of thiopental and methohexital.
| Feature | Thiopental | Methohexital |
|---|---|---|
| General EEG effect | Progressive suppression | Activates epileptic foci |
| Anticonvulsant | Yes | No |
| Burst suppression | Yes (high dose) | Yes (but also seizures reported) |
| Use in epilepsy surgery | Anticonvulsant — may suppress foci | Can activate and map epileptic foci intraoperatively |
| ECT | Reduces seizure quality | Prolongs/improves seizure quality |
Q34. What is the awareness risk with thiopental vs propofol?
A review of anaesthetic agents and awareness risk found:
- Benzodiazepines reduce awareness most effectively
- Ketamine and etomidate reduce wakefulness compared to thiopental
- Thiopental has an intermediate awareness profile — not the best for preventing awareness when used as a maintenance agent
- Propofol TIVA has a lower awareness risk than volatile agent-based techniques
Q35. What is the pKa of thiopental and why is it clinically significant?
pKa = 7.6 — very close to physiological pH (7.4). This means at body pH, a significant fraction exists in the unionised (lipid-soluble) form → rapid CNS penetration → fast onset of action.In acidosis (↓ pH), more drug is in unionised form → enhanced CNS effect → dose should be reduced in acidotic patients.
📝 RAPID-FIRE VIVA ROUND
| Question | Answer |
|---|---|
| First barbiturate synthesized | Barbituric acid (1864, Baeyer); first hypnotic barbiturate for anaesthesia — diethylbarbituric acid (1903, Fischer & von Mering) |
| First barbiturate used for induction | Hexobarbital (1927) |
| Thiopental first used clinically | 1934 — John Lundy & Ralph Waters |
| Thiopental concentration for induction | 2.5% solution |
| Methohexital concentration | 1% solution |
| Cannot mix thiopental with | Rocuronium, suxamethonium, midazolam, alfentanil, atracurium (precipitates) |
| Thiopental pKa | 7.6 |
| Thiopental protein binding | ~85% |
| Why avoid in porphyria | ↑ ALA synthetase → porphyrin crisis |
| Drug of choice for ECT | Methohexital |
| Intra-arterial injection treatment | Dilute + papaverine + sympathetic block + heparin |
| Thiopental after cardiac arrest? | NOT neuroprotective (global ischaemia) |
| Thiopental during aneurysm clipping? | YES neuroprotective (focal ischaemia) |
| Neonatal induction dose | 2–4 mg/kg IV |
| Rectal methohexital dose (paeds) | 25 mg/kg (10% solution) |
| Safe in bronchospasm? | NO — no bronchodilation, ↑ laryngospasm risk |
Examiner tip: The questions most commonly asked in MD viva on barbiturates are: (1) Why does thiopental have short action despite long half-life? (2) Why is it contraindicated in porphyria? (3) Intra-arterial injection management. (4) Why methohexital for ECT? (5) Mechanism of action vs benzodiazepines.
Generate a quick reference PDF cheat sheet on barbiturates
Reading File
Reading File
Reading File
Writing File
Running Command
Editing File
Running Command
Editing File
Running Command
Running Command
Your credits are exhausted. Your daily credits reset tomorrow, or you can purchase a top-up for more.
Generate a quick reference PDF cheat sheet on barbiturates
Running Command
Writing File
Running Command
Running Command
Your cheat sheet is ready!
Barbiturates CheatSheet
PDF Document · PDF
2-page A4 PDF with a dark navy + teal + amber theme, covering:
| Page | Sections |
|---|---|
| Page 1 | Chemistry & SAR · Classes & Incompatibilities · Mechanism of Action · Pharmacokinetics (table + key concepts) · CNS Effects (dose-response ladder) · CVS & Respiratory Effects |
| Page 2 | Clinical Uses & Dosing (full table) · Contraindications (absolute + relative) · Adverse Effects (inc. intra-arterial injection management steps) · Thiopental vs Methohexital vs Propofol comparison · 20-point Rapid Fire Viva panel |
CSF formation and circulation and anesthetic importance in MD Anaesthesia
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Finding Sources
Reading File
Finding Sources
Loading Image
I have all the content needed. Let me now compose a comprehensive, well-structured MD Anaesthesia-level answer.
CSF Formation, Circulation & Anaesthetic Importance
1. Introduction & Overview
Cerebrospinal fluid (CSF) is a clear, colourless, watery liquid that occupies the ventricular system of the brain and the subarachnoid space surrounding the brain and spinal cord. For the anaesthetist, a thorough understanding of CSF is indispensable because:
- It determines intracranial pressure (ICP)
- It is the medium through which intrathecal drugs are administered and distributed
- Its dynamics are directly altered by anaesthetic drugs, ventilation strategy, and patient positioning
- Its volume, along with blood and brain parenchyma, follows the Monro-Kellie doctrine
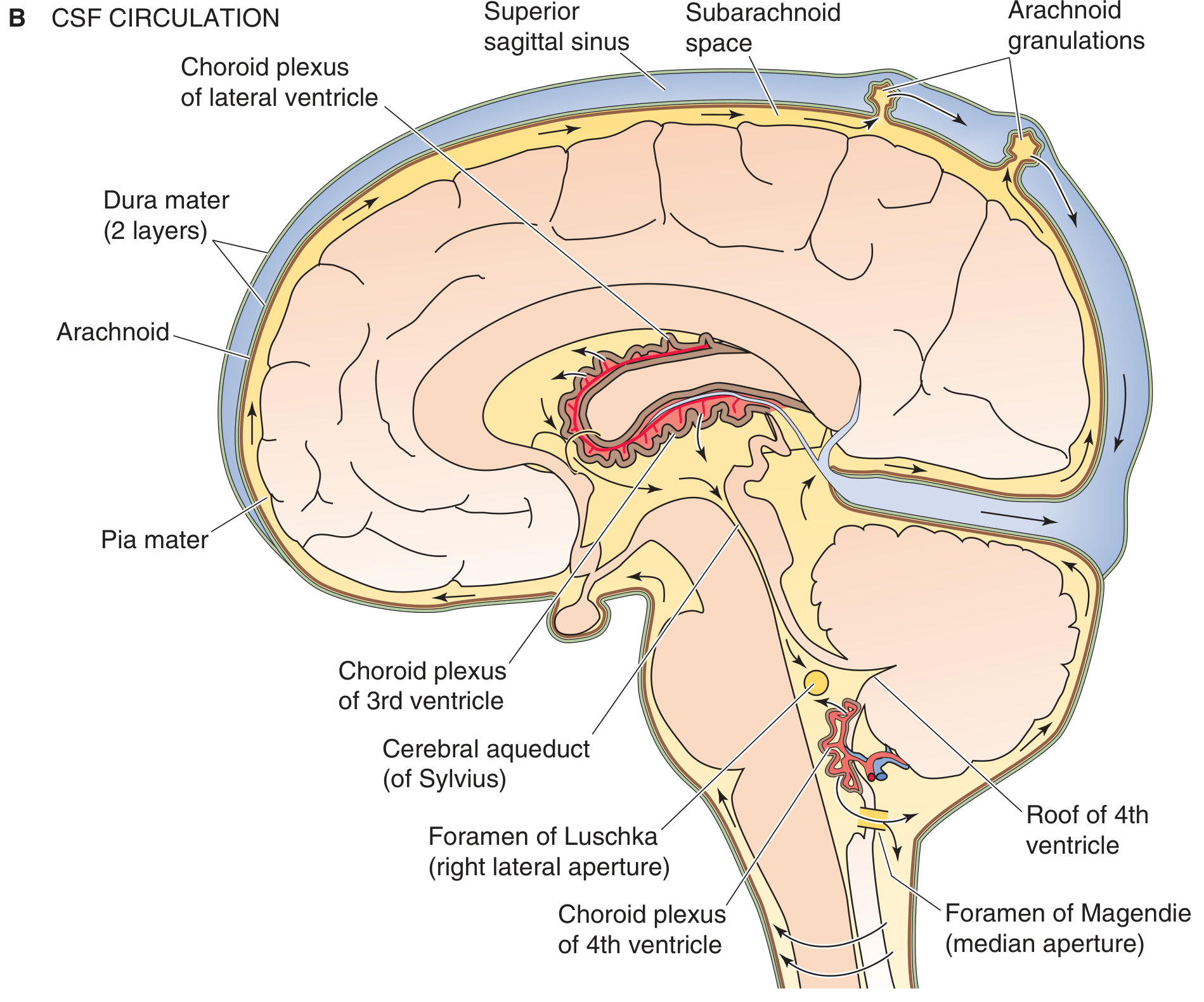
2. Anatomy of the CSF Compartments
Ventricles
There are four ventricles, each containing a choroid plexus:
| Ventricle | Location | Foramen |
|---|---|---|
| Two lateral ventricles | Within each cerebral hemisphere | Foramina of Monro → 3rd ventricle |
