Stomach Carcinoma gross n microscopic features diagram 2nd yr mbbs refer robins
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
Loading Image
Reading File
Loading Image
I now have all the material needed for a comprehensive answer. Here is the full Robbins-based breakdown:
Stomach Carcinoma — Gross & Microscopic Features
Robbins & Kumar Basic Pathology + Robbins, Cotran & Kumar Pathologic Basis of Disease
Overview
Adenocarcinoma is the most common malignancy of the stomach, comprising >90% of all gastric cancers. Classified into two major morphological types — intestinal-type and diffuse-type — which differ in gross appearance, histology, molecular pathogenesis, and prognosis.
Most distal tumors arise in the gastric antrum; the lesser curvature is involved more often than the greater curvature.
GROSS FEATURES
1. Intestinal-Type Adenocarcinoma
- Forms a bulky, exophytic mass with heaped-up, raised borders
- Central ulceration common — can mimic a benign peptic ulcer, but distinguished by irregular, raised, overhanging edges (versus the smooth, punched-out edges of peptic ulcer)
- Grows along broad cohesive fronts
- May be polypoid or fungating
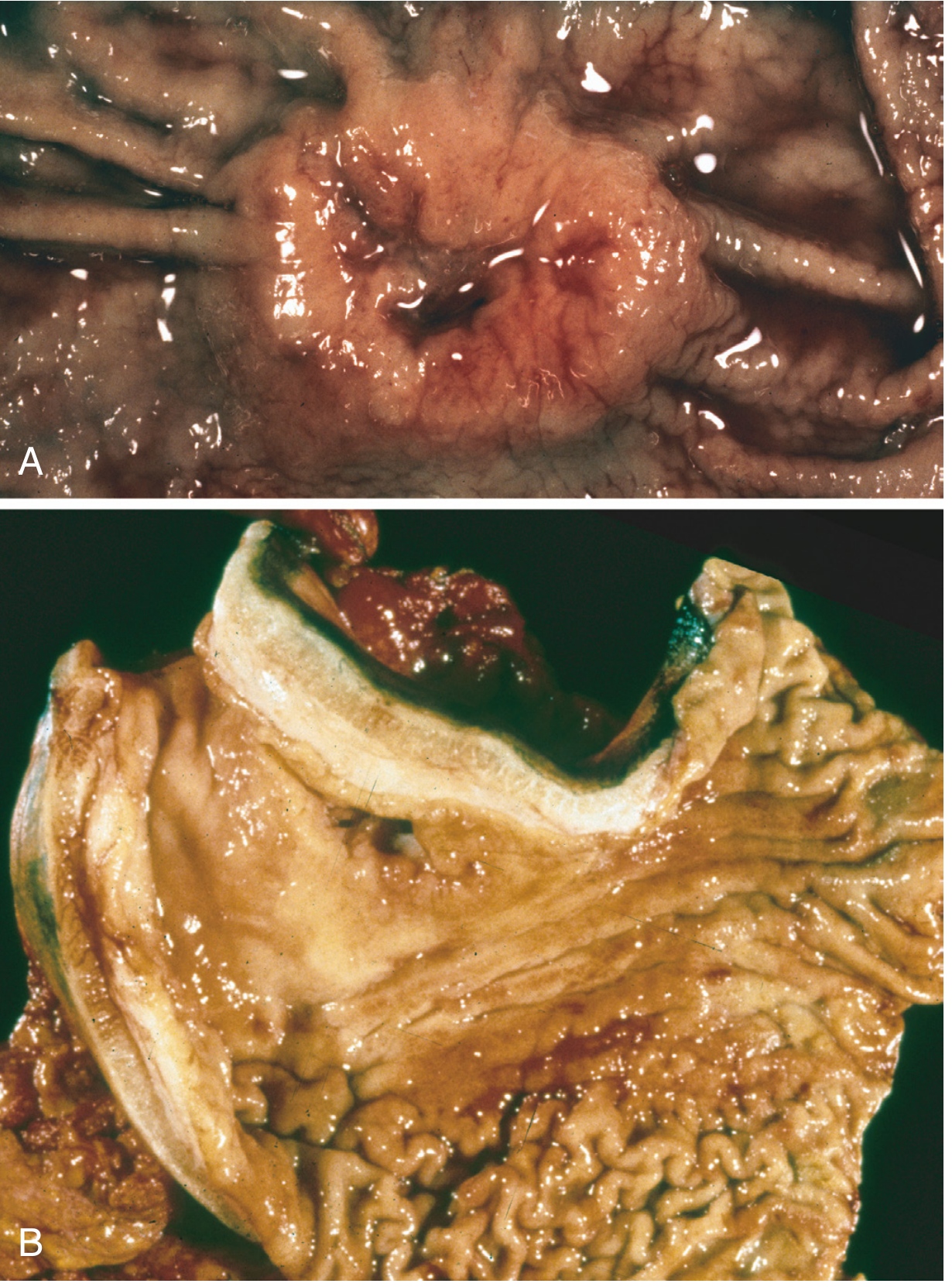
Fig. 17.20 — Robbins Pathologic Basis of Disease (9e). (A) Intestinal-type: elevated mass with heaped-up borders and central ulceration. (B) Linitis plastica due to diffuse infiltrative cancer — markedly thickened wall, rugal folds partially lost, no dominant mass.
