Oxygen cascade
Reading File
Finding Sources
Finding Sources
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Loading Image
Finding Sources
Reading File
Reading File
I now have comprehensive information to construct a detailed answer on the oxygen cascade. Let me compile this.
The Oxygen Cascade
The oxygen cascade describes the stepwise fall in partial pressure of oxygen (PO₂) as it travels from atmospheric air down to the mitochondria — the site of utilization. Each step involves a pressure "drop," driven by passive diffusion along concentration gradients.
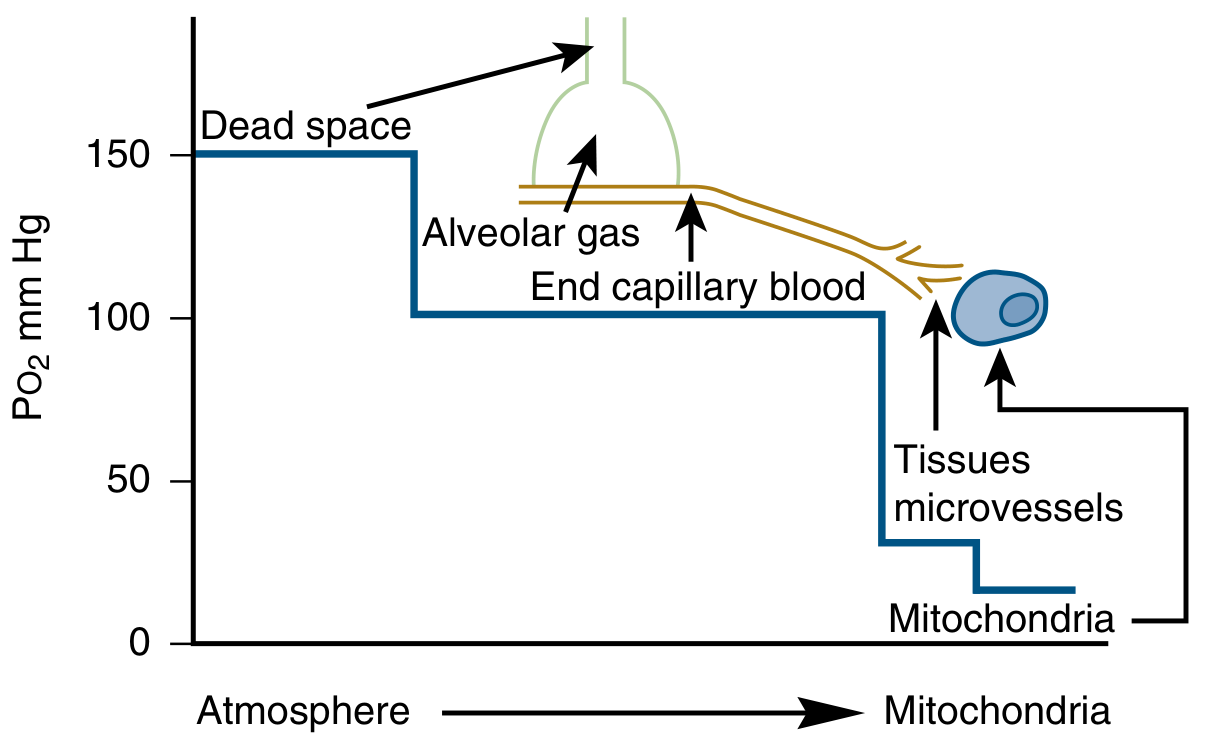
Steps of the Cascade
1. Atmospheric Air → Inspired Air
| Location | PO₂ (mmHg) |
|---|---|
| Dry atmosphere (sea level) | ~159 |
| Moist inspired air (trachea) | ~149 |
Atmospheric O₂ = 20.9% × 760 mmHg = 159 mmHg.
Once inhaled, air is warmed and humidified. Water vapor pressure at 37°C = 47 mmHg, so:
PO₂ (moist inspired) = 0.209 × (760 − 47) = 149 mmHg
2. Inspired Air → Alveolar Gas (~100 mmHg)
This is the largest single drop in the cascade. The alveolar PO₂ settles lower than inspired PO₂ because:
- Ventilation continuously adds O₂ to alveoli
- Pulmonary blood flow continuously removes O₂
The equilibrium is described by the Alveolar Gas Equation:
PAO₂ = PIO₂ − (PACO₂ / R)
Where:
- PIO₂ = inspired PO₂ (~149 mmHg)
- PACO₂ = alveolar PCO₂ (~40 mmHg, equal to arterial in normal lungs)
- R = respiratory exchange ratio (typically ~0.8)
PAO₂ = 149 − (40/0.8) = 149 − 50 = ~100 mmHg
The alveolar PO₂ varies only 3–4 mmHg with each breath because the FRC volume dampens oscillations. CO₂ displaces O₂ in the alveolus, accounting for most of this drop.
3. Alveolar Gas → Arterial Blood (~95 mmHg)
Even in the ideal lung, end-capillary blood would equal alveolar PO₂. In reality, arterial PO₂ is slightly lower due to the alveolar-to-arterial (A-a) gradient (normally 10–12 mmHg on air).
