Osteoarthritis
Reading File
Finding Sources
Searching PubMed
"osteoarthritis"[MeSH Terms] AND ("management" OR "treatment")
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
I now have comprehensive, well-sourced material to deliver a thorough answer on osteoarthritis.
Osteoarthritis
Definition
Osteoarthritis (OA) is characterized by failed repair of synovial joint tissues after intra-articular stress, resulting in breakdown of cartilage and bone that leads to pain, stiffness, and functional disability. Although the name implies inflammation, OA is primarily a degenerative disorder of articular cartilage, with inflammation acting as a secondary contributor. — Goldman-Cecil Medicine, Ch. 241; Robbins Basic Pathology, p. 789
Epidemiology
- Affects >500 million people worldwide; one of the top 15 causes of years lived with disability globally
- Incidence rises sharply between ages 50–75; ~40% of people >70 are affected
- Women disproportionately affected, especially for knee and hand OA
- Projected to affect ~25% of the total U.S. adult population by 2040
- Lifetime risk of symptomatic knee OA: ~40% in men, ~47% in women; rises to 60% in persons above healthy weight
- 80% of those affected have movement limitations; 25% cannot perform major activities of daily living
— Goldman-Cecil Medicine, p. 2766
Classification
| Type | Description |
|---|---|
| Primary (idiopathic) | No identifiable cause; appears as an aging phenomenon; oligoarticular (few joints); more common in women |
| Secondary | Identifiable predisposing condition in ~5% of cases; occurs at any age |
Secondary causes include:
- Trauma (acute or chronic repetitive)
- Metabolic/systemic: hemochromatosis, Wilson disease, ochronosis
- Endocrine: acromegaly, hypothyroidism, hyperparathyroidism, diabetes mellitus
- Crystal deposition: CPPD, gout
- Other: Rheumatoid arthritis, Paget disease, bone/joint dysplasias
— Grainger & Allison's Diagnostic Radiology
Pathogenesis
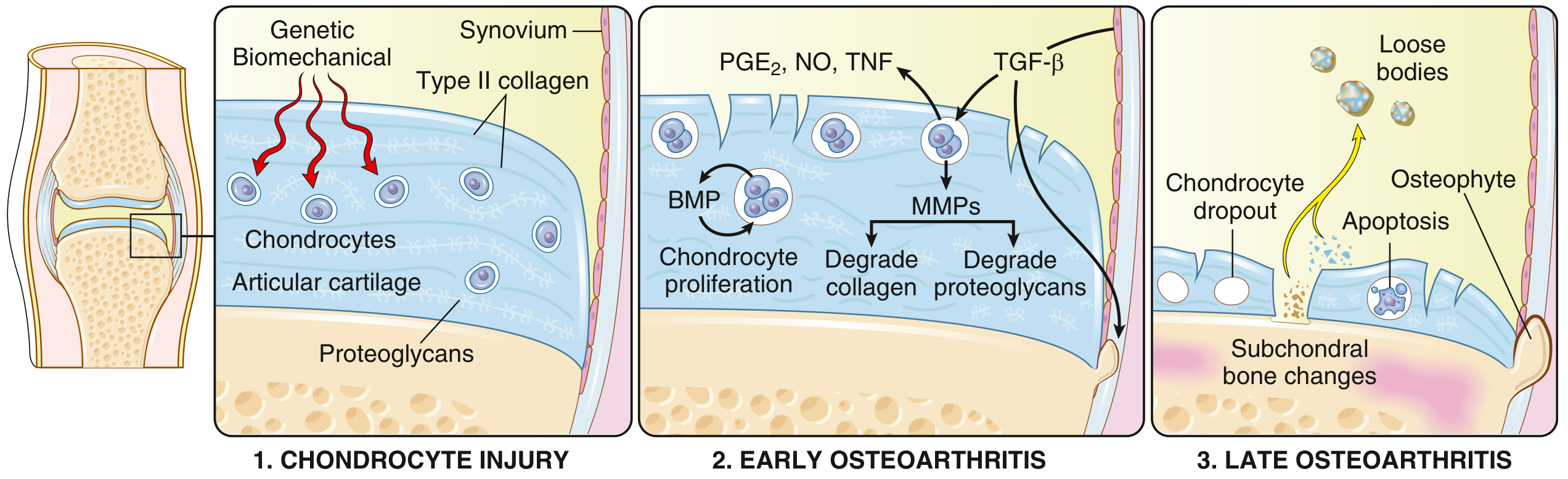
OA stems from an imbalance between repair and destruction of joint tissues driven by mechanical, inflammatory, and metabolic pathways:
- Chondrocyte injury — Biomechanical stress (± genetic predisposition) initiates damage to articular cartilage. Proteoglycans initially absorb water (cartilage swelling), then the type II collagen matrix becomes disrupted.
- Early OA — Chondrocytes proliferate in an attempted repair response, secreting matrix metalloproteinases (MMPs) that degrade type II collagen and proteoglycans. Pro-inflammatory mediators (PGE₂, NO, TNF) further deregulate chondrocyte function and stimulate synovial inflammation.