2. Diffuse-Type Adenocarcinoma (Linitis Plastica)
- No discrete mass — tumour permeates the wall diffusely
- Evokes a desmoplastic reaction → stiffening and thickening of the entire gastric wall
- Rugal folds flatten; stomach looks rigid and shrunken → classic "leather bottle" or linitis plastica appearance
- Wall thickening can be striking on gross inspection
Polypoid Variant (for completeness)
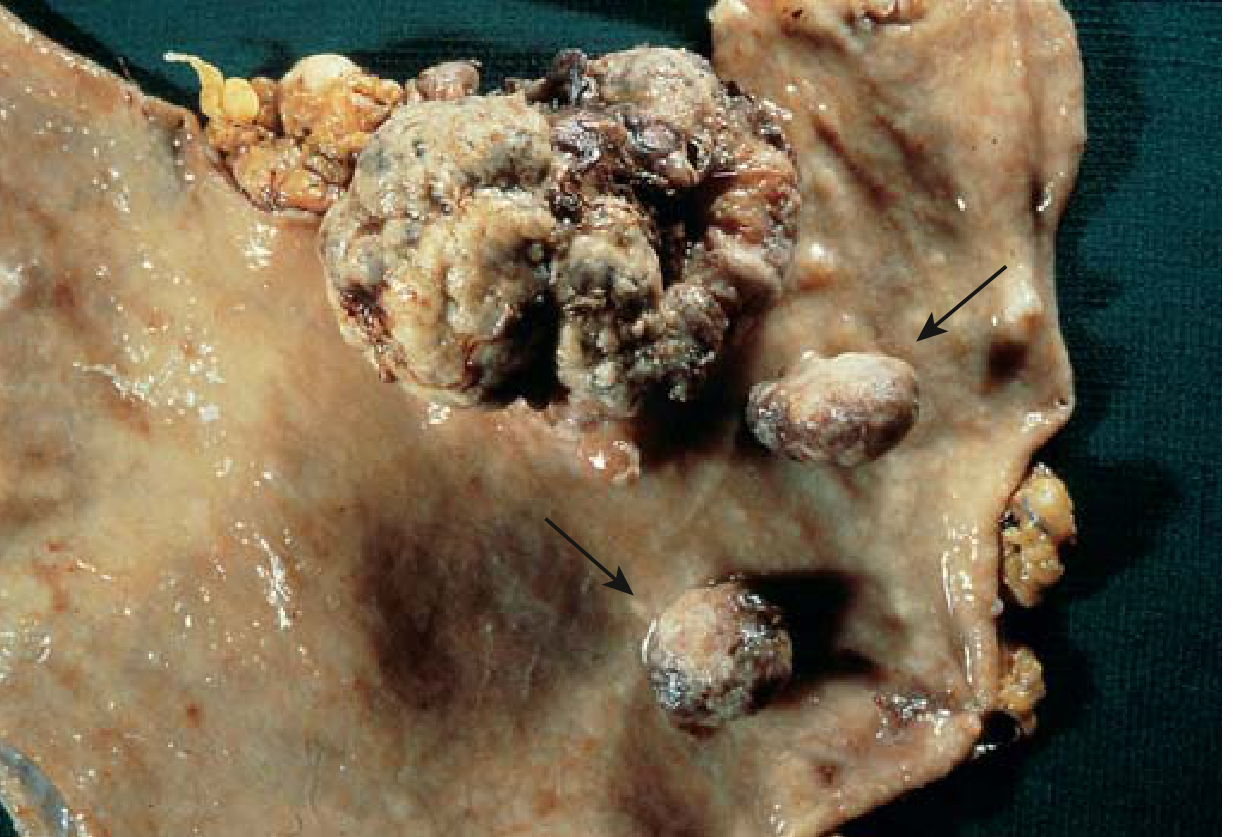
eFig. 13.4 — Robbins Basic Pathology. Polypoid gastric adenocarcinoma with two adjacent pedunculated adenomas (arrows). Note absence of rugal folds from background atrophic gastritis.