Three mechanisms widen this gradient:
- Diffusion limitation — thickened alveolar membrane (e.g., interstitial lung disease, extreme exercise at altitude)
- Ventilation-perfusion (V/Q) mismatch — the major contributor in most lung disease; low V/Q units contribute desaturated blood
- Shunt — true right-to-left shunt (anatomic or intrapulmonary); notably refractory to supplemental O₂ because raising FiO₂ cannot oxygenate non-ventilated alveoli
Gas exchange is normally perfusion-limited (not diffusion-limited): haemoglobin fully saturates in the first one-third of the pulmonary capillary under resting conditions.
4. Arterial Blood → Tissue Capillaries / Mixed Venous Blood (~40 mmHg)
Oxygen is delivered to tissues by:
- Haemoglobin (primary carrier): ~1.34 mL O₂/g Hb when fully saturated; SaO₂ ≈ 97–98% at PaO₂ ~95 mmHg
- Dissolved O₂: only 0.003 mL/100 mL/mmHg (minor contribution)
Oxygen delivery (DO₂):
DO₂ = CO × (Hb × 1.34 × SaO₂ + 0.003 × PaO₂)
At the tissue level, O₂ unloads from haemoglobin along the oxyhaemoglobin dissociation curve. The Bohr effect (↑PCO₂, ↑H⁺, ↑temperature, ↑2,3-DPG) shifts the curve rightward, lowering O₂ affinity and promoting unloading in metabolically active tissues. The P50 (PO₂ at 50% saturation) is normally ~27 mmHg.
Mixed venous PO₂ = ~40 mmHg (SvO₂ ~75%), reflecting the extraction by tissues.
5. Tissue Capillaries → Mitochondria (<5 mmHg)
Tissue extraction drops PO₂ a further ~55 mmHg. The mitochondrial PO₂ is not precisely measurable, but oxidative phosphorylation (cytochrome c oxidase) continues efficiently at only a few mmHg of PO₂.
Summary Table
| Step | Location | PO₂ (mmHg) | Mechanism of Drop |
|---|---|---|---|
| 1 | Dry atmosphere | 159 | — |
| 2 | Moist inspired air | 149 | Water vapour dilution |
| 3 | Alveolar gas | ~100 | CO₂ exchange; alveolar gas equation |
| 4 | Arterial blood | ~95 | A-a gradient (V/Q mismatch, shunt, diffusion) |
| 5 | Mixed venous blood | ~40 | Tissue O₂ extraction; Bohr effect |
| 6 | Mitochondria | <5 | Cellular utilization |
Clinical Relevance
| Perturbation | Step Affected | Example |
|---|---|---|
| High altitude | Step 1→2: ↓Pbar → ↓PIO₂ → ↓PAO₂ | AMS, HAPE |
| Hypoventilation | Step 2: ↑PACO₂ → ↓PAO₂ | Opioids, COPD exacerbation |
| V/Q mismatch | Step 3: ↑A-a gradient | PE, pneumonia, asthma |
| Shunt | Step 3: ↑A-a gradient, O₂-refractory | Hepatopulmonary syndrome, ARDS |
| Anaemia / ↓CO | Step 4: ↓DO₂ | Haemorrhage, cardiogenic shock |
| Mitochondrial dysfunction | Step 5→6: ↑venous O₂ (can't extract) | Cyanide poisoning, sepsis |
The cascade illustrates why hypoxia at tissue level can result from failure at any step — from environmental to cellular — and guides targeted therapy.
Sources: Murray & Nadel's Textbook of Respiratory Medicine; Harrison's Principles of Internal Medicine 22E; Costanzo Physiology 7th Edition; Goodman & Gilman's Pharmacological Basis of Therapeutics
Drugs and equipment used in general anaesthesia and spinal anaesthesia ( intubation tray)
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Reading File
Searching Images
intubation tray equipment laryngoscope endotracheal tube
Loading Image
Finding Sources
Reading File
I now have comprehensive information from multiple authoritative anaesthesia textbooks. Let me compose the complete answer.
Drugs & Equipment in General Anaesthesia and Spinal Anaesthesia
PART 1: GENERAL ANAESTHESIA
General anaesthesia involves three phases: induction → maintenance → reversal/emergence.
