vsd
Reading File
Finding Sources
Searching PubMed
"ventricular septal defect"[MeSH Terms] AND management
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Ventricular Septal Defect (VSD)
VSD is an abnormal communication between the left and right ventricles. It is the most common congenital cardiac anomaly diagnosed at birth, comprising 20–30% of all congenital heart disease.
Anatomy & Classification
The ventricular septum is formed by a muscular ridge growing upward from the apex fusing with a membranous partition growing downward from the endocardial cushions. The basal (membranous) region is the last to develop and accounts for ~90% of VSDs.
VSDs are classified into four types by location:
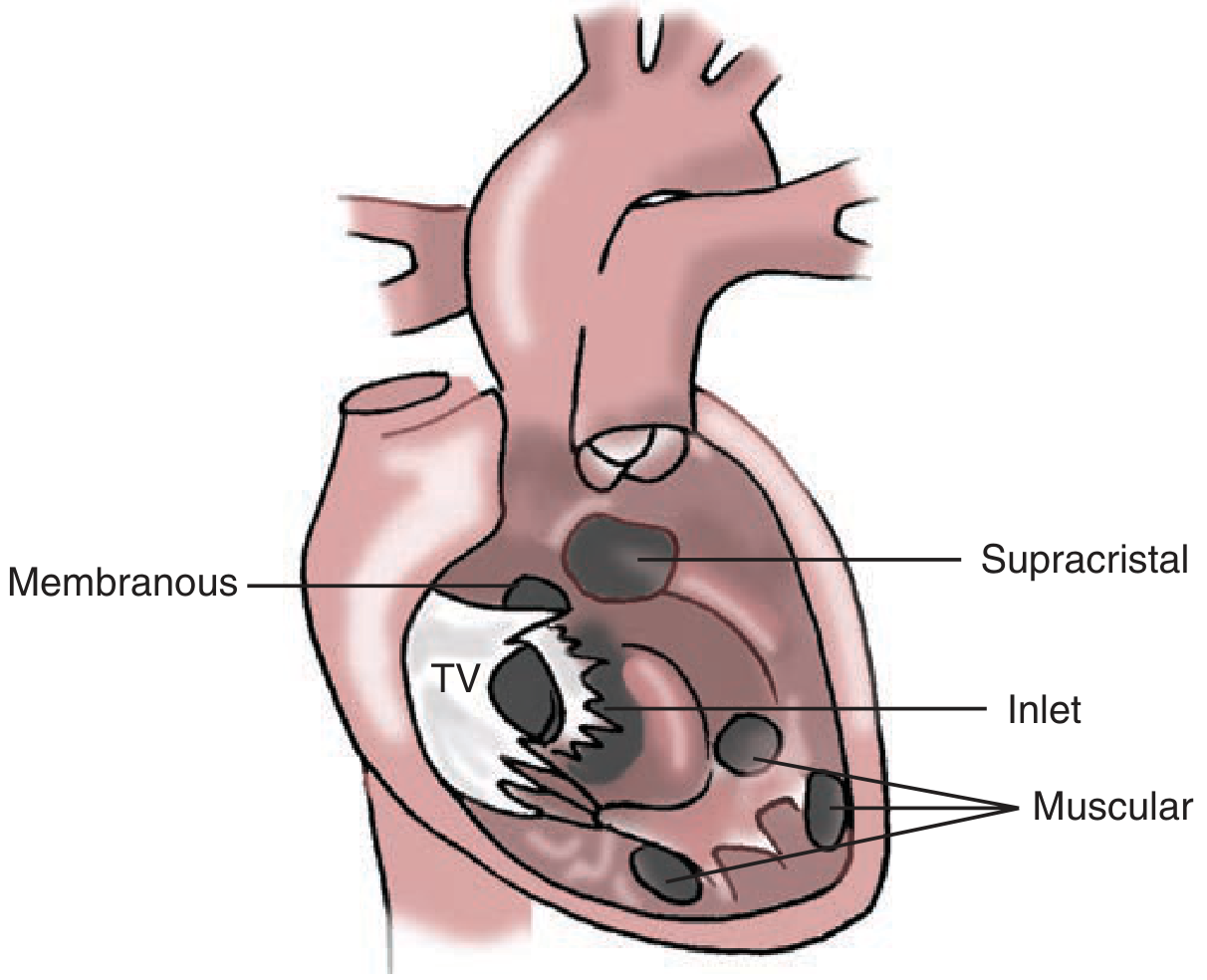
| Type | Features |
|---|---|
| Perimembranous (paramembranous) | Most common requiring surgery (~80% of surgical cases); involves membranous septum; includes malalignment defects of Tetralogy of Fallot |
| Muscular | Most common overall; surrounded by muscle; can be multiple ("Swiss-cheese" type); located anterior, midventricular, posterior, or apical |
| Inlet (AV canal) | Beneath the tricuspid valve; absent AV canal septum; usually associated with other anomalies |
| Supracristal (outlet) | Conal septum defect; limited by pulmonary valve; above the crista supraventricularis |
Pathophysiology
- Direction and magnitude of shunt depends on defect size and the ratio of pulmonary to systemic vascular resistance (PVR/SVR)
- Large (nonrestrictive) VSDs — diameter ≥ aortic annulus; allow free LV→RV flow; RV pressure equalizes with systemic; high Qp:Qs
- Small (restrictive) VSDs — offer significant resistance; RV pressure normal or minimally elevated; Qp:Qs rarely >1.5
- Chronic large left-to-right shunting → pulmonary hypertension → eventually reversal to right-to-left shunt = Eisenmenger syndrome (occurs in ~10% of VSDs)
- Small/moderate VSDs: jet lesions damage RV endocardium → risk of infective endocarditis
Clinical Presentation
| Defect size | Presentation |
|---|---|
| Small | Asymptomatic; loud grade ≥4 pansystolic murmur at 3rd–4th ICS left sternal border; otherwise normal exam |
| Moderate | Symptoms in childhood; often repaired before adulthood |
| Large | Severe CHF, recurrent respiratory infections, failure to thrive in infancy; in adults → Eisenmenger complex with cyanosis |
Key auscultatory finding: High-frequency, widely radiating holosystolic murmur maximal at the lower left sternal border (3rd–4th ICS) — the loud murmur with otherwise normal exam is a classic diagnostic clue.
