ABG
Asking for Preferences
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Loading Image
Finding Sources
Searching Images
acid-base disorder ABG interpretation flowchart
Here is a comprehensive guide to Arterial Blood Gas (ABG) — Acid-Base Disorders.
Normal ABG Values
| Parameter | Normal Value | Range |
|---|---|---|
| pH | 7.40 | 7.35–7.45 |
| PaCO₂ | 40 mmHg | 38–42 mmHg |
| PaO₂ | 100 mmHg | 75–100 mmHg |
| HCO₃⁻ | 24 mEq/L | 22–26 mEq/L |
Stepwise ABG Interpretation
Step 1 — Determine Acidemia vs. Alkalemia (pH)
- pH < 7.35 → Acidemia
- pH > 7.45 → Alkalemia
Step 2 — Identify the Primary Process
| pH | PaCO₂ | HCO₃⁻ | Disorder |
|---|---|---|---|
| ↓ | ↑ | — | Respiratory Acidosis |
| ↓ | ↓ | ↓ | Metabolic Acidosis |
| ↑ | ↓ | — | Respiratory Alkalosis |
| ↑ | ↑ | ↑ | Metabolic Alkalosis |
Key rule: In respiratory disorders, PaCO₂ moves opposite to pH. In metabolic disorders, PaCO₂ moves in the same direction as pH (as compensation).
Step 3 — Assess Compensation
Compensation never fully corrects pH (except sometimes in respiratory alkalosis).
| Primary Disorder | Compensation Formula |
|---|---|
| Metabolic acidosis | Expected PaCO₂ = 1.5 × [HCO₃⁻] + 8 ± 2 (Winter's Formula) |
| Metabolic alkalosis | Expected PaCO₂ = 0.7 × [HCO₃⁻] + 20 ± 5 |
| Acute respiratory acidosis | ↑ HCO₃⁻ = ΔPaCO₂ ÷ 10 |
| Chronic respiratory acidosis | ↑ HCO₃⁻ = 4 × (ΔPaCO₂ ÷ 10) |
| Acute respiratory alkalosis | ↓ HCO₃⁻ = 2 × (ΔPaCO₂ ÷ 10) |
| Chronic respiratory alkalosis | ↓ HCO₃⁻ = 5 × (ΔPaCO₂ ÷ 10) |
If compensation is more or less than expected → mixed disorder.
Step 4 — Calculate the Anion Gap (AG)
AG = Na⁺ − (Cl⁻ + HCO₃⁻)
Normal = 8–12 mEq/L (or up to 16 if using older norms)
Correct for albumin (hypoalbuminemia lowers AG):
Corrected AG = Measured AG + 2.5 × (4 − [albumin g/dL])
High AG metabolic acidosis causes (MUDPILES / GOLDMARK):
- Methanol, Uremia, DKA, Propylene glycol, Isoniazid/Iron, Lactic acidosis, Ethylene glycol, Salicylates
Normal AG (hyperchloremic) metabolic acidosis — check urine anion gap:
- Urine AG positive (renal cause): RTA, renal failure
- Urine AG negative (GI cause): diarrhea, fistula
Step 5 — Delta-Delta Ratio (Δ/Δ) — if AG is elevated
Δ/Δ = (Measured AG − 12) ÷ (24 − Measured HCO₃⁻)
| Δ/Δ Ratio | Interpretation |
|---|---|
| < 1.0 | High-AG acidosis + concurrent non-AG metabolic acidosis |
| 1.0–2.0 | Pure high-AG metabolic acidosis |
| > 2.0 | High-AG acidosis + concurrent metabolic alkalosis (or chronic respiratory acidosis) |
Step 6 — Base Excess / Base Deficit
Base excess (BE): Amount of base needed to titrate 1L of blood to pH 7.4 at 37°C.
- Normal: −2 to +2
- Positive (> +4) → metabolic alkalosis
- Negative (< −4, i.e., base deficit) → metabolic acidosis
Calculating base deficit from ABG:

For every ΔPaCO₂ of 10, pH changes 0.08 in the opposite direction. Base deficit = (Expected pH − Measured pH) × 2/3 × 150.