A. Drugs Used in General Anaesthesia
1. Induction Agents (IV)
| Drug | Dose | Key Features | Caution |
|---|---|---|---|
| Propofol | 1.5–2.5 mg/kg IV | Most commonly used; antiemetic; bronchodilator; rapid onset | ↓BP 25–40%; central apnoea; pain on injection |
| Ketamine | 1–2 mg/kg IV or 4–6 mg/kg IM | Dissociative; ↑BP, HR, CO; bronchodilator; analgesic; preserves airway reflexes | Hallucinations/emergence delirium; ↑salivation (give glycopyrrolate) |
| Etomidate | 0.2–0.3 mg/kg IV | Haemodynamically neutral; preferred in CV compromise, ↑ICP, trauma | Adrenocortical suppression; myoclonus; PONV; pain on injection |
| Thiopental | 3–5 mg/kg IV | Barbiturate; rapid onset; ↓ICP | No longer widely available in US; cardiovascular depression |
| Midazolam | 0.05–0.1 mg/kg | Anxiolysis/sedation/amnesia as adjunct; can be sole agent in high dose | Prolonged sedation at induction doses; respiratory depression synergistic with opioids |
2. Maintenance Agents
Inhaled volatile agents:
| Agent | Key Properties |
|---|---|
| Sevoflurane | Smooth induction; least airway irritation; preferred for mask induction |
| Isoflurane | Economical; potent bronchodilator |
| Desflurane | Very rapid emergence; airway irritant — not for mask induction |
| Nitrous oxide (N₂O) | Analgesic adjunct; reduces volatile agent requirements (↓MAC) |
IV maintenance: Propofol infusion (TIVA — total IV anaesthesia), remifentanil infusion
3. Neuromuscular Blocking Agents (NMBAs)
| Class | Drug | Onset | Duration | Notes |
|---|---|---|---|---|
| Depolarising | Succinylcholine | 45–60 s | 10–15 min | Rapid onset; used for RSI/suspected difficult airway |
| Non-depolarising | Rocuronium | 60–90 s (1.2 mg/kg for RSI) | 30–60 min | Reversible with sugammadex |
| Vecuronium | 3–5 min | 25–40 min | Cardiac stability | |
| Cisatracurium | 3–5 min | 40–75 min | Hofmann elimination; preferred in liver/renal failure | |
| Atracurium | 2–3 min | 20–35 min | Histamine release |
Reversal:
- Neostigmine + glycopyrrolate (anticholinesterase; must give with anticholinergic)
- Sugammadex — selective reversal of rocuronium/vecuronium; rapid, complete
4. Opioids (Analgesia / Blunting Laryngoscopy Response)
| Drug | Notes |
|---|---|
| Fentanyl | 1–3 µg/kg; most widely used intraoperative opioid |
| Remifentanil | Ultra-short; infusion only; no accumulation |
| Morphine | Longer duration; suitable for postoperative analgesia |
| Sufentanil | 5–10× more potent than fentanyl |
5. Adjuncts / Premedication
| Drug | Role |
|---|---|
| Midazolam | Anxiolysis, anterograde amnesia |
| Glycopyrrolate / Atropine | Antisialogogue; prevent bradycardia |
| Lidocaine IV | Blunts ↑ICP/BP response to intubation; adjunct to propofol (reduces injection pain) |
| Dexmedetomidine | α₂-agonist; sedation with preserved respiratory drive; used for awake fibreoptic intubation |
| Ondansetron / Dexamethasone | PONV prophylaxis |
| Ephedrine / Phenylephrine | Vasopressors for induction hypotension |
B. Equipment for General Anaesthesia / Intubation Tray
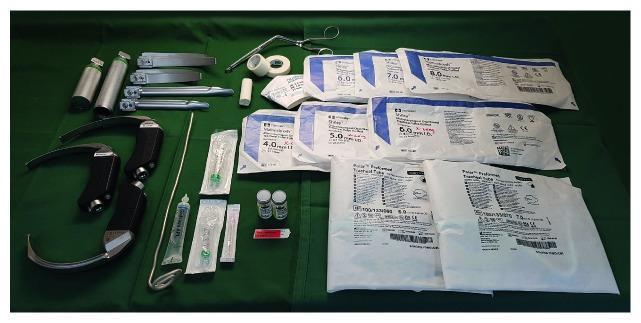
Airway / Intubation Tray Contents
Laryngoscopes:
- Direct laryngoscope with handle + blades:
- Macintosh (curved) — sizes 2, 3 (standard adult), 4 (large adult); tip in vallecula
- Miller (straight) — sizes 2–3; tip lifts epiglottis directly; preferred for paediatrics/anterior larynx
- Video laryngoscope (e.g., GlideScope, McGrath) — for anticipated difficult airway
Endotracheal Tubes (ETTs):
- Adult female: ID 7.0–7.5 mm | Adult male: ID 8.0–8.5 mm
- Cuffed (standard); uncuffed (children <8 y traditionally, though cuffed now accepted)
- Depth of insertion: ~23 cm (F), ~25 cm (M) at lips
Paediatric ETT sizing:
- ETT ID (mm) = (Age/4) + 4 (uncuffed) or (Age/4) + 3.5 (cuffed)
- Depth = ID × 3 cm
| Age | ETT Size | Blade |
|---|---|---|
| Neonate/premature | 3.0 uncuffed | 0 straight |
| 0–6 months | 3.5 cuffed | 1 straight |
| 1–2 years | 4.5 cuffed | 1.5 straight |
| 5–6 years | 5.0 cuffed | 2 curved |
| Adult female | 7.0–7.5 | Mac 3 |
| Adult male | 8.0–8.5 | Mac 3–4 |
Other intubation tray equipment:
- Stylet / bougie (gum elastic bougie) — for difficult/anterior larynx
- Magill forceps — nasotracheal intubation / foreign body removal
- 10 mL syringe — cuff inflation
- Tape / tie — ETT fixation
- Bite block
- Lubricating gel / lignocaine jelly
- Suction (Yankauer catheter)
- Bag-valve-mask (BVM / Ambu bag) with appropriate mask — preoxygenation and rescue ventilation
- Oropharyngeal airways (Guedel) — sizes 0–4 (adult 3–4)
- Nasopharyngeal airway — alternative in semi-conscious patients
Monitoring (before induction):
- SpO₂, ECG, NIBP (minimum)
- Capnography (ETCO₂) — mandatory confirmation of intubation
- Temperature probe, BIS (depth of anaesthesia monitoring)
Anaesthetic Machine:
- Vaporisers, breathing circuit, CO₂ absorber, ventilator
- Functioning suction
- IV access × 2 (minimum large-bore)
PART 2: SPINAL ANAESTHESIA
Spinal (subarachnoid) anaesthesia = injection of local anaesthetic ± adjuncts into the subarachnoid (intrathecal) space.