Diagnosis
- Echo (transthoracic): First-line — identifies defect, estimates shunt size, assesses LV/RV size and PA pressures
- CXR: Cardiomegaly, pulmonary overcirculation with large defects
- ECG: LV hypertrophy (moderate/large); biventricular hypertrophy with large defects; left atrial enlargement
- Cardiac catheterization: Reserved for pre-surgical assessment in older patients or those with pulmonary hypertension to measure PVR reactivity
Natural History
- ~50% of all native VSDs are small; >50% of small VSDs close spontaneously
- Moderate/large VSDs: close spontaneously in only ~10%
- Closure rate is highest in the first decade of life and inversely related to age:
- 1 month old → ~80% chance of spontaneous closure
- 12 months old → ~25% chance
- Spontaneous closure in adult life is unusual
Treatment
Indications for closure:
- Large defects — repair in infancy (irreversible pulmonary vascular changes can develop in first year of life)
- Moderate defects — Qp:Qs >1.5 with hemodynamic significance
- Eisenmenger complex — contraindicates surgery (prohibitive PVR); manage medically
Surgical repair:
- Cardiopulmonary bypass with moderate hypothermia and cardioplegic arrest
- Right atrial approach preferred for most defects
- Right ventriculotomy for apical muscular defects
- Pulmonary arteriotomy or subpulmonary RV incision for supracristal defects
- Patch closure (synthetic or pericardial) with interrupted or continuous suture technique
Transcatheter closure:
- For perimembranous VSDs, transcatheter device closure is as effective as open surgery and is now the preferred approach in suitable anatomy
Eisenmenger complex management:
- Medical complications of cyanosis (erythrocytosis, paradoxical embolism, hemoptysis)
- Lung or heart-lung transplantation in selected patients
Sources: Schwartz's Principles of Surgery (11th ed.), Goldman-Cecil Medicine, Robbins & Kumar Basic Pathology
infective endocarditis
Finding Sources
Searching PubMed
"infective endocarditis"[MeSH Terms] AND management
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Loading Image
Infective Endocarditis (IE)
Definition
Infective endocarditis is a microbial infection of the endocardial surface of the heart — primarily cardiac valves — leading to formation of vegetations composed of fibrin, inflammatory cells, and microorganisms, often with underlying tissue destruction. Prosthetic valves, pacemaker leads, and other intracardiac devices may also be involved.