Quick Summary Table
| Disorder | pH | PaCO₂ | HCO₃⁻ | Common Causes |
|---|---|---|---|---|
| Respiratory acidosis | ↓ | ↑ | ↑ (compensatory) | COPD, opioids, obesity hypoventilation, NMJ disease |
| Respiratory alkalosis | ↑ | ↓ | ↓ (compensatory) | Anxiety, PE, sepsis, altitude, pregnancy |
| Metabolic acidosis | ↓ | ↓ (compensatory) | ↓ | DKA, lactic acidosis, renal failure, diarrhea |
| Metabolic alkalosis | ↑ | ↑ (compensatory) | ↑ | Vomiting, diuretics, hyperaldosteronism, antacid overuse |
Clinical Example
84-year-old on IV morphine, rib fractures, AKI → pH 7.18, PaCO₂ 73, HCO₃⁻ 28, normal AG:
- pH < 7.35 → acidemia
- PaCO₂ ↑ → primary respiratory acidosis
- HCO₃⁻ only mildly elevated → no significant metabolic compensation → acute process
- Normal anion gap
- Diagnosis: Acute respiratory acidosis from opioid-induced respiratory depression → treat with naloxone
Sources: Current Surgical Therapy 14e, p. 1608 | Barash Clinical Anesthesia 9e, p. 1158–1161
Still more depth
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Searching Images
metabolic acidosis anion gap causes MUDPILES GOLDMARK
Searching Images
acid base nomogram Davenport HCO3 pH PCO2
Loading Image
Loading Image
Here is the deep-dive on ABG acid-base disorders.
ABG Acid-Base Disorders — In Depth
The Henderson-Hasselbalch Framework
All acid-base physiology rests on:
pH = pKa + log([HCO₃⁻] / α·PCO₂)
The Kassirer-Bleich approximation is more practical at the bedside:
[H⁺] = 24 × PaCO₂ / [HCO₃⁻]
With [H⁺] in nmol/L. Key reference points: [H⁺] of 25 ≈ pH 7.6, 40 ≈ pH 7.4, 63 ≈ pH 7.2.
This equation shows the two control levers: the lungs (PaCO₂) and the kidneys (HCO₃⁻). These are independent and compensatory — each responds to disturbances in the other.
The Acid-Base Nomogram
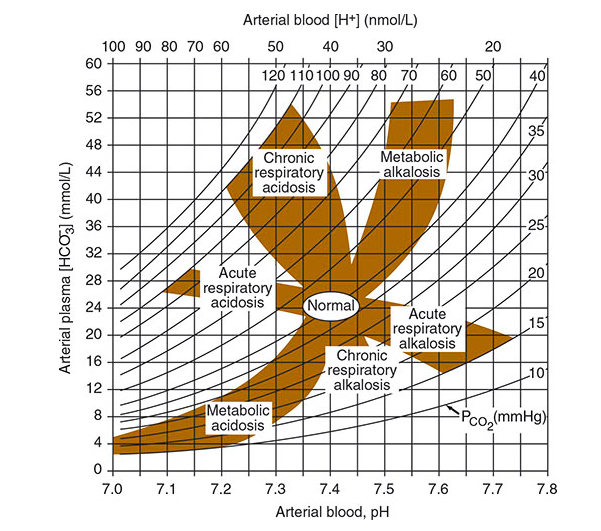
This nomogram plots pH (x-axis) against arterial plasma [HCO₃⁻] (y-axis) with diagonal PCO₂ isobars. Each shaded region represents the expected range for a simple disorder with compensation. If an ABG plots outside the confidence band for its primary disorder → mixed disorder present.
1. Metabolic Acidosis
Pathophysiology
Three mechanisms:
- Excess endogenous acid production (lactic acid, ketoacids) — overwhelms renal excretion
- Loss of HCO₃⁻ (diarrhea, RTA) — kidneys not at fault, but bicarbonate is gone
- Impaired renal acid excretion (CKD, RTA) — ammoniagenesis fails
Respiratory compensation (hyperventilation) begins within minutes; full renal compensation takes 3–5 days. Winter's formula predicts the expected PaCO₂:
Expected PaCO₂ = 1.5 × [HCO₃⁻] + 8 ± 2
Kussmaul respiration (slow, deep breathing) is the classic sign — minimizes dead-space and maximizes CO₂ elimination.
Classification by Anion Gap
AG = Na⁺ − (Cl⁻ + HCO₃⁻) | Normal: 8–12 mEq/L
Always correct for albumin: each 1 g/dL drop in albumin below 4 g/dL → AG underestimated by ~2.5 mEq/L.