A. Drugs Used in Spinal Anaesthesia
1. Local Anaesthetics
| Drug | Dose | Baricity | Duration | Notes |
|---|---|---|---|---|
| Bupivacaine (hyperbaric) | 10–15 mg (2–3 mL 0.5%) | Hyperbaric (in dextrose) | 2–4 h | Most commonly used; reliable block |
| Bupivacaine (isobaric) | 10–15 mg | Isobaric | 2–4 h | Less predictable spread |
| Lidocaine | 30–100 mg (2–5% hyperbaric) | Hyperbaric | 1–2 h | Fast onset; transient neurological symptoms risk |
| Ropivacaine | 15–22.5 mg | Isobaric | 2–4 h | Less motor block than bupivacaine |
| Chloroprocaine | 40–60 mg | — | 30–60 min | Short procedures |
| Tetracaine | 5–20 mg | Hyperbaric or hypobaric | 2–4 h | Traditional; slower onset |
Baricity determines spread:
- Hyperbaric (+ dextrose) → sinks with gravity → predictable, lower spread
- Isobaric → position independent
- Hypobaric (+ sterile water) → rises → useful for prone/lithotomy
2. Adjuncts Added to Spinal Injectate
| Drug | Dose | Effect |
|---|---|---|
| Fentanyl | 10–25 µg | ↑ block quality; ↑ duration of analgesia; reduces LA dose needed |
| Morphine (preservative-free) | 0.1–0.3 mg | Prolonged postoperative analgesia (up to 24 h); risk of delayed respiratory depression |
| Sufentanil | 5–7.5 µg | Rapid, dense analgesia |
| Epinephrine | 0.1–0.2 mg | Prolongs block; vasoconstricts; ↑ quality of block (esp. with short-acting LAs) |
| Clonidine | 15–45 µg | α₂-agonist; prolongs sensory/motor block; off-label |
| Neostigmine | 10–50 µg | Prolongs analgesia; more nausea |
3. Vasopressors (for Post-Spinal Hypotension)
| Drug | Notes |
|---|---|
| Phenylephrine | Now first-line for spinal hypotension; α₁-agonist; ↓HR (reflex bradycardia) |
| Ephedrine | Mixed α/β; preserves HR; historical first-line; more fetal acidosis than phenylephrine |
| Norepinephrine | Emerging alternative; maintains CO better than phenylephrine |
| Atropine / Epinephrine | High spinal / bradycardia / cardiac arrest |
B. Equipment for Spinal Anaesthesia
| Item | Detail |
|---|---|
| Spinal needle | Preferred: pencil-point (Whitacre, Sprotte) — atraumatic, ↓PDPH risk; 24–27 gauge. Quincke (cutting bevel) — 22–25 G, more PDPH |
| Introducer needle | Used to guide small-gauge pencil-point needles through skin/ligament |
| Syringe | 5 mL (for local anaesthetic) + 2 mL (for skin LA) |
| Skin cleaning | Chlorhexidine (preferred) or povidone-iodine; sterile field |
| Sterile drape + gloves | Full aseptic technique |
| Local anaesthetic for skin | 1% lignocaine for skin wheal |
| Drug ampoules | Hyperbaric bupivacaine 0.5%; opioid additives; vasopressor drawn up |
| IV cannula + fluids | Mandatory — for fluid co-loading / vasopressor administration |
| Patient position aids | Pillow for lateral decubitus; C-spine support; stool for sitting position |
| Monitoring | SpO₂, ECG, NIBP — continuous throughout |
| Resuscitation equipment | Must be immediately available: airway kit, BVM, epinephrine, atropine, vasopressors |
| Block height assessment | Ice/cold spray, pinprick, light touch |
Dermatomal levels required:
| Surgery | Minimum level |
|---|---|
| Perianal / perineum | S2–5 |
| Lower limb | L2–3 |
| Hips | L1–2 |
| Lower abdomen / TURP | T10 |
| Caesarean section | T4 (nipple line) |
| Upper abdomen | T4–6 (with caution) |
Key Differences Summary
| Feature | General Anaesthesia | Spinal Anaesthesia |
|---|---|---|
| Airway required | Yes — intubation/LMA | No (unless high block or emergency) |
| Consciousness | Lost | Preserved (usually with sedation) |
| Main drugs | Propofol/ketamine/etomidate + NMBA + volatile agent | Hyperbaric bupivacaine ± fentanyl/morphine |
| Duration limit | Unlimited | ~2–4 h (single shot) |
| Major complications | Aspiration, failed intubation, awareness | High spinal, PDPH, hypotension, TNS |
| Ideal for | All surgery types | Lower limb, pelvic, caesarean, urological |
Sources: Miller's Anesthesia 10e; Morgan & Mikhail's Clinical Anesthesiology 7e; Cummings Otolaryngology; Sabiston Textbook of Surgery; Barash Clinical Anesthesia 9e; Tintinalli's Emergency Medicine
Oxygen dissociation curve with Clinical correlation
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Loading Image
Loading Image
Loading Image
Reading File
Finding Sources
Finding Sources
Loading Image
Reading File
Finding Sources
Reading File
Reading File
Loading Image
Reading File
Excellent — I have comprehensive textbook content and all necessary images. Let me now compose the full answer.