MICROSCOPIC FEATURES
1. Intestinal-Type
- Gland-forming columnar cells — resembles colonic or oesophageal adenocarcinoma
- Neoplastic cells contain apical mucin vacuoles; abundant mucin in gland lumina
- Cells arranged in irregular, back-to-back glands infiltrating through desmoplastic stroma
- Nuclear enlargement, hyperchromasia, prominent nucleoli, mitoses
2. Diffuse-Type (Signet-Ring Cell)
- No gland formation — cells permeate as single cells or small discohesive clusters
- Large cytoplasmic mucin vacuoles expand the cytoplasm and push the nucleus to the periphery → classic signet-ring cell morphology
- Signet-ring cells can be mistaken for macrophages at low power
- Loss of E-cadherin expression (CDH1 mutations) is the molecular basis for discohesion
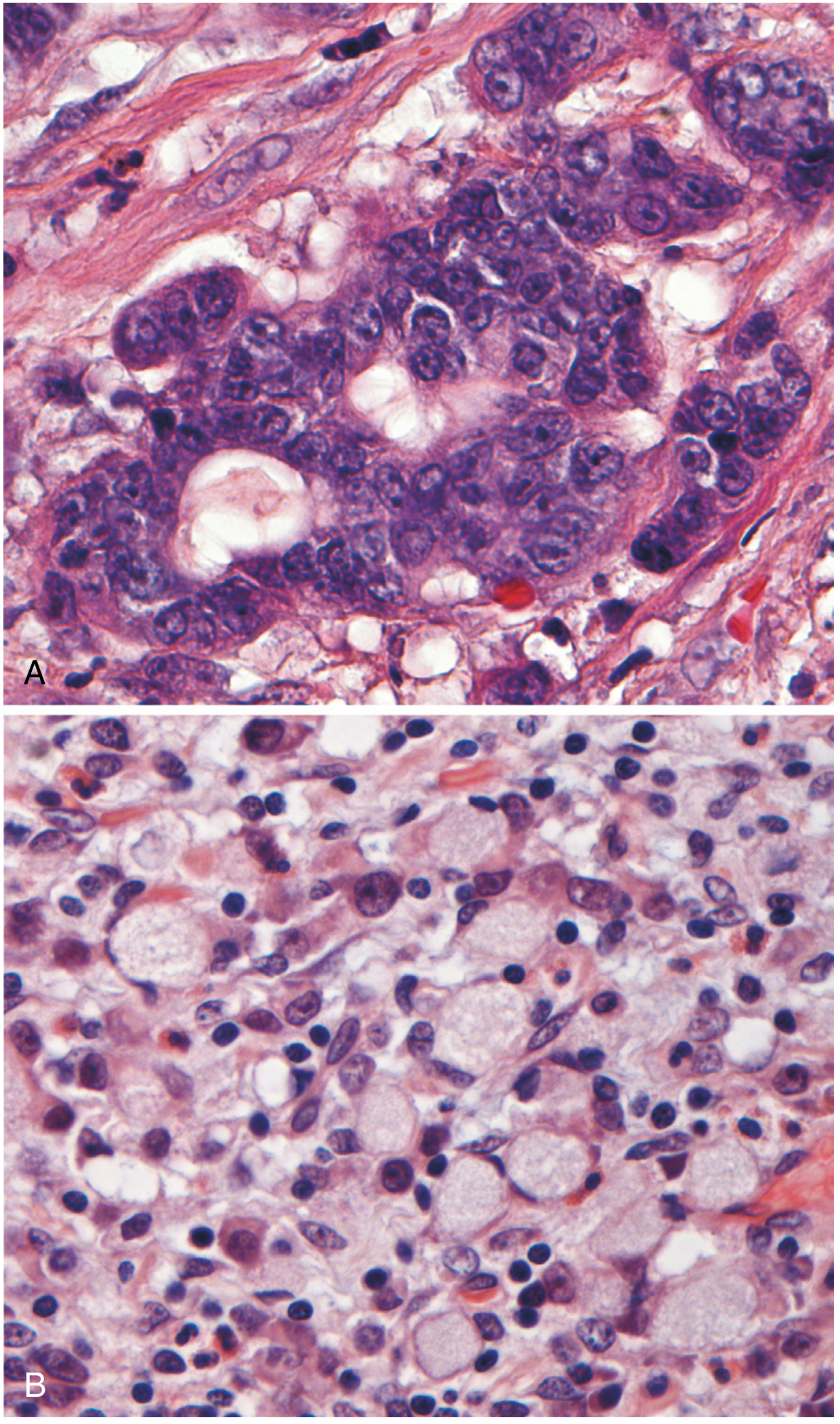
Fig. 17.21 — Robbins PBD. (A) Intestinal-type: gland-forming columnar cells in desmoplastic stroma. (B) Diffuse/signet-ring cell type: large mucin vacuoles push nuclei to periphery; no gland formation.
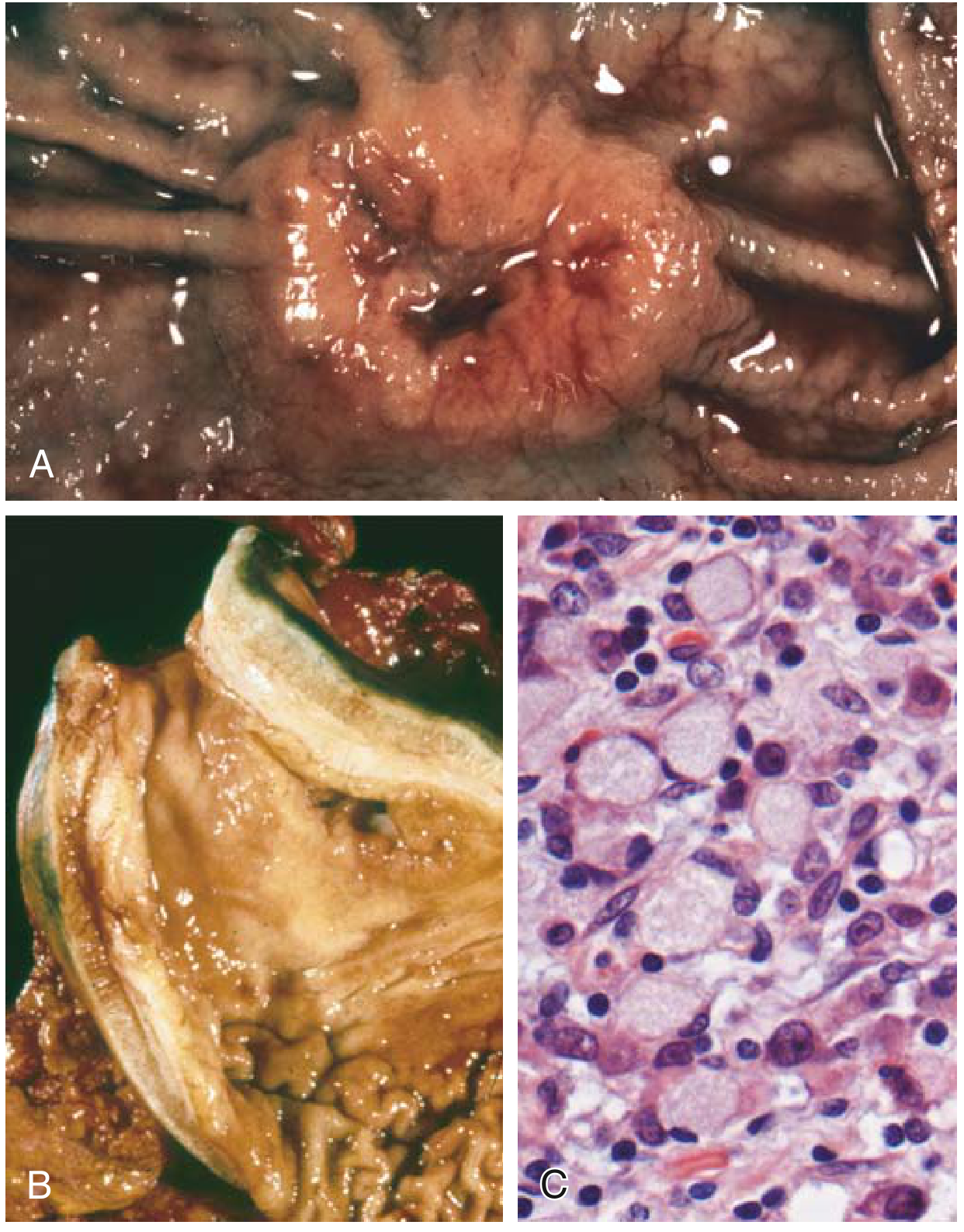