| Third ventricle | Diencephalon (between thalami) | Cerebral aqueduct of Sylvius → 4th |
| Fourth ventricle | Between pons/medulla and cerebellum | Foramina of Luschka (×2, lateral) and Foramen of Magendie (median) → subarachnoid space |
CSF flows from the fourth ventricle into the subarachnoid space via these three foramina. Distended regions of the subarachnoid space are called subarachnoid cisterns (cisterna magna, pontine cistern, interpeduncular cistern).
3. Formation of CSF
Site of Production
CSF is produced primarily by the choroid plexus in the lateral, third, and fourth ventricles. Small additional contributions come from:
- Cerebral endothelial cells
- Fluid produced as a by-product of neuronal metabolic activity
Rate of Production
- ~450–500 mL/day (some sources: 500 mL/day; Miller's: 450 mL/day)
- Rate of formation: ~0.35 mL/min (≈ 20 mL/hr)
- Total CSF volume: ~150 mL (intracranial ~100 mL + spinal ~50 mL)
- Therefore, CSF turns over approximately 3–4 times per day
Mechanism of Formation
CSF formation is an active secretory process, not simple ultrafiltration. Two phases:
- Hydrostatic efflux — fluid moves from choroidal capillaries into the perivascular space by hydrostatic pressure gradient
- Active transport — choroid plexus epithelial cells (resembling renal distal tubular cells) actively transport ions and water into the ventricles:
- Secreted into CSF: Na⁺, Cl⁻, HCO₃⁻, water
- Reabsorbed from CSF into blood: K⁺
- Proteins, cholesterol, and large molecules are excluded (large molecular size)
The Blood-CSF Barrier (Choroid Plexus Barrier)
Three layers:
- Capillary endothelial cells + basement membrane
- Neuroglial membrane
- Epithelial cells of the choroid plexus (joined by tight junctions)
Circadian Rhythm
CSF production is under circadian influence — peak production occurs during sleep. This is relevant because the glymphatic system (see below) is also most active during sleep and general anaesthesia.
4. CSF Composition
| Component | CSF vs Plasma |
|---|---|
| Na⁺ | Equal to plasma |
| Cl⁻ | Equal to plasma |
| HCO₃⁻ | Equal to plasma |
| Osmolarity | Equal to plasma |
| K⁺ | Lower than plasma |
| Ca²⁺ | Lower than plasma |
| Glucose | Lower than plasma (60–80% of plasma) |
| Amino acids | Lower than plasma |
| Protein | Negligible (~15–45 mg/dL; plasma 6000–8000 mg/dL) |
| Cholesterol | Negligible |
| Mg²⁺ | Higher than plasma |
| pH | Slightly lower than plasma (7.32–7.34) |
Normal CSF pressure: 70–180 mmH₂O (5–15 mmHg) in lateral decubitus.
5. Circulation of CSF
The CSF circulation follows a predictable pathway:
Choroid plexus (lateral ventricles)
↓ [Foramina of Monro]
Third ventricle
↓ [Cerebral aqueduct of Sylvius]
Fourth ventricle
↓ [Foramina of Luschka (×2) + Foramen of Magendie (×1)]
Subarachnoid space (around brain and spinal cord)
↓ [Convection/bulk flow upward over cerebral convexities]
Arachnoid granulations (in dural venous sinuses)
↓ [One-way bulk flow]
Superior sagittal sinus → Venous blood
Additional Drainage Routes (Minor)
- Along cranial and peripheral nerve sheaths
- Perivascular (perivenous) routes
- Along white matter tracts (transependymal flow)
- Meningeal and cervical lymphatic vessels
Driving Force for Circulation
CSF flows primarily by pulsatile bulk flow driven by:
- Choroidal arterial pulsations
- Respiratory pressure variations
- Cilia on ependymal cells lining the ventricles
6. The Glymphatic System — Anaesthetic Relevance
The glymphatic pathway (discovered ~2013) is a newly understood waste-clearance system of the brain:
Mechanism:
- CSF enters periarterial spaces (bounded by vessel walls and astrocyte end-feet)
- Aquaporin-4 channels on astrocyte end-feet facilitate water exchange
- CSF is transported by convection to brain parenchyma
- Accumulates in perivenous space → drains into cervical lymphatics and along cranial nerves
Clinical significance for anaesthetists:
-
The periarterial space increases significantly during sleep AND during general anaesthesia → waste clearance enhanced
-
Among anaesthetic agents:
- Volatile agents → reduce lymphatic/glymphatic transport
- Dexmedetomidine → less reduction; better preserves glymphatic transport
- This may be relevant to the pathogenesis of postoperative cognitive dysfunction (POCD)
-
Miller's Anesthesia, 10e, p. 930
7. Absorption of CSF
Primary Route: Arachnoid Granulations
- One-way valve-like projections into the dural venous sinuses (mainly superior sagittal sinus)
- Bulk flow from CSF to venous blood when CSF pressure > venous pressure
- Open at a threshold pressure of ~68 mmH₂O
Secondary Routes
- Perivenous and perineural pathways
- Spinal arachnoid granulations
- Nasal mucosal lymphatics (via cribriform plate)
8. Monro-Kellie Doctrine & ICP
The cranial vault is a rigid closed box. The three incompressible contents are:
- Brain parenchyma: ~80%
- Blood (cerebrovascular): ~12%
- CSF: ~8% (~100 mL intracranial)
Monro-Kellie Doctrine: The total intracranial volume is constant. An increase in one component must be compensated by a decrease in another.