Classification
| Type | Features |
|---|---|
| Acute IE | Rapidly progressive, destructive; high morbidity/mortality even with treatment; typically S. aureus on healthy valves |
| Subacute IE | Insidious onset; protracted weeks-to-months course; typically viridans streptococci on damaged valves; most recover with antibiotics |
A clear delineation is not always possible — many cases fall along a spectrum.
Epidemiology & Risk Factors
- Incidence: ~3–14 cases per 100,000 persons/year (Western Europe, US)
- ~10,000–20,000 new cases/year in the United States
- Predisposing conditions:
| More Common | Less Common |
|---|---|
| Mitral valve prolapse (with regurgitation) — leading risk factor | Rheumatic heart disease |
| Degenerative valvular disease | Idiopathic hypertrophic subaortic stenosis |
| Injection drug use | Coarctation of the aorta |
| Congenital heart disease (esp. uncorrected VSD) | Complex cyanotic congenital heart disease |
| Previous endocarditis | |
| Prosthetic valves (10–20% of all IE cases) |
- Host risk factors: neutropenia, immunodeficiency, malignancy, diabetes, alcohol use, IV drug use
Microbiology
| Setting | Predominant Organisms |
|---|---|
| Community-acquired (native valve) | S. viridans (50–60%); subacute presentation |
| Healthcare-associated / Hospital | S. aureus — now the #1 cause in high-income countries; acute; attacks healthy valves |
| IV drug users | S. aureus (~70%); tricuspid valve predominance |
| Prosthetic valve (early, <60 days) | S. aureus, coagulase-negative staphylococci |
| Prosthetic valve (late) | Staphylococci (still #1), VGS, S. gallolyticus |
| Culture-negative (~10–15%) | Prior antibiotics, fastidious organisms (Bartonella, Coxiella burnetii, Legionella, fungi) |
HACEK group (oral commensals): Haemophilus, Aggregatibacter, Cardiobacterium, Eikenella, Kingella
Pathology & Morphology
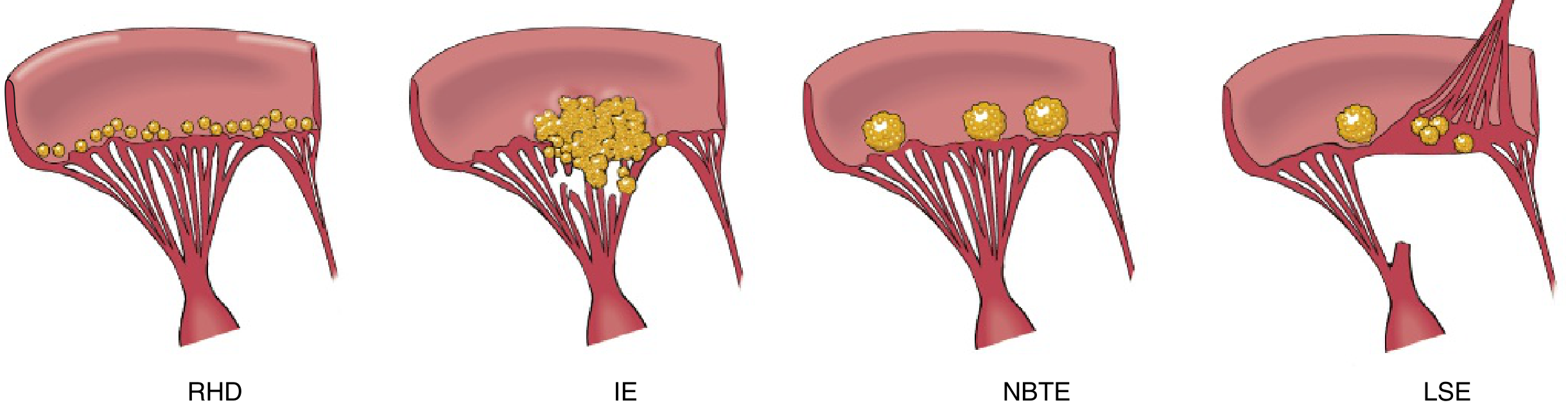
- Vegetations in IE are friable, bulky, and destructive, containing fibrin, inflammatory cells, and organisms
- Most common sites: Aortic and mitral valves (left-sided); tricuspid in IV drug users
- Ring abscess: Vegetation erodes into underlying myocardium → abscess cavity
- Mycotic aneurysms: Emboli seed arterial walls → local bacterial infection
- Septic emboli → septic infarcts (brain, kidney, spleen, lungs in right-sided IE)
Clinical Features
Symptoms:
- Fever — most consistent sign (may be absent in subacute IE in the elderly)
- Fatigue, weight loss, flu-like illness (subacute); chills, rigors (acute)
- Murmur present in ~90% of left-sided IE
Classic peripheral stigmata:
| Sign | Description |
|---|---|
| Osler nodes | Painful fingertip nodules (immunologic) |
| Janeway lesions | Painless erythematous palm/sole lesions (embolic) |
| Roth spots | Retinal hemorrhages with pale centers |
| Splinter hemorrhages | Nail bed linear hemorrhages |
| Petechiae | From microemboli |
Complications: Glomerulonephritis (immune complex deposition → hematuria, proteinuria, renal failure), arrhythmias (septal extension), heart failure, stroke.
Diagnosis — 2023 Duke Criteria
Definite IE: 2 major criteria OR 1 major + 3 minor OR 5 minor criteria
Possible IE: 1 major + 1 minor OR 3 minor criteria
Possible IE: 1 major + 1 minor OR 3 minor criteria
Major Criteria
A. Microbiologic:
- ≥2 separate blood cultures positive for typical IE organisms (VGS, S. gallolyticus, S. aureus, HACEK, enterococci)
- OR persistently positive blood cultures (≥2 drawn >12 hr apart; or ≥3 of ≥4 drawn ≥1 hr apart)
- OR single positive culture / elevated IgG titer for Coxiella burnetii (Q fever)
- OR positive PCR for C. burnetii, Bartonella, or T. whipplei
B. Imaging:
- Echo/CT: vegetation, valvular perforation/aneurysm, abscess, pseudoaneurysm, intracardiac fistula, new valvular regurgitation, new prosthetic valve dehiscence
- ¹⁸F-FDG PET/CT: abnormal metabolic activity involving native/prosthetic valve or intracardiac device (≥3 months post-implant)
C. Surgical: Direct intraoperative visualization of IE
Minor Criteria
- Predisposing heart condition or IV drug use
- Fever >38°C (>36°C in 2023 update)
- Vascular phenomena: arterial emboli, septic pulmonary infarcts, mycotic aneurysm, Janeway lesions, intracranial hemorrhage, conjunctival hemorrhages
- Immunologic phenomena: glomerulonephritis, Osler nodes, Roth spots, rheumatoid factor
- Microbiologic evidence not meeting major criteria
Investigations
- Blood cultures: Draw ≥3 sets from different sites before antibiotics; critical for diagnosis and guiding therapy
- Echo:
- TTE first in most patients
- TEE when: TTE is non-diagnostic, prosthetic valve, high clinical suspicion, complicated IE (suspected abscess)
- CBC: Normochromic normocytic anemia; leukocytosis (more acute); thrombocytopenia (~10%)
- ESR/CRP: Elevated in ~60%
- Urinalysis: Hematuria, proteinuria (glomerulonephritis)
- CT/MRI: For embolic complications (brain, spleen, kidneys); also CT of chest/abdomen/pelvis
Treatment
Antibiotic Therapy (general principles)
Prolonged IV bactericidal therapy is required. All patients should be managed in an inpatient setting with a multidisciplinary "IE team" (Infectious Disease, Cardiology, Cardiac Surgery).