High-AG Metabolic Acidosis
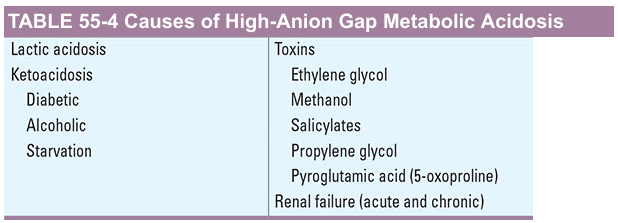
Mnemonic — GOLDMARK:
- G — Glycols (ethylene glycol → oxalate; propylene glycol)
- O — 5-Oxoproline (pyroglutamic acid — chronic paracetamol users, malnourished)
- L — L-lactic acidosis (sepsis, shock, mesenteric ischaemia, metformin)
- D — D-lactic acidosis (short bowel, bacterial overgrowth)
- M — Methanol (→ formic acid; causes bilateral putaminal necrosis)
- A — Aspirin/salicylates (mixed HAGMA + respiratory alkalosis due to CNS stimulation)
- R — Renal failure (GFR <20 mL/min; sulfate, phosphate, lactate accumulate)
- K — Ketoacidosis (DKA, alcoholic, starvation)
Special points:
- Toxic alcohols: Initially raise the osmolar gap (measured − calculated osmolality >10). As the alcohol is metabolised to acid, osmolar gap falls and AG rises. So early on, both gaps may be elevated.
- Salicylate OD: Produces a mixed picture — primary respiratory alkalosis (direct medullary stimulation) + HAGMA (lactic acid, ketone formation). Treat with IV NaHCO₃ to alkalinise urine and trap salicylate ions.
- DKA: May present as HAGMA or normal-AG (hyperchloremic) acidosis, depending on hydration status and how fast ketones are renally excreted.
Normal-AG (Hyperchloremic) Metabolic Acidosis
HCO₃⁻ replaced by Cl⁻, so AG unchanged. Causes:
| Category | Examples |
|---|---|
| GI HCO₃⁻ loss | Diarrhea (most common), ileostomy, pancreatic/biliary fistula, cholestyramine |
| Renal HCO₃⁻ loss | Proximal RTA (Type 2), carbonic anhydrase inhibitors (acetazolamide) |
| Impaired renal H⁺ excretion | Distal RTA (Type 1), Type 4 RTA (hypoaldosteronism), early CKD (GFR 20–50) |
| Iatrogenic | Large-volume normal saline infusion (dilutional), ammonium chloride, TPN amino acids |
| Urologic diversion | Uretero-sigmoidostomy (ileum/colon Cl⁻/HCO₃⁻ exchangers absorb urine Cl⁻ → lose HCO₃⁻) |
Differentiating GI vs. renal cause → Urine Anion Gap (UAG):
UAG = Urine (Na⁺ + K⁺) − Urine Cl⁻
| UAG | Interpretation |
|---|---|
| Negative (< 0) | Renal NH₄⁺ excretion is intact → GI cause (diarrhea) |
| Positive (> +20) | Kidneys failing to excrete NH₄⁺ → RTA or renal failure |
Note: UAG is unreliable in HAGMA because unmeasured anions (lactate, ketones) are present in urine instead of NH₄⁺.
RTA Subtypes
| Type | Defect | Urine pH | K⁺ | Cause |
|---|---|---|---|---|
| 1 (Distal) | H⁺ secretion failure in collecting duct | >5.5 | ↓ (hypokalemia) | Sjögren's, amphotericin, medullary sponge kidney |
| 2 (Proximal) | HCO₃⁻ reabsorption failure | <5.5 (when acidotic) | ↓ | Fanconi syndrome, acetazolamide, multiple myeloma |
| 4 | Hypoaldosteronism → ↓ NH₄⁺ excretion | <5.5 | ↑ (hyperkalemia) | DM nephropathy, NSAIDs, ACEi/ARBs, heparin |
Treatment of Metabolic Acidosis
- Priority is treating the underlying cause (insulin for DKA, fluids/vasopressors for lactic acidosis, naloxone for opioid OD)
- NaHCO₃ is indicated for:
- Chronic hyperchloremic acidosis (RTA) — prevents bone catabolism
- pH <7.1 with severe hyperkalemia (shifts K⁺ intracellularly)
- Salicylate/tricyclic OD (urinary alkalinisation)
- Severe metabolic acidosis with concurrent respiratory acidosis (mixed)
- NaHCO₃ is NOT clearly beneficial in DKA, lactic acidosis due to sepsis/shock — risks include paradoxical intracellular acidosis (CO₂ generated crosses membranes faster than HCO₃⁻), left-shift of O₂–Hb dissociation curve, decreased ionised Ca²⁺
2. Metabolic Alkalosis
Pathophysiology
Occurs when H⁺ is lost or HCO₃⁻ is gained. The kidney normally excretes excess HCO₃⁻ easily — alkalosis is maintained only when the kidney's ability to excrete HCO₃⁻ is impaired. The key perpetuating factor is usually volume depletion (activates RAAS → Na⁺/H⁺ exchange keeps reclaiming HCO₃⁻) or hypokalemia (drives H⁺ into cells, increases renal H⁺ secretion).