Oxygen Dissociation Curve — With Clinical Correlations
1. The Curve: What It Is
The oxyhaemoglobin dissociation curve (ODC) describes the relationship between the partial pressure of oxygen (PO₂) in blood and the percentage saturation of haemoglobin with oxygen (SaO₂). It is sigmoid (S-shaped), not a straight line — a direct consequence of the cooperative binding of O₂ to haemoglobin's four haem groups.
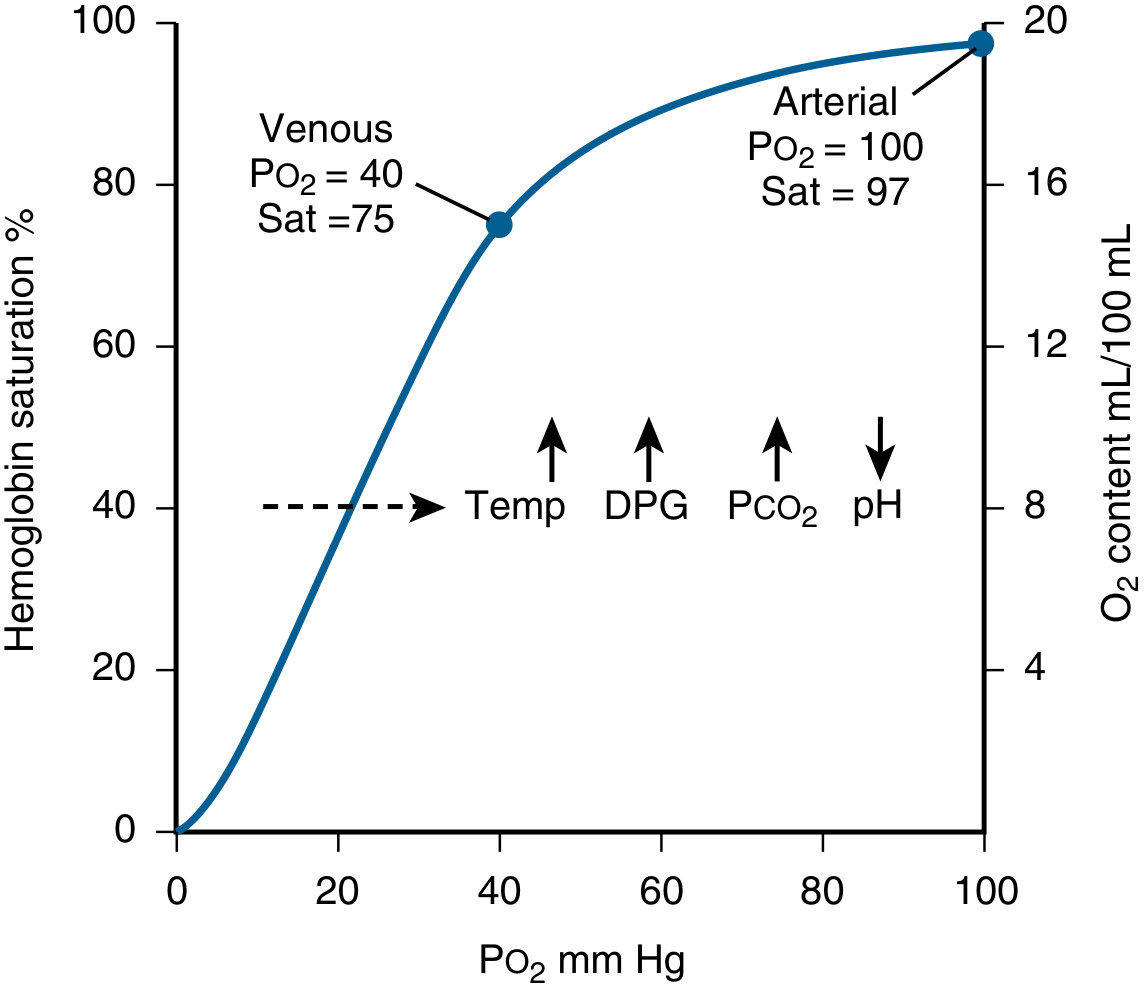
2. Key Values (Normal, at pH 7.4, 37°C)
| Parameter | Value |
|---|---|
| Normal arterial PO₂ | ~100 mmHg → SaO₂ ~97% |
| Mixed venous PO₂ | ~40 mmHg → SvO₂ ~75% |
| P50 (PO₂ at 50% saturation) | 26.5 mmHg |
| O₂ capacity (Hb 15 g/dL) | ~20.8 mL O₂/100 mL blood |
| O₂ content formula | (1.39 × Hb × SaO₂) + (0.003 × PaO₂) |
| Dissolved O₂ only | 0.003 mL/100 mL/mmHg |
Each gram of haemoglobin carries 1.34–1.39 mL O₂ when fully saturated. Normal resting O₂ extraction is ~25% (only one-quarter of delivered O₂ is used at rest).