- Late OA — Continued MMP activity exceeds repair capacity → chondrocyte dropout via apoptosis, full-thickness cartilage loss, subchondral bone changes, osteophyte formation, and loose bodies.
Cartilage/bone breakdown products stimulate the synovium to become hyperplastic and produce cytokines (IL-1β, TNF, IL-6), which further perpetuate cartilage damage. Subchondral bone undergoes remodeling, with trabecular thickening and cyst formation.
— Goldman-Cecil Medicine, p. 2767; Robbins Basic Pathology, p. 790
Morphology (Gross & Histologic)
| Feature | Description |
|---|---|
| Cartilage fibrillation | Earliest change — vertical clefts in articular cartilage surface |
| Cartilage erosion | Progressive loss → full-thickness sloughing |
| Loose bodies (joint mice) | Dislodged fragments of cartilage ± bone |
| Bone eburnation | Exposed subchondral bone becomes the articular surface; polished ivory appearance from friction |
| Subchondral cysts | Synovial fluid forced into bone via ball-valve mechanism through fracture gaps |
| Osteophytes | Marginal bony outgrowths capped by fibrocartilage → gradually ossify |
| Synovium | Only mildly congested and fibrotic; scattered chronic inflammatory cells (contrast with RA) |
— Robbins Basic Pathology, p. 790
OA vs. Rheumatoid Arthritis: Key Differences
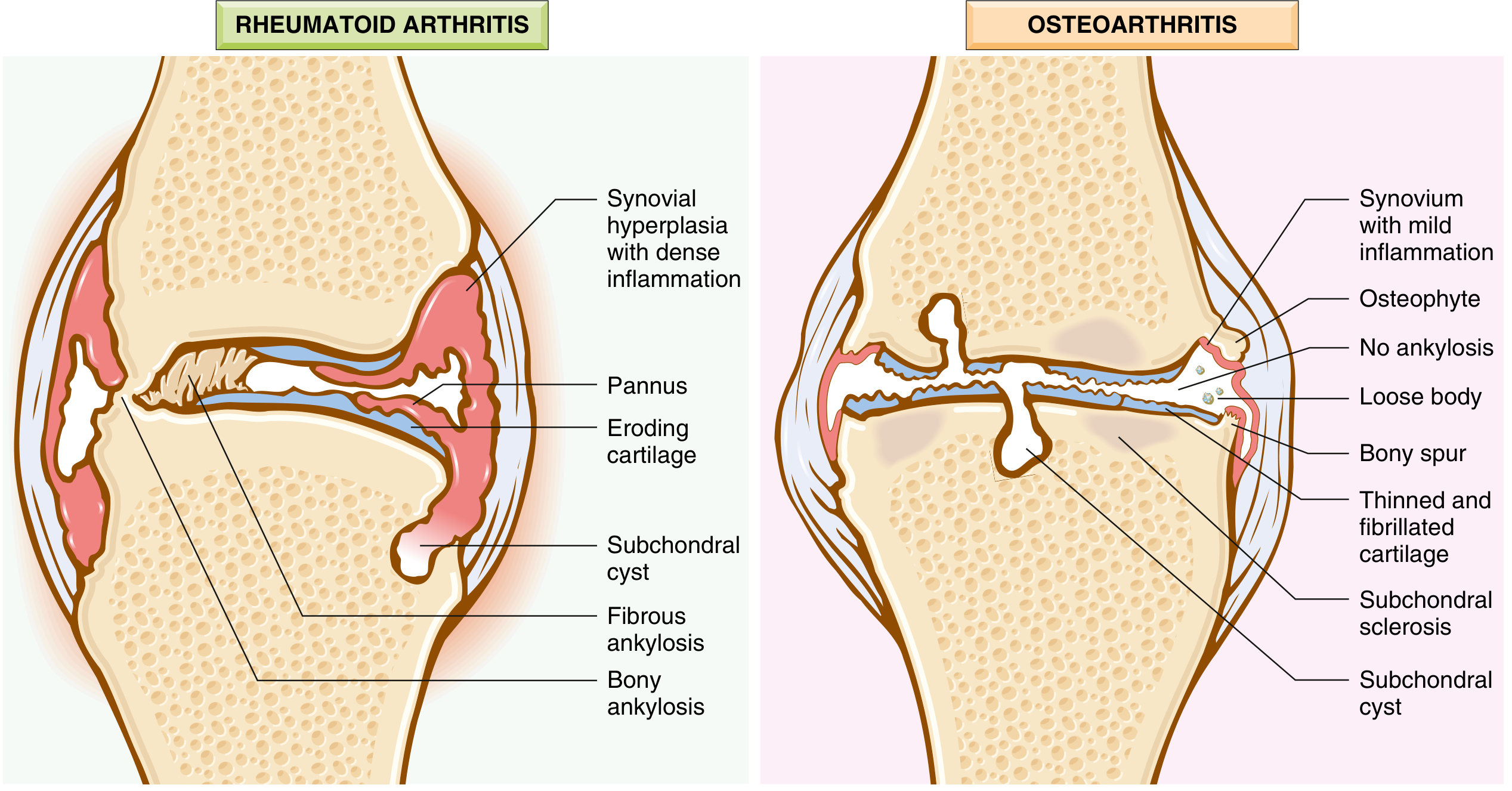
| Feature | Osteoarthritis | Rheumatoid Arthritis |
|---|---|---|
| Primary mechanism | Mechanical cartilage injury | Autoimmunity (T cells, antibodies) |
| Inflammation | Secondary | Primary |
| Joints | Weight-bearing (knee, hip) | Small joints of hands first; multiple |
| Serum antibodies | None | ACPA, rheumatoid factor |
| Ankylosis | Does NOT occur | Can occur |
| Systemic involvement | No | Yes (lungs, heart, etc.) |
Clinical Features
- Onset: usually >50 years; insidious
- Pain: worsens with use/activity; eased by rest (early) — contrast with inflammatory arthritis where rest worsens stiffness
- Morning stiffness: brief (<30 min); "gelling" after inactivity
- Crepitus on movement
- Limited range of motion
- Joint deformity over time (but no fusion)
- Heberden nodes: osteophytes at distal interphalangeal (DIP) joints — more common in women
- Bouchard nodes: osteophytes at proximal interphalangeal (PIP) joints
- Spinal OA: osteophytes impinge on foramina → radicular pain, muscle spasm, neurologic deficits
Commonly involved joints: hips, knees, lower lumbar/cervical spine, DIP/PIP fingers, 1st carpometacarpal, 1st tarsometatarsal
Important: Radiographic severity correlates poorly with pain and disability.