Compensatory hypoventilation raises PaCO₂ by ~0.6–0.7 mmHg per 1 mEq/L rise in HCO₃⁻. Compensation rarely exceeds PaCO₂ of 55 mmHg (hypoxia drives ventilation back up).
Classification: Chloride-Responsive vs. Chloride-Resistant
| Chloride-Responsive | Chloride-Resistant | |
|---|---|---|
| Urine Cl⁻ | < 20 mEq/L | > 40 mEq/L |
| Mechanism | Volume/Cl⁻ depletion → RAAS activation | Mineralocorticoid excess or severe K⁺ depletion |
| Causes | Vomiting/NG suction, diuretics (remote), posthypercapnia, diarrhoea with Cl⁻ loss, villous adenoma | Hyperaldosteronism, Cushing's, Bartter/Gitelman syndrome, exogenous steroids, renal artery stenosis, severe hypokalemia, milk-alkali syndrome |
| Treatment | IV 0.9% saline + KCl → kidneys excrete HCO₃⁻ | Treat underlying cause; spironolactone, K⁺ repletion; acetazolamide; HCl infusion (central line) for refractory cases |
Posthypercapnia alkalosis: When chronic respiratory acidosis (↑ PaCO₂ → ↑ HCO₃⁻ compensation) is rapidly corrected (e.g., intubation of a COPD patient), PaCO₂ normalises but the HCO₃⁻ takes days to correct → metabolic alkalosis appears.
Clinical Features of Severe Alkalosis
- Neuromuscular: Tetany, muscle cramps, weakness (from ↓ ionised Ca²⁺, hypokalaemia)
- Cardiac: Arrhythmias (especially with concurrent hypokalaemia)
- Respiratory: Hypoventilation (compensation) — can worsen respiratory failure
- CNS: Confusion, obtundation
3. Respiratory Acidosis
Pathophysiology
Any cause of alveolar hypoventilation → CO₂ retention → ↑ PaCO₂ → acidaemia.
Buffering timeline:
- Acute (<24 h): Cellular buffers (proteins, phosphate) raise HCO₃⁻ by ~1 mEq/L per 10 mmHg ↑ PaCO₂
- Chronic (>24–48 h): Renal adaptation raises HCO₃⁻ by ~4 mEq/L per 10 mmHg ↑ PaCO₂ (max ~38 mEq/L)
Causes (Organised by Level)
| Level | Examples |
|---|---|
| Respiratory centre depression | Opioids, benzodiazepines, anaesthetic agents, head trauma, brainstem stroke, obesity hypoventilation, sleep apnoea |
| Neuromuscular | GBS, MG, ALS, diaphragm paralysis, hypophosphataemia, hypokalaemia, botulism |
| Airways/lungs | Severe asthma, COPD exacerbation, ARDS (late), pneumonia, pulmonary oedema |
| Mechanical ventilation | Inadequate tidal volume/rate, high PEEP causing dead space, ventilator circuit leak |
| Thoracic wall | Flail chest, kyphoscoliosis, morbid obesity |
Clinical Features
| Severity | Features |
|---|---|
| Mild/chronic | Sleep disturbance, daytime somnolence, personality change, memory loss |
| Moderate | Anxiety, confusion, asterixis, tremor, headache, papilloedema (cerebral vasodilation from CO₂) |
| Severe/acute | Psychosis, seizures, coma |
Cerebral vasodilation from hypercapnia can mimic raised ICP with papilloedema and focal signs.