3. The Sigmoid Shape: Two Physiologically Critical Segments
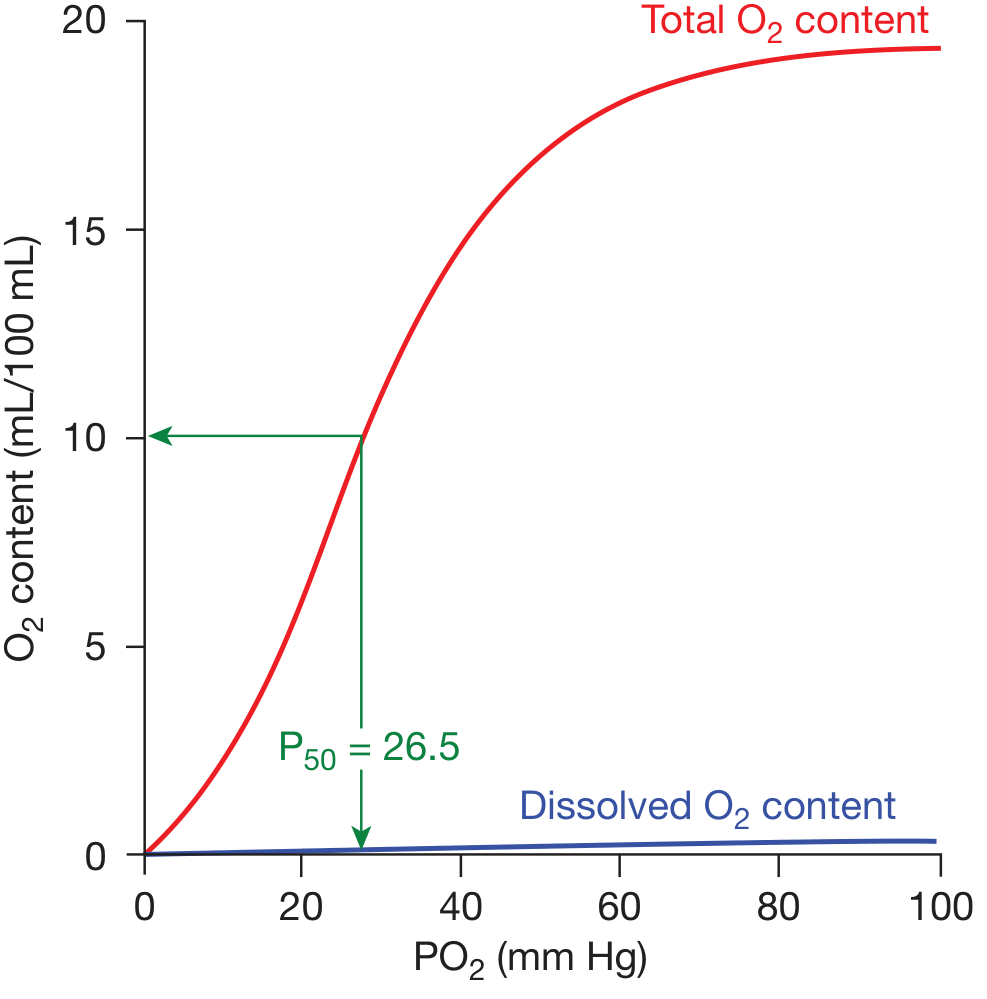
Upper Flat Portion (PO₂ 60–100 mmHg)
- Small drop in PaO₂ (e.g., 100 → 70 mmHg) causes only minor fall in SaO₂
- Clinical advantage: Patients with mild/moderate hypoxaemia (PaO₂ 60–100) remain reasonably saturated — a "safety margin"
- Clinical trap: Pulse oximetry appears reassuringly normal even with significant PaO₂ fall — e.g., PaO₂ can drop from 100 → 60 mmHg while SpO₂ only falls from 97% → 90%
Lower Steep Portion (PO₂ 20–60 mmHg)
- A small drop in PO₂ releases large amounts of O₂ to tissues
- Facilitates efficient unloading at tissue capillaries (PO₂ 40 → 20 mmHg)
- Maintains adequate diffusion gradient into mitochondria
4. Shifts of the Curve
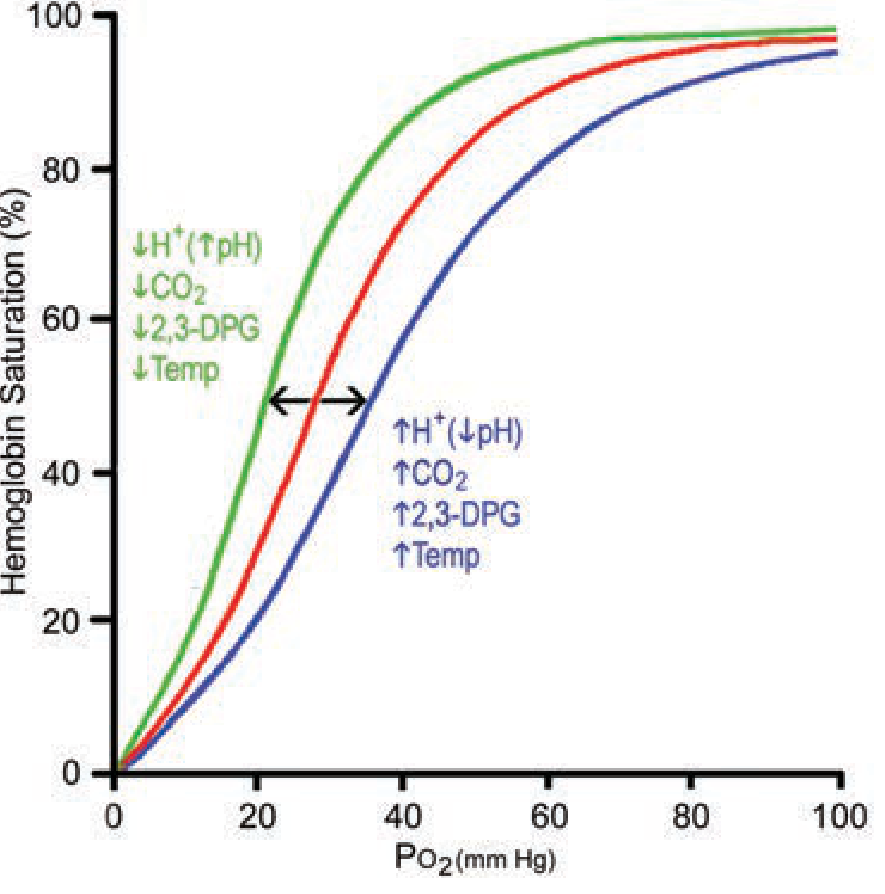
Right Shift (↑P50 — decreased O₂ affinity → more O₂ released to tissues)
| Factor | Mechanism | Mnemonic |
|---|---|---|
| ↑Temperature | Disrupts Hb-O₂ bonds | CADET |
| ↑PCO₂ | Bohr effect | CO₂ |
| ↓pH (↑H⁺) | Bohr effect | Acidosis |
| ↑2,3-DPG | Binds β-chains, stabilises deoxyHb | DPG |
| ↑Altitude (chronic) | ↑2,3-DPG response | Exercise/altitude |