Fig. 13.16 — Robbins Basic Pathology. (A) Gross: intestinal-type mass. (B) Gross: linitis plastica. (C) Microscopy: high-power signet-ring cells.
Comparison Table
| Feature | Intestinal-Type | Diffuse-Type |
|---|---|---|
| Gross appearance | Bulky exophytic/ulcerated mass | Diffuse wall thickening (linitis plastica) |
| Growth pattern | Broad cohesive fronts | Infiltrative, single cells/small clusters |
| Histology | Gland-forming (columnar cells) | Signet-ring cells, NO glands |
| Mucin | Apical vacuoles, luminal mucin | Large cytoplasmic vacuole pushes nucleus aside |
| Stroma | Desmoplastic | Desmoplastic (causes leather-bottle stomach) |
| Key mutation | TP53, KRAS, HER2, APC | CDH1 (E-cadherin loss), RHOA |
| Precursor lesion | Atrophic gastritis → intestinal metaplasia → dysplasia | None identified |
| Geography | High incidence in Japan, Chile, E. Europe | Uniform worldwide |
| Age/sex | Mean 55 yrs; M:F = 2:1 | Mean 40 yrs (if CDH1 germline); M≈F |
| H. pylori link | Strong | Less direct |
Key Points for Exams
- Extracellular mucin released from either type can form mucin lakes that dissect tissue planes
- Metastatic sites: Virchow node (L. supraclavicular), Sister Mary Joseph nodule (periumbilical), Krukenberg tumour (ovaries — bilateral signet-ring metastases)
- Early gastric cancer = confined to mucosa/submucosa (regardless of lymph node status) → >90% 5-year survival
- Advanced gastric cancer → <20% 5-year survival
- Linitis plastica produces a rigid "leather bottle" stomach — one of the most tested gross features
Sources: Robbins, Cotran & Kumar Pathologic Basis of Disease, 10e (Fig. 17.20, 17.21) | Robbins & Kumar Basic Pathology, 11e (Fig. 13.16, eFig. 13.4)
Gastric Carcinoma 2nd yr mbbs harsh mohan
Finding Sources
Finding Sources
Harsh Mohan's Textbook of Pathology is not in the library. However, since Harsh Mohan closely follows Robbins and is structured similarly (and actually draws from Robbins for much of its content), I'll give you a Harsh Mohan–style write-up — organized exactly as it appears in that textbook — based on the Robbins content already retrieved. Harsh Mohan covers gastric carcinoma under Chapter 20: The Gastrointestinal Tract.