Permissive Hypercapnia
Intentional strategy in ARDS/severe asthma — accept ↑ PaCO₂ to use lung-protective low tidal volumes (6 mL/kg IBW). Target: pH ≥7.20–7.25. Caution: Worsens if concurrent metabolic acidosis (lactic acidosis) is present. Short-term NaHCO₃ may be used if pH is critically low.
Treatment
- Remove the cause (naloxone for opioids, bronchodilators for asthma/COPD, NIV/intubation)
- NIV (BiPAP) is first-line for COPD exacerbations and obesity hypoventilation
- Caution when correcting chronic respiratory acidosis rapidly — if renal HCO₃⁻ retention has occurred, normalising PaCO₂ quickly leaves behind a metabolic alkalosis (posthypercapnic alkalosis)
4. Respiratory Alkalosis
Pathophysiology
Alveolar hyperventilation → CO₂ blown off → ↓ PaCO₂ → alkalemia.
Compensation:
- Acute: HCO₃⁻ falls ~2 mEq/L per 10 mmHg ↓ PaCO₂ (cellular buffering)
- Chronic (>2–6 h → days): Kidneys reduce NH₄⁺/titratable acid excretion, decrease HCO₃⁻ reabsorption → HCO₃⁻ falls ~4–5 mEq/L per 10 mmHg ↓ PaCO₂
Chronic respiratory alkalosis is the only disorder that can be fully compensated (pH may normalise).
Causes
| Category | Examples |
|---|---|
| CNS stimulation | Anxiety/panic (hyperventilation syndrome), pain, fever, CVA, subarachnoid haemorrhage |
| Hypoxia-driven | PE, pneumonia, high altitude, severe anaemia, heart failure |
| Drugs | Salicylates (early, before HAGMA develops), progesterone (pregnancy), theophylline, catecholamines |
| Hepatic | Hepatic encephalopathy (direct CNS stimulation) |
| Sepsis | Early sepsis/SIRS — most common cause in ICU |
| Mechanical ventilation | Over-ventilation — always check settings |
Key clinical pearl: In early sepsis, the ABG typically shows a respiratory alkalosis due to the hyperdynamic phase. As sepsis progresses → lactic acidosis (HAGMA) superimposes → the pH may be falsely normalised despite two concurrent disorders.
Clinical Features
- Acute: Circumoral and peripheral paraesthesias, light-headedness, carpopedal spasm (↓ ionised Ca²⁺ from pH change), dizziness, rarely seizures
- Cardiac: Arrhythmias (coronary vasospasm from alkalosis + left-shift of O₂–Hb curve → myocardial ischaemia)
- Hypocapnia itself reduces cerebral blood flow — CNS signs even without hypoxia
Treatment
Treat the underlying cause. For panic-attack hyperventilation: paper-bag rebreathing raises PaCO₂. In mechanically ventilated patients: reduce RR or tidal volume.
5. Mixed Acid-Base Disorders
A mixed disorder is present when two or more primary disorders coexist simultaneously. Clues:
- pH and PaCO₂ move in the same direction (normally they oppose each other)
- Compensation is more or less than expected per formula
- ABG plots outside the nomogram confidence band
- Clinical history strongly suggests multiple concurrent processes
Common Mixed Disorders
| Combination | Clinical Scenario | ABG Pattern |
|---|---|---|
| Met. acidosis + Resp. alkalosis | Sepsis, salicylate OD, hepatic failure | pH may be normal; PaCO₂ lower than predicted by Winter's |
| Met. acidosis + Resp. acidosis | Cardiac arrest, severe COPD + renal failure, over-sedation in sepsis | pH very low; PaCO₂ paradoxically high |
| Met. alkalosis + Resp. acidosis | COPD on diuretics; NG suction + chronic hypoventilation | HCO₃⁻ very high; pH may be near-normal |
| Met. alkalosis + Resp. alkalosis | Liver cirrhosis + vomiting | pH very high; PaCO₂ lower than expected |
| HAGMA + Met. alkalosis | DKA + vomiting; AKI + nasogastric drainage | AG elevated but HCO₃⁻ near normal → Δ/Δ >2 |
| HAGMA + Normal-AG acidosis | DKA + diarrhoea; toluene toxicity; uremia + saline resuscitation | Δ/Δ <1; HCO₃⁻ falls more than AG rises |
| Triple disorder | Alcoholic ketoacidosis + vomiting + hepatic dysfunction | AG acidosis + met. alkalosis + resp. alkalosis |
The Delta-Delta Ratio Revisited
Δ/Δ = (AG − 12) ÷ (24 − HCO₃⁻)
This compares how much the AG has risen vs. how much HCO₃⁻ has fallen. In a pure HAGMA, every extra anion titrates exactly one HCO₃⁻, so Δ/Δ = 1.