| Anaemia (chronic) | ↑2,3-DPG compensation | Temperature |
Left Shift (↓P50 — increased O₂ affinity → less O₂ released to tissues)
| Factor | Clinical Example |
|---|---|
| ↓Temperature | Hypothermia, stored blood |
| ↓PCO₂ | Hyperventilation (alkalosis) |
| ↑pH | Metabolic alkalosis |
| ↓2,3-DPG | Stored/banked blood (depletes within 7 days) |
| Carboxyhaemoglobin (COHb) | CO poisoning |
| Methaemoglobin | Drug-induced (dapsone, nitrites) |
| Fetal haemoglobin (HbF) | Newborns |
| High O₂-affinity Hb variants | Polycythaemia (rare mutations) |
5. The Bohr Effect
The Bohr effect is the right shift of the ODC caused by ↑CO₂ and ↑H⁺:
- At tissues: Metabolising cells produce CO₂ and H⁺ → blood PCO₂ and H⁺ rise → ODC shifts right → O₂ released more readily to tissues
- At lungs: CO₂ diffuses out → blood PCO₂ and H⁺ fall → ODC shifts left → Hb reloads O₂ efficiently from alveolar gas
Quantitatively, the Bohr effect augments O₂ delivery by only 2–3% at rest, but becomes more significant during strenuous exercise when lactic acid production causes a marked acidosis (pH may fall to 7.2 → P50 rises from ~27 to ~38 mmHg).