Compensatory mechanisms for rising ICP:
- Displacement of CSF into spinal subarachnoid space (most readily displaced)
- Reduction in cerebral venous blood volume
- ↓ CSF production
- Once compensation is exhausted → exponential rise in ICP (decompensation)
Normal ICP: 5–15 mmHg (70–180 mmH₂O)
Cerebral Perfusion Pressure (CPP):
CPP = MAP − ICP (or MAP − CVP, whichever is higher)Target: CPP ≥ 60 mmHg (brain-injured patients: ≥ 70 mmHg)
9. Anaesthetic Importance of CSF — Comprehensive
9.1 Spinal (Subarachnoid) Anaesthesia
This is the most direct anaesthetic application of CSF knowledge:
Mechanism: Local anaesthetic injected into the lumbar cistern (L3-4 or L4-5) mixes with CSF and blocks nerve roots in the subarachnoid space.
Baricity and CSF density:
| Preparation | Density | Behaviour | Clinical use |
|---|---|---|---|
| Hyperbaric (heavy) | > CSF (denser) | Sinks with gravity | Positioned to desired level; most controllable |
| Isobaric | = CSF | Minimal positional spread | More predictable regardless of position |
| Hypobaric (light) | < CSF | Rises against gravity | Hip arthroplasty in lateral position |
CSF specific gravity: 1.003–1.008 at 37°CHyperbaric bupivacaine (0.5% + 8% glucose) = heavy bupivacaine, most commonly used
Factors affecting level of spinal block:
- Baricity — most important factor
- Dose (volume × concentration)
- Patient position at and immediately after injection
- Level of injection (L3-4 vs L4-5)
- Speed of injection
- CSF volume — reduced in pregnancy (epidural venous engorgement compresses subarachnoid space), obesity, elderly → unpredictable high block risk
PDPH (Post-Dural Puncture Headache):
- CSF leaks through dural puncture site → ↓ CSF volume and pressure → traction on pain-sensitive intracranial structures
- Positional: worse sitting/standing, relieved lying flat
- Prevented by: small-gauge pencil-point needles (Whitacre, Sprotte)
- Treated by: hydration, caffeine, epidural blood patch (gold standard)
9.2 Epidural Anaesthesia and CSF
- Epidural space is external to the dura — no direct CSF contact
- Accidental dural puncture (wet tap) → PDPH (especially with large Tuohy needle, 16–18G)
- Epidural blood patch: 20 mL autologous blood injected epidurally → seals CSF leak → clot increases epidural pressure temporarily and seals dural puncture → 85–90% success rate
- Total spinal: accidental intrathecal injection of epidural volume → high/total spinal block
9.3 Anaesthesia in Raised ICP
Anaesthetic goals in raised ICP:
- Maintain CPP ≥ 60 mmHg (MAP 80–100 + ICP control)
- Reduce ICP by:
- Reducing cerebral blood volume (CBV)
- Reducing CSF volume
- Reducing brain water/oedema
Effect of anaesthetic agents on ICP/CSF:
| Agent | Effect on ICP | Mechanism |
|---|---|---|
| Thiopental | ↓↓ ICP | ↓ CBF, ↓ CMRO₂, ↓ CBV |
| Propofol | ↓↓ ICP | ↓ CBF, ↓ CMRO₂, ↓ CBV |
| Ketamine | ↑ ICP | ↑ CBF, ↑ CMRO₂ — avoid in raised ICP |
| Etomidate | ↓ ICP | ↓ CBF, ↓ CMRO₂ |
| Midazolam/BZDs | ↓ mild | ↓ CMRO₂ |
| Volatile agents | ↑ ICP (dose-dep.) | Direct cerebral vasodilation → ↑ CBV |
| Isoflurane | Mild ↑ ICP; blunted by hyperventilation | |
| Sevoflurane | Mild ↑ ICP at >1 MAC | Preserves autoregulation best among volatiles |
| Desflurane | ↑ ICP slightly more than sevo/iso | Also ↑ HR and MAP via airway irritation |
| N₂O | ↑ ICP | ↑ CBF, ↑ CMRO₂ — avoid in neurosurgery |
CSF production and resorption:
- Volatile anaesthetics have modest, variable effects on CSF production and resorption — "clinically far less important than their effects on CBF" (Barash, 9e)
- The dominant mechanism of ICP change with volatiles is through changes in CBV (cerebral blood flow), not through direct effects on CSF
9.4 Hyperventilation and ICP
PaCO₂ is the most potent acute regulator of CBF and ICP:
- ↓ PaCO₂ → cerebral vasoconstriction → ↓ CBV → ↓ ICP
- Each 1 mmHg ↓ PaCO₂ → ↓ CBF by ~3%
- Target PaCO₂: 35 mmHg (routine) or 30–35 mmHg (acute ICP crisis)
- Prophylactic hyperventilation to PaCO₂ <30 mmHg is not recommended (cerebral ischaemia risk)
- Effect is temporary — CSF pH normalizes within 6–12 hours (bicarbonate compensates)
9.5 Drugs That Reduce CSF Production
| Drug | Mechanism | Clinical use |
|---|---|---|
| Acetazolamide | Inhibits carbonic anhydrase in choroid plexus → ↓ HCO₃⁻ secretion → ↓ CSF production by ~50% | Raised ICP, idiopathic intracranial hypertension |
| Furosemide | Inhibits Cl⁻ transport in choroid plexus | Adjunct in ICP management |
| Mannitol | Osmotic → ↓ brain water; also ↓ blood viscosity → ↓ CBF reflexly | Acute ICP management; 0.25–1 g/kg IV |
| Hypertonic saline | Osmotic; ↑ serum osmolarity → draws water from brain | 3–23.4% saline for ICP |
| Corticosteroids | ↓ permeability; effective for vasogenic oedema (tumours) | Preoperative brain tumour, post-irradiation |
| Barbiturates | Burst suppression → ↓ CMRO₂ → ↓ CBF → ↓ CBV → ↓ ICP | Barbiturate coma (refractory ICP) |
9.6 CSF Drainage as a Neurosurgical Adjunct
- Lumbar CSF drain or ventricular drain (EVD): deliberate removal of CSF during surgery to reduce ICP and improve surgical access to deep structures
- External Ventricular Drain (EVD): catheter in lateral ventricle → continuous ICP monitoring + CSF drainage