| Organism | Regimen |
|---|---|
| Penicillin-susceptible streptococci (MIC ≤0.12 μg/mL) | Penicillin G or ceftriaxone 2 g IV daily × 4 weeks (or 2 weeks + gentamicin for uncomplicated NVE) |
| Relatively resistant streptococci (MIC 0.12–0.5 μg/mL) | Penicillin G or ceftriaxone × 4 weeks + gentamicin × 2 weeks |
| MSSA | Nafcillin/oxacillin × 4–6 weeks |
| MRSA | Vancomycin × 6 weeks (target trough 15–20 μg/mL) |
| Enterococci | Ampicillin + gentamicin × 4–6 weeks; or ampicillin + ceftriaxone for aminoglycoside-resistant strains |
| HACEK organisms | Ceftriaxone 2 g IV once daily × 4 weeks |
| Fungi | Amphotericin B ± azole; surgery almost always required |
| Penicillin-allergic patients | Vancomycin (avoid clindamycin due to C. difficile risk; doxycycline as alternative) |
- Native valve IE (NVE): 4-week minimum
- Prosthetic valve IE (PVE): 6-week minimum preferred
Antibiotic Prophylaxis (AHA 2021 Update)
Prophylaxis is recommended only for high-risk patients undergoing invasive dental procedures:
- Prosthetic cardiac valves or valve repair with devices
- Previous IE
- Unrepaired cyanotic congenital heart disease
- Left ventricular assist devices / implantable hearts
Preferred regimen: Amoxicillin 2 g PO 30–60 min before procedure
Penicillin-allergic: Doxycycline (clindamycin no longer recommended)
Key change (2021): 90% of "penicillin-allergic" patients will have negative skin testing — allergy screening before defaulting to alternative agents.
Penicillin-allergic: Doxycycline (clindamycin no longer recommended)
Key change (2021): 90% of "penicillin-allergic" patients will have negative skin testing — allergy screening before defaulting to alternative agents.
Indications for Surgery
| Indication | Class |
|---|---|
| Acute AI or MR with heart failure | I |
| Fungal endocarditis | I |
| Annular/aortic abscess, fistula, valve dehiscence/perforation/rupture | I |
| Persistent infection despite 7–10 days appropriate antibiotics (no noncardiac source) | I |
| Recurrent emboli after appropriate antibiotic therapy | I |
| Gram-negative enteric or antibiotic-resistant organisms with valve dysfunction | I |
| Anterior mitral leaflet vegetation >10 mm OR persistent vegetation after embolization | IIa |
| Vegetation enlarging despite therapy | IIb |
| Prosthetic valve IE — early PVE (<2 months post-op), heart failure, nonstreptococcal/non-HACEK organisms, perivalvular leak/abscess/fistula/new conduction disturbance | I |
There is no survival benefit to delaying surgery >7 days after ischemic stroke when valve replacement is otherwise indicated.
Prognosis
- Untreated IE is universally fatal
- With appropriate antibiotics ± surgery, mortality is substantially reduced but remains significant (~20–30% in-hospital mortality overall)
- Worse prognosis: S. aureus, prosthetic valve IE, Eisenmenger complex, abscess formation, CNS embolization, heart failure
Sources: Robbins & Kumar Basic Pathology, Braunwald's Heart Disease (11th ed.), Goldman-Cecil Medicine, Harrison's Principles of Internal Medicine 22E (2025)
This is a shared conversation. Sign in to Orris to start your own chat.