| Δ/Δ | Meaning |
|---|---|
| < 1.0 | HCO₃⁻ has fallen more than AG rose → concurrent non-AG acidosis |
| 1.0–2.0 | Pure HAGMA |
| > 2.0 | HCO₃⁻ hasn't fallen as much as expected → concurrent met. alkalosis OR chronic resp. acidosis with renal HCO₃⁻ retention |
Nuance: In lactic acidosis, Δ/Δ often >1 because lactate can be redistributed intracellularly. In DKA, Δ/Δ is often <1 because excreted ketone salts represent lost potential HCO₃⁻.
6. ABG in Key Clinical Contexts
DKA
- Classic: HAGMA (AG >12), low HCO₃⁻, low pH, compensatory low PaCO₂
- Can be normo-AG if well-hydrated and excreting ketones rapidly
- Hyperkalemia at presentation despite total body K⁺ depletion (insulin deficiency + acidosis shifts K⁺ out of cells)
- During treatment: As ketones clear → watch for postherapeutic alkalosis (ketones metabolised to HCO₃⁻)
COPD Exacerbation
- Chronic resp. acidosis: ↑ PaCO₂, ↑ HCO₃⁻ (compensated), near-normal pH
- Acute-on-chronic: ↑ PaCO₂ further, pH drops — look for inadequate HCO₃⁻ rise for degree of hypercapnia
- Post-NIV/intubation: Rapid CO₂ correction → posthypercapnic metabolic alkalosis
Sepsis
- Early: Respiratory alkalosis (hyperventilation)
- Late: HAGMA from lactic acidosis (type A — impaired tissue O₂ delivery)
- Often mixed: resp. alkalosis + met. acidosis → pH deceptively near-normal
- Metformin accumulates in sepsis/AKI → can cause Type B lactic acidosis
Renal Failure
- GFR 20–50 mL/min: Hyperchloremic non-AG acidosis (impaired NH₄⁺ excretion)
- GFR <20 mL/min: HAGMA (sulfate, phosphate, organic acids accumulate)
- Chronic metabolic acidosis accelerates bone demineralisation and muscle catabolism
Pregnancy
- Chronic respiratory alkalosis (progesterone stimulates ventilation)
- Normal ABG in pregnancy: pH ~7.44, PaCO₂ ~30 mmHg, HCO₃⁻ ~20 mEq/L
- What looks like "acidosis" in a non-pregnant patient may be normal in pregnancy
7. Physiologic Effects of Severe pH Derangements
| Effect | Acidemia (pH <7.2) | Alkalemia (pH >7.6) |
|---|---|---|
| Cardiac | ↓ contractility, ↓ CO, vasodilation, ventricular arrhythmias | Arrhythmias, coronary vasospasm |
| Respiratory | Hyperventilation (met. acidosis), fatigue | Hypoventilation |
| Electrolytes | Hyperkalemia (H⁺/K⁺ exchange), ↑ ionised Ca²⁺ | Hypokalemia, ↓ ionised Ca²⁺ (tetany) |
| O₂ delivery | Right-shift O₂–Hb curve (↑ O₂ off-loading) | Left-shift O₂–Hb curve (↓ O₂ off-loading) |
| CNS | Confusion → coma | Seizures, altered consciousness |
| Enzyme function | Denaturation, impaired coagulation cascade | Reduced enzyme efficiency |
Sources:
- Brenner and Rector's The Kidney, pp. 707–715
- Murray & Nadel's Textbook of Respiratory Medicine, pp. 274–281
- Barash Clinical Anesthesia 9e, pp. 1153–1161
- Harrison's Principles of Internal Medicine 22e, pp. 1465–1471
- Fishman's Pulmonary Diseases and Disorders, pp. 3228–3236
This is a shared conversation. Sign in to Orris to start your own chat.