- Used in: subarachnoid haemorrhage (SAH), TBI, post-craniotomy ICP management, hydrocephalus
- Anaesthetic relevance: sudden rapid CSF drainage → risk of brain herniation (especially with posterior fossa tumours and aqueductal obstruction)
9.7 Lumbar Puncture — Anaesthetic Considerations
Diagnostic LP:
- Performed at L3-4 or L4-5 (below conus medullaris, which ends at L1-2 in adults)
- Patient position: lateral decubitus (knees to chest) or sitting flexed
- Normal opening pressure: 70–180 mmH₂O
- Contraindications to LP:
- Raised ICP with papilloedema or mass lesion (risk of transtentorial herniation)
- Local infection at puncture site
- Coagulopathy (INR >1.5, platelets <50,000, therapeutic anticoagulation)
- Anticoagulant therapy — timing per regional anaesthesia guidelines (ASRA/ESRA)
CSF examination in anaesthetic context:
- Blood-tinged CSF: traumatic tap (clears with successive samples) vs SAH (xanthochromia)
- CSF glucose: always compare with blood glucose — ratio <0.5 = bacterial meningitis
9.8 CSF and Neuraxial Drug Spread
Intrathecal opioids:
- Lipophilic (fentanyl, sufentanil): rapid uptake into cord → segmental analgesia, minimal rostral spread, shorter duration
- Hydrophilic (morphine, diamorphine): slow uptake → rostral spread in CSF → prolonged analgesia + risk of delayed respiratory depression (up to 18–24 hours with morphine)
- Requires 24-hour respiratory monitoring after intrathecal morphine
Intrathecal adjuvants: clonidine, dexmedetomidine, neostigmine — all act via CSF distribution
9.9 Position and CSF Pressure
| Position | Effect on ICP |
|---|---|
| Head-up 30° | ↓ ICP (facilitates CSF and venous drainage) — standard for raised ICP |
| Head-down (Trendelenburg) | ↑ ICP — use cautiously in neuro patients |
| Head rotation/flexion | Can obstruct jugular venous drainage → ↑ ICP |
| Lateral decubitus | Preferred for LP (opens intervertebral spaces) |
| Sitting (beach chair) | ↓ ICP but risk of venous air embolism in posterior fossa surgery |
9.10 The Glymphatic System and POCD
Of emerging anaesthetic relevance:
- General anaesthesia increases glymphatic transport (periarterial space enlarges)
- Volatile agents reduce glymphatic function relative to sleep
- Dexmedetomidine (which produces sleep-like state) better preserves glymphatic flow
- Accumulation of amyloid-β and tau (AD pathology markers) may relate to impaired glymphatic clearance
- This underlies ongoing research linking choice of anaesthetic agent to long-term cognitive outcomes
10. Summary Table — Anaesthetic Effects on CSF/ICP
| Intervention | ICP Effect | Mechanism |
|---|---|---|
| Propofol | ↓↓ | ↓ CMRO₂ → ↓ CBF → ↓ CBV |
| Thiopental | ↓↓ | ↓ CMRO₂ → ↓ CBF → ↓ CBV; burst suppression |
| Ketamine | ↑↑ | ↑ CMRO₂, ↑ CBF, ↑ CBV — avoid in raised ICP |
| Volatile (>1 MAC) | ↑ (mild) | Direct vasodilation → ↑ CBV |
| Sevoflurane ≤1 MAC | Minimal | Preserves autoregulation best |
| Desflurane | ↑ (most of volatiles) | Greatest vasodilation + airway reflex ↑ MAP |
| N₂O | ↑ | ↑ CBF, ↑ CMRO₂ |
| Hyperventilation (PaCO₂ ↓) | ↓ | Cerebral vasoconstriction |
| Hypoventilation (PaCO₂ ↑) | ↑↑ | Cerebral vasodilation |
| Mannitol | ↓ | Osmotic dehydration of brain |
| Head-up 30° | ↓ | Facilitates venous + CSF drainage |
| CSF drainage (EVD/LP) | ↓ | Direct volume removal |
| Acetazolamide | ↓ | ↓ CSF production via CA inhibition |
| Coughing/straining | ↑↑ | ↑ intrathoracic pressure → ↑ CBV |
11. Key Viva Points
| Q | A |
|---|---|
| Rate of CSF production | ~450–500 mL/day (0.35 mL/min) |
| Total CSF volume | ~150 mL |
| Turnover rate | 3–4 times per day |
| Site of production | Choroid plexus (lateral, 3rd, 4th ventricles) |
| Site of absorption | Arachnoid granulations in superior sagittal sinus |
| Foramen from 4th ventricle | Luschka (×2, lateral) + Magendie (×1, median) |
| Normal ICP | 5–15 mmHg (70–180 mmH₂O) |
| CPP formula | MAP − ICP |
| Target CPP | ≥60 mmHg (≥70 in TBI) |
| Most potent regulator of CBF | PaCO₂ |
| Drug that best reduces CSF production | Acetazolamide (CA inhibitor) |
| Anaesthetic that raises ICP most | Ketamine; N₂O |
| Safest volatile for neuro | Sevoflurane ≤1 MAC |
| Why avoid ketamine in ICP? | ↑ CMRO₂ + ↑ CBF + ↑ CBV |
| Monro-Kellie doctrine | Brain + blood + CSF = constant intracranial volume |
| Baricity importance in spinal | Hyperbaric sinks; hypobaric floats; position determines spread |
| PDPH mechanism | CSF leak → ↓ CSF pressure → traction on intracranial structures |
| Treatment of PDPH | Epidural blood patch (gold standard) |
| Intrathecal morphine risk | Delayed respiratory depression (up to 18–24 h) |
| Glymphatic and dexmedetomidine | Dex better preserves glymphatic transport than volatiles |
Sources: Miller's Anesthesia 10e | Barash Clinical Anesthesia 9e | Costanzo Physiology 7e | Boron & Boulpaep Medical Physiology
Make a ppt and cheat sheet
Writing File
Writing File
!
Failed to create session: unauthorized: authentication failed: Bearer token is invalid
We encountered an error while processing your request. Please try again or reach out to us for help.
Make a ppt
Running Command
Writing File
Running Command
Running Command
Your presentation is ready!