GASTRIC CARCINOMA
As per Harsh Mohan — Textbook of Pathology
INCIDENCE & EPIDEMIOLOGY
- Accounts for >90% of all gastric malignancies
- Worldwide: one of the most common cancers; 8% of all cancer deaths globally
- High incidence: Japan, Chile, Costa Rica, Eastern Europe (up to 20× higher than Western countries)
- Low incidence: North America, Northern Europe, Africa, Southeast Asia
- Incidence in the West has fallen >85% since the 1930s — mainly the intestinal type has declined; diffuse type remains stable
- Cancer of gastric cardia is rising (linked to Barrett oesophagus, obesity, GERD)
- Age: Mean 55 years (intestinal type); 40 years (diffuse/hereditary CDH1)
- Sex: M:F = 2:1 (intestinal type); nearly equal in diffuse type
ETIOPATHOGENESIS (Risk Factors)
Environmental/Dietary Factors
| Factor | Effect |
|---|---|
| H. pylori infection | Most important; induces chronic gastritis → atrophy → intestinal metaplasia → dysplasia → carcinoma (Correa cascade) |
| High salt, smoked/preserved foods | N-nitroso compounds, benzo[a]pyrene act as carcinogens |
| Low fruit & vegetable intake | Reduced antioxidants |
| Refrigeration (protective) | Decreased need for salt preservation → falling incidence |
| Smoking, alcohol | Modest risk increase |
| Nitrates in food | Converted to carcinogenic nitrites by bacteria |
Pre-malignant Conditions (important MCQ topic)
- Chronic atrophic gastritis with intestinal metaplasia (especially type III / incomplete metaplasia)
- Gastric adenoma — risk ↑ with size (up to 30% in >2 cm lesions)
- Pernicious anaemia (autoimmune atrophic gastritis) — 3–5× increased risk
- Post-gastrectomy stomach (Billroth II) — bile reflux, hypochlorhydria
- Ménétrier disease (hypertrophic gastropathy)
- Gastric ulcer — NOT a pre-malignant condition per se (contrast: peptic ulcer actually has reduced risk of cancer)
Molecular/Genetic Factors
- CDH1 (E-cadherin) mutations → hereditary diffuse gastric cancer; also 50% of sporadic diffuse type
- TP53 mutations — both types
- HER2 amplification (~10–20%) — therapeutic target
- Microsatellite instability (MSI) — subset of intestinal type
- EBV association — ~10% of gastric cancers (proximal stomach, diffuse pattern, lymphocytic infiltrate, PIK3CA mutations)
- RHOA mutations / CLDN18::ARHGAP26 fusions — diffuse type
CLASSIFICATION (Lauren Classification — most used)
| Intestinal Type | Diffuse Type | |
|---|---|---|
| Histology | Gland-forming | Signet-ring cells, discohesive |
| Precursor | Intestinal metaplasia | None identified |
| Growth | Cohesive, expansile | Infiltrative |
| Genetics | CIN, MSI; TP53, HER2, KRAS | CDH1 loss, RHOA |
| Epidemiology | High-incidence regions, declining | Uniform worldwide, stable |
| Age/Sex | Older (55 yrs), M>F | Younger (40 yrs), M=F |
| Prognosis | Relatively better | Worse |
GROSS MORPHOLOGY
SITE
- Antrum/pylorus — most common (50–60%)
- Lesser curvature — more than greater curvature
- Cardia — increasing incidence
- Body/fundus — least common
GROSS TYPES (Borrmann Classification)
| Type | Description |
|---|---|
| Type I — Polypoid/Fungating | Projecting mass, cauliflower-like, well-defined; best prognosis |
| Type II — Ulcerating (with raised margins) | Ulcer with heaped-up, everted edges; most common type seen |
| Type III — Infiltrating-Ulcerating | Ulcer with partial infiltration of surrounding wall |
| Type IV — Diffuse Infiltrating (Linitis Plastica) | No discrete mass; entire wall thickened, rigid, rugae flattened — "leather bottle stomach"; worst prognosis |
Key gross distinction from peptic ulcer:
- Peptic ulcer → smooth, punched-out edges, flat margins, mucosal folds radiate to edge
- Carcinomatous ulcer → irregular, raised/heaped-up edges, nodular base, folds do not reach ulcer margin
Gross images from Robbins:

(A) Intestinal-type: elevated ulcerating mass with heaped-up borders. (B) Linitis plastica: thickened, rigid gastric wall with loss of rugal folds — no discrete mass.