— Robbins Basic Pathology, p. 791; Goldman-Cecil Medicine, p. 2768
Risk Factors
| Category | Factors |
|---|---|
| Systemic | Age, female sex, genetics, obesity, metabolic syndrome |
| Local/mechanical | Previous joint injury, malalignment, muscle weakness, occupation (repetitive loading), ligamentous laxity |
| Modifiable | Obesity (↑ mechanical load + metabolic/inflammatory effect), physical inactivity, quadriceps weakness |
Diagnosis
Primarily clinical — imaging and labs are rarely needed for initial diagnosis.
Clinical criteria (knee OA): age >50, pain with activity, brief morning stiffness, crepitus, bony enlargement, no warmth
Imaging (when used):
- X-ray: joint space narrowing, subchondral sclerosis, osteophytes, subchondral cysts
- MRI: earlier cartilage changes, bone marrow lesions, synovial hypertrophy
Labs: generally normal. Useful to exclude inflammatory arthritis (check ESR/CRP, rheumatoid factor, synovial fluid for crystals) when features are atypical.
Management
Management is individually tailored with a core–adjunct–surgical framework:
Core (First-line for all patients)
- Self-management education and health literacy
- Exercise — land-based (aerobic, resistance) and aquatic exercise are equally effective; reduces pain and improves function
- Weight management — even 5–10% weight loss reduces knee pain; crucial in obese patients
Pharmacological
| Agent | Use |
|---|---|
| Topical NSAIDs (e.g., diclofenac gel) | First-line pharmacotherapy, especially knee/hand OA |
| Oral NSAIDs | Effective for pain; use lowest dose/duration; GI/CV/renal risk |
| Paracetamol (acetaminophen) | Modest benefit; safer than NSAIDs for short-term use |
| Duloxetine | Useful when central sensitization or co-morbid depression present |
| Intra-articular corticosteroids | Short-term pain relief; esp. when effusion present |
| Intra-articular hyaluronic acid | Modest benefit; more controversial |
| Opioids | Reserved for refractory cases; significant adverse effects limit use |
Surgical
- Total joint replacement (arthroplasty) — highly effective for severe, refractory knee or hip OA; greatest benefit when functional limitation is marked
- Unicompartmental knee replacement, osteotomy — selected patients
Note: No treatment has been proven to prevent or halt OA progression (no disease-modifying OA drug [DMOAD] is currently approved).
— Goldman-Cecil Medicine, pp. 2770–2772; Robbins Basic Pathology, p. 791
Recent Evidence (2024–2025)
- A 2025 network meta-analysis in the BMJ (PMID: 41093618) compared exercise modalities in knee OA — all forms reduce pain, with resistance and aerobic exercise showing the greatest benefit.
- A 2024 systematic review (PMID: 38963824) updated evaluation and management recommendations, reinforcing exercise and weight loss as first-line strategies.
- A 2025 meta-analysis (PMID: 39212129) found collagen supplementation provided modest benefit in knee OA, though effect sizes were small.
Sources: Goldman-Cecil Medicine (Ch. 241), Robbins & Kumar Basic Pathology (Ch. 19), Grainger & Allison's Diagnostic Radiology (Ch. 44), Textbook of Family Medicine 9e
Multiple sclerosis for nurses
Reading File
Finding Sources
Searching PubMed
"multiple sclerosis"[MeSH Terms] AND "nursing"[MeSH Terms]
Finding Sources
Searching PubMed
"multiple sclerosis"[MeSH Terms] AND "disease-modifying"
Finding Sources
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
You've run out of free credits. Sign up for a free account to continue using Orris.
This is a shared conversation. Sign in to Orris to start your own chat.