CSF Anaesthesia
Presentation · PPTX
11 slides — deep navy + teal + amber color scheme:
| Slide | Content |
|---|---|
| 1 | Title slide |
| 2 | Outline |
| 3 | Anatomy of CSF Compartments (ventricular pathway + spinal landmarks) |
| 4 | Formation of CSF (2 phases, barrier, rate/volume, circadian) |
| 5 | CSF Composition Table + Circulation pathway diagram |
| 6 | Monro-Kellie Doctrine, 3 components, CPP formula, compensatory mechanisms |
| 7 | Anaesthetic effects on ICP/CSF — full comparison table (10 agents) |
| 8 | Spinal anaesthesia — baricity, block factors, PDPH, intrathecal opioids |
| 9 | ICP management — ventilation/positioning, pharmacology, CSF drainage |
| 10 | Glymphatic system — mechanism + anaesthetic relevance |
| 11 | Viva summary — 6 category rapid-fire reference |
Test and types of autoregulation in CNS in Anesthesia
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Finding Sources
Loading Image
Reading File
Reading File
Reading File
I now have comprehensive content. Let me compose the full MD Anaesthesia-level answer.
Autoregulation in the CNS — Types, Tests & Anaesthetic Importance
1. What is Cerebral Autoregulation?
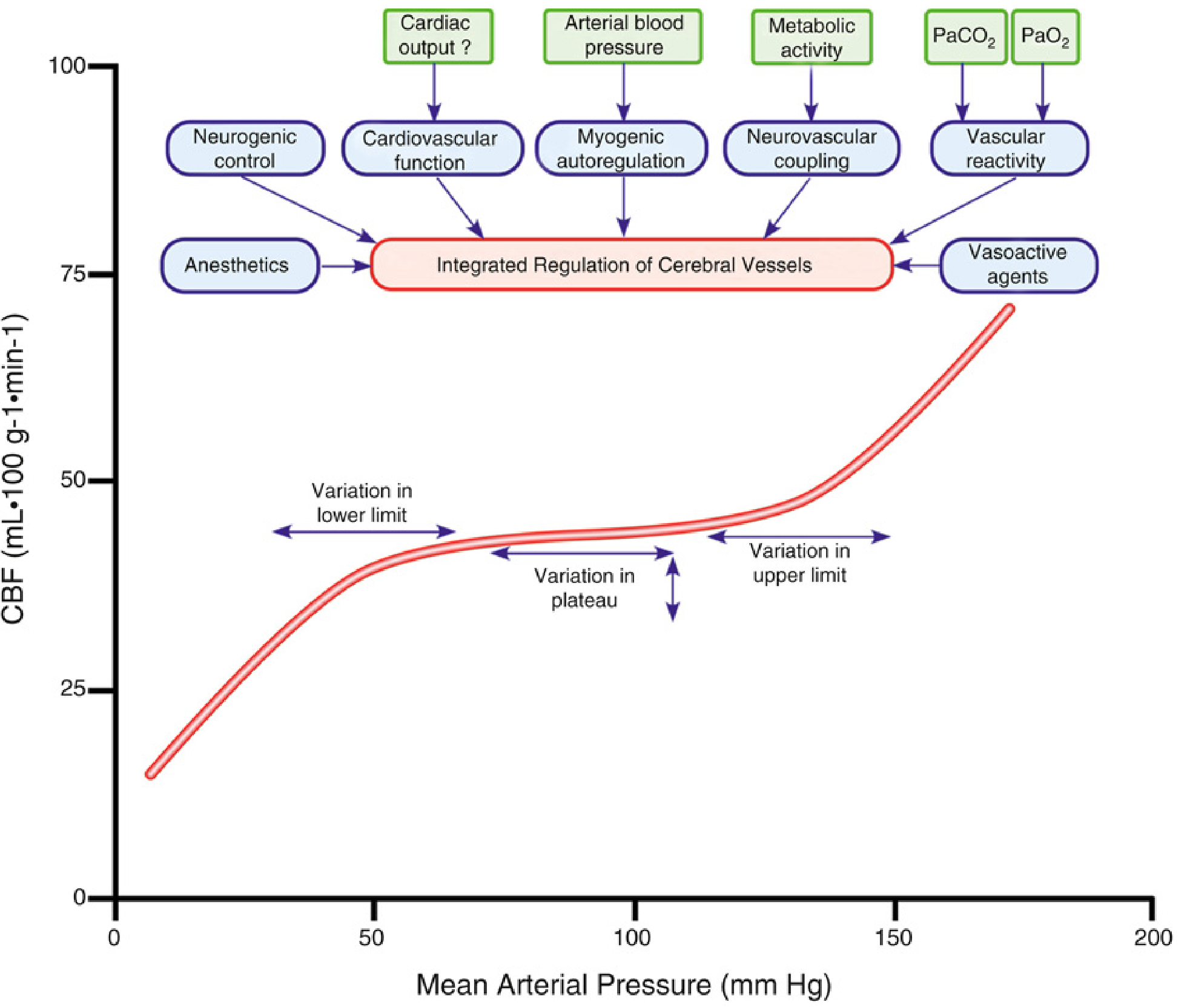
Autoregulation refers to the intrinsic capacity of the cerebral circulation to maintain a relatively constant cerebral blood flow (CBF) over a wide range of mean arterial pressure (MAP) values, despite changes in perfusion pressure.
The concept originates from the classic review by Lassen (1959), which described CBF being held constant at approximately 50 mL/100 g/min over a MAP range of 50–150 mmHg.
However, the current view in Miller's Anesthesia (10e) emphasizes that the "static, flat plateau" of Lassen is outdated. Autoregulation is now understood as a dynamic, integrative process influenced by multiple interdependent variables — not a simple pressure-passive or pressure-independent switch.
2. Regulation of Cerebral Blood Flow — Overview
Normal CBF: 50 mL/100 g/min (grey matter ~80 mL/100 g/min; white matter ~20 mL/100 g/min)
The mechanisms regulating CBF include:
- Myogenic (pressure autoregulation)
- Metabolic / Chemical (CO₂, O₂, pH)
- Neurogenic
- Neurovascular coupling (flow-metabolism coupling)
- Endothelial (NO, prostaglandins, endothelin)
These are described in Miller's Table 10.1 as chemical, myogenic, and neurogenic factors — and they act in an interdependent, integrated manner.
3. Types of Autoregulation
3.1 Pressure Autoregulation (Myogenic Autoregulation)
Mechanism:
- The Bayliss myogenic response — smooth muscle in cerebrovascular walls responds to transmural pressure changes
- ↑ perfusion pressure → active vasoconstriction (prevents overperfusion)
- ↓ perfusion pressure → active vasodilation (prevents underperfusion)
- Operates predominantly in pial arteries and arterioles
Classic Lassen Curve:
| Pressure Range | CBF Behaviour |
|---|---|
| MAP < 50 mmHg (below LLA) | CBF falls (pressure-passive) |
| MAP 50–150 mmHg (plateau) | CBF maintained ~50 mL/100 g/min |
| MAP > 150 mmHg (above ULA) | CBF rises (breakthrough, forced dilatation) |
Modern revision of Lassen:
- The plateau is not flat — has a gentle upslope
- The range of pressure-passivity is wider than classically believed
- Lower and upper limits vary considerably between individuals
- Measured autoregulatory curves show variation in lower limit, plateau, and upper limit
Static vs Dynamic autoregulation:
| Parameter | Static Autoregulation | Dynamic Autoregulation |
|---|---|---|
| Time frame | Minutes (~10 min) | Seconds to minutes |
| BP change | Slow, sustained | Rapid, transient |
| Method | Pharmacologic MAP manipulation | Thigh cuff deflation → rapid MAP drop |
| Measurement | CBF at steady state | MCAfv (TCD) response |
| Plateau | Wider, flat-looking | Narrower, more slope |
| Clinical use | Pharmacologic testing | Bedside assessment (PRx, COx) |
Static autoregulation allows the measured variables to achieve steady state; dynamic autoregulation uses rapid, transient MAP reductions (e.g., thigh cuff deflation) to assess the buffering capacity of the cerebral circulation.