MICROSCOPIC MORPHOLOGY
Intestinal-Type
- Well to moderately differentiated gland-forming adenocarcinoma
- Columnar cells arranged in irregular, back-to-back glands
- Cells have apical mucin vacuoles; mucin may pool in gland lumina
- Stroma is desmoplastic (fibrous)
- Resembles intestinal/colonic adenocarcinoma
- Nuclear enlargement, hyperchromasia, prominent nucleoli, mitoses
Diffuse-Type (Signet-Ring Cell Carcinoma)
- No gland formation
- Cells are discohesive, infiltrate singly or in small clusters
- Hallmark: Signet-ring cells — large mucin vacuole fills cytoplasm, pushes nucleus to periphery → crescent-shaped eccentric nucleus
- Loss of E-cadherin accounts for discohesion
- Desmoplastic stroma → rigidity of gastric wall
- Can be mistaken for inflammatory cells at low magnification
- Release of mucin → mucin lakes dissecting tissue planes
Microscopic images from Robbins:

(A) Intestinal-type: irregular malignant glands in desmoplastic stroma. (B) Diffuse/signet-ring type: cells with large clear mucin vacuoles, crescent-shaped peripheral nuclei — no glands.

High power: signet-ring cells with large clear cytoplasmic mucin and eccentrically placed crescent nuclei.
SPREAD OF GASTRIC CARCINOMA
Direct Spread
- Duodenum, pancreas, transverse colon, liver, peritoneum
Lymphatic Spread
| Site | Eponym |
|---|---|
| Left supraclavicular node | Virchow node / Troisier sign |
| Periumbilical nodes | Sister Mary Joseph nodule |
| Left axillary node | Irish node |
Blood-Borne Spread
- Liver (most common), lungs, bone, brain
Transcoelomic Spread
- Peritoneal seedlings → Krukenberg tumour (bilateral ovarian metastasis of signet-ring cells)
- Pouch of Douglas seedlings
CLINICAL FEATURES
- Early (often silent): dyspepsia, nausea, epigastric discomfort — mimics peptic ulcer/gastritis
- Late (advanced): weight loss, anorexia, early satiety (diffuse type), dysphagia (cardia), haematemesis/melaena, iron-deficiency anaemia, palpable epigastric mass
- Paraneoplastic: acanthosis nigricans, Leser-Trélat sign (sudden onset of multiple seborrhoeic keratoses)
STAGING & PROGNOSIS
- Early gastric cancer = confined to mucosa + submucosa (regardless of node status)
-
90% 5-year survival after surgical resection
-
- Advanced gastric cancer (beyond submucosa)
- <20% 5-year survival overall in Western countries
- Most important prognostic factor: depth of invasion + lymph node + distant metastasis (TNM staging)
- In Japan (mass screening): 35% detected as early gastric cancer
SUMMARY BOX (High-Yield for MBBS)
| Point | Fact |
|---|---|
| Most common gastric malignancy | Adenocarcinoma (>90%) |
| Most common site | Antrum, lesser curvature |
| Most important risk factor | H. pylori |
| Pre-malignant conditions | Chronic atrophic gastritis, adenoma, pernicious anaemia, post-gastrectomy |
| Gross classification | Borrmann Types I–IV |
| "Leather bottle stomach" | Linitis plastica (Borrmann Type IV, diffuse type) |
| Key microscopic hallmark of diffuse type | Signet-ring cells (E-cadherin loss) |
| L. supraclavicular node met | Virchow node |
| Bilateral ovarian met | Krukenberg tumour |
| Early gastric cancer prognosis | >90% 5-yr survival |
| Falling incidence linked to | Reduced H. pylori, refrigeration, fewer preserved foods |
Note: Harsh Mohan's Textbook of Pathology (8th edition) is not currently in the library database. This write-up is structured as per Harsh Mohan's chapter organization and terminology, synthesized from Robbins & Kumar Basic Pathology (11e) and Robbins, Cotran & Kumar Pathologic Basis of Disease (10e), which form the primary reference base for Harsh Mohan.
Enumerate and describe the types of Reed Sternberg cells with well labelled diagram
Finding Sources
Your credits are exhausted. Your daily credits reset tomorrow, or you can purchase a top-up for more.
This is a shared conversation. Sign in to Orris to start your own chat.