3.2 Metabolic / Chemical Autoregulation
3.2.1 CO₂ Reactivity (PaCO₂)
The most powerful acute regulator of CBF. CBF varies linearly with PaCO₂ in the range of 20–80 mmHg.
Mechanism:
- CO₂ freely diffuses across the blood-brain barrier
- Alters extracellular pH in the perivascular space
- H⁺-mediated NO release, adenosine, arachidonic acid metabolites, reactive oxygen species → vasodilation
- Acidosis (↑CO₂) → vasodilation → ↑CBF
- Alkalosis (↓CO₂) → vasoconstriction → ↓CBF
Quantitative:
- Each 1 mmHg ↑ PaCO₂ → ↑ CBF ~3% (linear response ~20–80 mmHg)
- Below PaCO₂ ~25 mmHg: further CBF reduction is limited (vasospasm limit)
- Above PaCO₂ ~75–80 mmHg: response is attenuated
Effect on autoregulation:
- Hypercarbia → cerebral vasodilation → attenuates autoregulatory response to hypertension → autoregulation becomes pressure-passive at lower MAP
- Hypocapnia → vasoconstriction → autoregulation maintained over wider MAP range
Duration:
- Effect is not sustained — despite maintained arterial pH change, CBF returns toward normal in 6–8 hours as bicarbonate is extruded from CSF (pH normalizes)
Clinical consequence (rebound):
- After prolonged hyperventilation: acute return to normal PaCO₂ → CSF acidosis → ↑CBF → ↑ICP
- After prolonged hypoventilation: acute normalization → CSF alkalosis → risk of ischaemia
Relationship to MAP:
- Moderate hypotension (MAP ↓ <33%): CO₂ responsiveness is significantly attenuated
- Severe hypotension (MAP ↓ ~66%): CO₂ responsiveness is abolished
3.2.2 Oxygen (PaO₂)
- PaO₂ 60–300 mmHg: little influence on CBF
- PaO₂ < 60 mmHg → rapid ↑ CBF (critical threshold corresponding to fall in SpO₂)
- Relationship between CBF and haemoglobin saturation is inversely linear
- Mechanism: hypoxia → local adenosine release, ↓pH, NO release → vasodilation
- Haematic hypoxia (e.g., anaemia): reduction in oxygen content with normal PaO₂ also triggers compensatory ↑CBF
Anaemia and CBF:
- Haematocrit ↓ → ↓ viscosity + ↓ O₂ content → ↑CBF
- At low haematocrit, the benefit of ↓ viscosity is outweighed by ↓O₂ carrying capacity
- Optimal haematocrit for cerebral O₂ delivery: ~30–35% (balance of viscosity and O₂ content)
3.2.3 Metabolic/Flow-Metabolism Coupling (Neurovascular Coupling)
Principle: CBF is tightly coupled to local neuronal metabolic activity (CMRO₂). An area of increased neural activity receives proportionally increased flow.
Mechanism:
- Neuronal activity → release of K⁺, H⁺, adenosine, CO₂, arachidonic acid metabolites → perivascular vasodilation
- Astrocytes (glial cells) act as intermediaries → release vasoactive substances (NO, arachidonic acid derivatives)
- Potassium siphoning by astrocytes also contributes
Anaesthetic relevance:
- Volatile agents cause uncoupling — they increase CBF despite decreasing CMRO₂ (direct vasodilation exceeds metabolic suppression)
- IV agents (propofol, thiopental) maintain or improve coupling — they reduce CMRO₂ and CBF in parallel
Dose-response with barbiturates:
- Progressive reduction in CMRO₂ and CBF with increasing barbiturate dose
- Maximum effect at isoelectric EEG (electrophysiologic silence)
- Beyond this point, no further reduction — only basal metabolic activity (cellular homeostasis) persists
- Additional barbiturate causes no further CMRO₂ or CBF reduction
3.3 Neurogenic Autoregulation
Innervation:
- Cerebral vessels receive sympathetic, parasympathetic, and sensory innervation
- Sympathetic: from superior cervical ganglia → travels with cerebral arteries → supplies large + penetrating arteries
- Parasympathetic: from pterygopalatine ganglion → cholinergic vasodilator input
- Sensory: trigeminal nerve → peptide-mediated (CGRP, substance P)
Normally limited role:
- Mild-to-moderate sympathetic stimulation causes little change in CBF — myogenic and metabolic mechanisms override
- Sympathetic influence becomes important in protecting against acute severe hypertension — prevents high pressure from reaching small vessels and causing haemorrhage (stroke prevention)
Clinical importance:
- Medications affecting sympathetic tone (β-agonists, α₂-agonists, ACE inhibitors, ARBs, calcium channel blockers) all modulate autoregulation
- Dexmedetomidine (α₂-agonist): reduces sympathetic outflow → affects cerebrovascular tone
3.4 Endothelial Autoregulation
The vascular endothelium produces and responds to vasoactive mediators:
| Mediator | Effect | Stimulus |
|---|---|---|
| Nitric oxide (NO) | Vasodilation | Shear stress, acetylcholine, hypoxia, CO₂ |
| Prostacyclin (PGI₂) | Vasodilation | Shear stress |
| Endothelin-1 | Vasoconstriction | Stretch, angiotensin II, thrombin |
| Thromboxane A₂ | Vasoconstriction | Platelet activation |
| Adenosine | Vasodilation | Metabolic demand, hypoxia |
4. Static vs Dynamic Autoregulation — Detailed Comparison
| Feature | Static | Dynamic |
|---|---|---|
| Definition | CBF maintained constant with slow MAP changes | CBF buffering with rapid transient MAP changes |
| Time frame | ~10 min per BP step | Seconds to ~2 min |
| BP change method | Pharmacologic (phenylephrine, nitroprusside, tilt) | Thigh cuff deflation, sit-to-stand, Valsalva |
| Measurement | CBF (Xe-133, PET, MRI, TCD) | Middle cerebral artery flow velocity (TCD) |
| Outcome measure | Autoregulatory index (plateau slope) | Phase shift, gain, transfer function, PRx |
| Plateau width | ~50–150 mmHg (Lassen); ~70–150 mmHg (modern) | Narrower (rapid changes less buffered) |
| Clinical use | Pharmacologic BP management | Bedside continuous monitoring (ICP/BP) |
| Rate-dependence | Minimal | Critical — faster changes = less buffering |
5. Tests of Cerebral Autoregulation
5.1 Transcranial Doppler (TCD) — Most Used Clinically
Measures: Middle cerebral artery flow velocity (MCAfv) as a surrogate for CBF
Tests:
| Test | Method | Measure |
|---|---|---|
| Thigh cuff test | Rapid cuff deflation → transient ↓MAP | MCAfv recovery; ARI (Autoregulatory Index) |
| Transient hyperaemic response test (THRT) | Brief carotid compression → release | Hyperaemic overshoot = intact autoregulation |
| Tilt-table / head-up tilt | Postural BP change | MCAfv vs MAP phase relationship |
| Transfer function analysis | Spontaneous BP oscillations (0.07–0.2 Hz) | Gain and phase between MAP and MCAfv |
Autoregulatory Index (ARI):
- Scale of 0–9 (Tiecks et al.)
- 0 = complete pressure-passivity (no autoregulation)
- 9 = perfect autoregulation
- ARI ≥ 4 considered normal; ARI < 4 = impaired
5.2 Pressure Reactivity Index (PRx) — ICP-Based
Used in: ICU/neurocritical care, TBI monitoring
Method:
- Continuous simultaneous recording of ABP and ICP
- Calculate rolling correlation coefficient between MAP and ICP over 5-minute windows
- PRx = Pearson correlation coefficient (MAP vs ICP)
Interpretation:
| PRx value | Meaning |
|---|---|
| Negative (−1 to 0) | Active autoregulation — ICP inversely reactive to MAP |
| Near zero | No relationship — intermediate |
| Positive (+0.3 to +1) | Impaired autoregulation — pressure-passive CBF |
Optimal CPP (CPPopt):
- U-shaped relationship between PRx and CPP
- The MAP at the nadir (lowest PRx) = optimal CPP where autoregulation is most effective
- Managing TBI patients to CPPopt is associated with better outcomes
5.3 Cerebral Oximetry Index (COx / TOx)
Method:
- Correlation between MAP and regional cerebral oxygen saturation (rSO₂) measured by Near-Infrared Spectroscopy (NIRS)
- COx = correlation coefficient between MAP and rSO₂
Interpretation:
- COx near 0 or negative = intact autoregulation (rSO₂ buffered from MAP changes)
- COx > 0.3–0.4 = impaired autoregulation
- Used in cardiac surgery, TBI — can derive optimal MAP from U-shaped COx-MAP curve
5.4 Laser Doppler Flowmetry
- Continuous cortical CBF measurement
- Invasive (intraoperative) or perioperative
- Provides real-time CBF data for autoregulation assessment
- Used in experimental and intraoperative neurosurgical settings
5.5 Xe-133 Clearance / PET / MRI
- Gold standard measurement of absolute CBF
- Used in research and select clinical settings
- Static autoregulation curves derived from multiple BP levels
5.6 Near-Infrared Spectroscopy (NIRS) — Non-Invasive Bedside
Hemoglobin Reactivity Index (HVRx or HbD reactivity index):
- Non-invasive; measures oxygenated vs deoxygenated Hb changes with BP fluctuations
- Used in neonates, cardiac surgery, TBI
- Optimal MAP for cardiac surgery derived from NIRS-based autoregulation monitoring
6. Conditions Affecting Autoregulation
6.1 Physiological Shifts
| Condition | Effect on Autoregulation |
|---|---|
| Hypercarbia (↑PaCO₂) | Attenuates; narrows plateau; LLA rises |
| Hypocapnia (↓PaCO₂) | Enhances; widens plateau |
| Hypoxia (PaO₂ <60 mmHg) | Impairs |
| Chronic hypertension | Shifts entire curve to RIGHT (higher MAP range) |
| Acute severe hypotension | CO₂ reactivity abolished |
6.2 Disease States
| Condition | Effect |
|---|---|
| Traumatic brain injury (TBI) | Commonly impaired; pressure-passive CBF |
| Subarachnoid haemorrhage | Impaired especially in vasospasm period |
| Stroke / acute ischaemia | Abolished in penumbral tissue |
| Preeclampsia | Impaired → pressure-dependent CBF → oedema, seizures |
| Sepsis | Impaired in severe cases |
| Prematurity (neonates) | Poorly developed; pressure-passive |
| Chronic hypertension | Shifted right; vulnerable to rapid BP reduction |
| Atherosclerosis, old age | Impaired |
| Hypoglycaemia | Can impair |
7. Anaesthetic Agents and Autoregulation
7.1 Intravenous Agents
| Agent | Effect on Autoregulation | Mechanism |
|---|---|---|
| Propofol | PRESERVED | ↓ CMRO₂ → ↓ CBF proportionally; vasoconstriction coupled to metabolism |
| Thiopental | PRESERVED (at anaesthetic doses) | ↓ CMRO₂ → ↓ CBF; cerebrovascular tone maintained |
| Etomidate | PRESERVED | ↓ CMRO₂ → ↓ CBF |
| Ketamine | IMPAIRED | ↑ CMRO₂ → ↑ CBF; direct vasodilation; uncoupling |
| Midazolam | Largely PRESERVED | Mild CMRO₂ reduction |
| Dexmedetomidine | PRESERVED | α₂ agonist; coupled reduction in CBF and CMRO₂; reduces sympathetic tone |
| Opioids | PRESERVED | Minimal direct effect; ↓ CMRO₂ |
7.2 Volatile Agents
All volatile agents impair autoregulation in a dose-dependent manner:
- Mechanism: Direct cerebral vasodilation → pressure-passive CBF at high doses
- Net effect = sum of indirect vasoconstriction (via ↓CMRO₂) and direct vasodilation
| Agent | Autoregulation | Vasodilation potency | Notes |
|---|---|---|---|
| Sevoflurane | Best preserved (~1 MAC) | Least | Preserves up to ~1 MAC; dynamic ARi better than isoflurane at 1.5 MAC |
| Isoflurane | Impaired dose-dependently | Moderate | Hyperventilation blunts increase in ICP |
| Desflurane | Impaired | Most | Airway irritation → ↑MAP → ↑ICP; avoid in raised ICP |
| Halothane | Most impaired | Maximum | Vasodilates even at low concentrations (0.5 MAC); no longer used |
| N₂O | Impaired | Moderate | ↑ CMRO₂ and CBF; avoid in raised ICP |
At high anaesthetic doses (>1.5–2 MAC), CBF becomes essentially pressure-passive with all volatile agents.
8. The Flow-Metabolism Uncoupling with Volatiles
- Volatile agents decrease CMRO₂ but simultaneously cause direct vasodilation
- This is called "functional uncoupling" — CBF ↑ or → unchanged despite CMRO₂ ↓
- From a mechanistic standpoint, true uncoupling may not occur: there is still a coupled CMRO₂-driven vasoconstriction, opposed by a direct vasodilatory effect
- Net result depends on which effect dominates at a given MAC
9. Factors Modulating Autoregulation — Summary
| Factor | Effect |
|---|---|
| ↑ PaCO₂ | Impairs (vasodilation → pressure-passive) |
| ↓ PaCO₂ | Enhances (widens autoregulatory range) |
| Hypoxia (PaO₂ <60) | Impairs |
| Severe hypotension | Abolishes CO₂ reactivity |
| Volatile anaesthetics | Dose-dependent impairment |
| IV agents (propofol, thiopental) | Preserved or enhanced |
| Sympathomimetics | Modulate (α₁ agents: ↑ SVR, may ↓ CO) |
| Nitrates, Ca²⁺ channel blockers | Impair (vasodilators) |
| ACE inhibitors, ARBs | Modulate |
| β-agonists | Modulate via CO effect |
| Chronic hypertension | Rightward shift of curve |
| TBI, stroke, SAH | Impaired/abolished |
| Age, atherosclerosis | Impaired |
10. Clinical Applications of Autoregulation in Anaesthesia
10.1 Blood Pressure Management Intraoperatively
- Within autoregulatory range: BP changes have minimal CBF effect
- Outside autoregulatory range (pressure-passive): every MAP change directly changes CBF and ICP
- Vasopressor choice matters:
- Phenylephrine (α₁-agonist): ↑ SVR → may ↓ CO → compromised CBF (especially with bolus)
- Ephedrine (α+β): maintains or ↑ CO + MAP → better CBF maintenance
- After volume optimization, prefer agents that maintain/increase both CO and MAP
10.2 Anaesthetic Agent Selection for Neurosurgery
- Prefer TIVA (propofol + opioid) for raised ICP → preserved autoregulation, ↓ CBV
- If volatile required: sevoflurane ≤ 1 MAC + normocapnia → autoregulation maintained
- Avoid desflurane in patients with raised ICP
- Ketamine: avoid in raised ICP (uncoupling, ↑ CBF, ↑ CMRO₂)
10.3 Targeted MAP Management — CPPopt
- In TBI patients: use PRx monitoring to identify CPPopt
- Manage MAP to keep CPP within optimal range where PRx is most negative
- Prevents both: ischaemia (CPP too low) and hyperaemia/oedema (CPP too high)
10.4 CO₂ Management
- Normocapnia (PaCO₂ 35–40 mmHg): standard
- Hyperventilation (PaCO₂ 30–35 mmHg): for acute ICP crisis only — bridge therapy
- PaCO₂ < 30 mmHg: risk of ischaemia (vasoconstriction beyond safety threshold)
- Chronic hyperventilation not sustained — pH normalizes in 6–8 hours
10.5 Cardiac Surgery
- Prolonged CPB → potential impairment of autoregulation
- NIRS-based autoregulation monitoring (COx/HVRx) allows determination of individual optimal MAP
- Above the ULA: risk of cerebral hyperaemia, oedema, intracerebral haemorrhage
- Below the LLA: risk of ischaemia, cognitive dysfunction, stroke
11. Key Viva Questions
| Q | A |
|---|---|
| Define autoregulation | Intrinsic capacity of cerebral circulation to maintain constant CBF over MAP 50–150 mmHg |
| Classic LLA and ULA | LLA ~50 mmHg, ULA ~150 mmHg (Lassen); modern view: variation exists |
| Normal CBF | ~50 mL/100 g/min |
| Types of autoregulation | Pressure (myogenic), metabolic (CO₂, O₂, pH), neurogenic, endothelial, neurovascular coupling |
| Static vs dynamic autoregulation | Static = slow BP changes over ~10 min; Dynamic = rapid transient BP changes (thigh cuff) |
| Best test of autoregulation at bedside | PRx (ICP-based) or COx/NIRS (non-invasive) |
| PRx > +0.3 means? | Impaired autoregulation — pressure-passive CBF |
| CPPopt definition | MAP at nadir of U-shaped PRx curve — where autoregulation most effective |
| Each 1 mmHg ↑ PaCO₂ | ↑ CBF ~3% |
| Why does CBF not decrease below PaCO₂ 25 mmHg? | Further vasoconstriction limited — ischaemia threshold reached |
| Effect of hypercarbia on autoregulation | Attenuates; narrows autoregulatory plateau; LLA rises |
| Effect of hypocapnia | Enhances; widens autoregulatory range |
| Why CO₂ reactivity is abolished in severe hypotension | MAP reduction ~66% → cerebral vasculature already maximally dilated |
| Best volatile for neuroanaesthesia | Sevoflurane ≤ 1 MAC — best preserves autoregulation |
| Why ketamine contraindicated in raised ICP | Uncouples flow-metabolism → ↑ CBF + ↑ CMRO₂ + direct vasodilation |
| Shift of autoregulation curve in hypertension | Rightward — higher MAP range needed for adequate perfusion; rapid BP reduction risks ischaemia |
| ARI scale | 0–9: ARI 0 = no autoregulation; ARI 9 = perfect |
| Bayliss response | Myogenic response — vessel wall contracts in response to increased transmural pressure |
Sources: Miller's Anesthesia 10e (Chapter 10) | Barash Clinical Anesthesia 9e | Guyton & Hall Medical Physiology | Boron & Boulpaep Medical Physiology
Make a ppt
Running Command
Writing File
Running Command
Running Command
Your presentation is ready!
CNS Autoregulation
Presentation · PPTX
11 slides — deep navy + teal/green/amber accent scheme:
| Slide | Content |
|---|---|
| 1 | Title slide |
| 2 | Outline |
| 3 | Definition & Classical Lassen Curve (with graph + modern revision) |
| 4 | 6 Types of Autoregulation (myogenic, metabolic, neurogenic, neurovascular, endothelial, flow-metabolism coupling) |
| 5 | CO₂ & O₂ Reactivity (detailed tables, anaemia, CO₂-AR interaction) |
| 6 | Static vs Dynamic Autoregulation (comparison table) |
| 7 | Tests — TCD/ARI, PRx, NIRS/COx (3-column detail) |
| 8 | Anaesthetic Agents — IV agents + Volatile agents comparison tables |
| 9 | Conditions affecting autoregulation (physiological + diseases + hypertension shift) |
| 10 | Clinical Applications (BP management, neuroanaesthesia, CPPopt/TBI) |
| 11 | Viva Summary — 6-category rapid-fire reference |
This is a shared conversation. Sign in to Orris to start your own chat.