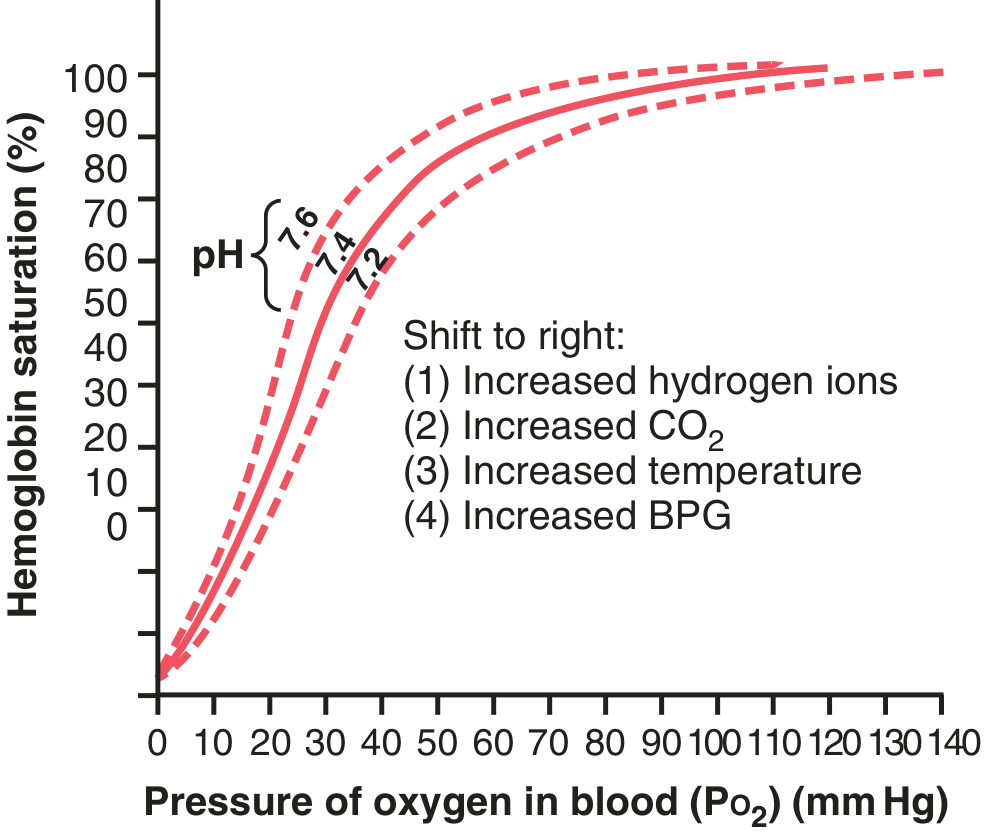
6. 2,3-DPG: Mechanism and Clinical Significance
2,3-DPG (= 2,3-bisphosphoglycerate, 2,3-BPG) is produced by glycolysis in red cells. It:
- Enters the central cavity of deoxyhaemoglobin between the β-chains (electrostatic binding)
- Stabilises the T-state (tense/deoxy) → requires higher PO₂ to load O₂ → right shift
- Normal concentration: ~5 mM
Clinical importance of ↑2,3-DPG (right shift):
- Chronic anaemia → compensatory ↑2,3-DPG → tissues receive more O₂ per gram Hb
- Chronic altitude exposure → ↑2,3-DPG (adaptive)
Clinical importance of ↓2,3-DPG (left shift):
- Stored blood: 2,3-DPG degrades significantly within 7 days of refrigerated storage → left-shifted curve → impaired O₂ unloading to tissues despite transfusion
- Restores to ~50% at 7 hours post-transfusion; ~95% by 48 hours
7. Clinical Correlations
A. Carbon Monoxide (CO) Poisoning
- CO binds Hb with 250× greater affinity than O₂
- COHb has two effects:
- Functional anaemia — CO occupies O₂-binding sites
- Left shift — CO on remaining haem sites increases O₂ affinity of other sites → O₂ won't unload at tissues
- Danger: SpO₂ falsely normal (pulse ox cannot distinguish COHb from OxyHb) — must use co-oximetry
- Treatment: 100% O₂ → displaces CO (half-life: 5 h on room air → 80 min on 100% O₂ → 20 min on hyperbaric O₂)
B. Methaemoglobinaemia
- Fe²⁺ oxidised to Fe³⁺ (cannot carry O₂)
- ↑O₂ affinity of remaining Hb → left shift → tissue hypoxia despite adequate PaO₂
- Causes: dapsone, nitrites, prilocaine, amyl nitrite, phenazopyridine
- SpO₂ reads ~85% regardless of true saturation
- Treatment: methylene blue 1–2 mg/kg IV
C. Fetal Haemoglobin (HbF)
- γ-chains replace β-chains → 2,3-DPG cannot bind effectively
- HbF curve shifted left → P50 ~20 mmHg (vs 27 for HbA)
- Higher O₂ affinity allows fetus to extract O₂ from maternal blood across placenta
- At placenta: fetal blood "steals" O₂ from maternal blood (Bohr effect assists — maternal blood acidifies slightly as it unloads O₂, shifting maternal curve right, facilitating fetal loading)
D. Sickle Cell Disease (HbS)
- HbS has reduced O₂ affinity (right shift)
- At low PO₂ (tissues), deoxyHbS polymerises → sickling → vaso-occlusion and haemolysis
- Voxelotor (new treatment): small molecule that binds α-chain N-terminus → increases O₂ affinity (left shift) → keeps Hb oxygenated longer → reduces sickling
E. Anaemia
- Hb↓ → O₂ content ↓ at any given PaO₂ and SaO₂
- PaO₂ and SpO₂ may be normal, but oxygen content and delivery (DO₂) are reduced
- Compensatory: ↑2,3-DPG (right shift), ↑cardiac output, ↑O₂ extraction
F. Altitude / Chronic Hypoxia
- ↓PaO₂ → immediate: hyperventilation → ↑pH → left shift (transient, impairs unloading)
- Days–weeks: ↑2,3-DPG, ↑erythropoiesis → right shift (adaptive, improves O₂ delivery)
- Chronic mountain dwellers also show ↑Hb and ↑capillary density
G. Transfusion of Stored Blood
- Stored RBCs: ↓2,3-DPG → left-shifted curve → Hb clings to O₂, won't release to tissues
- Especially relevant in massive transfusion, cardiac surgery
- 2,3-DPG recovers within 48 hours post-transfusion
H. Exercise
- Muscle: ↑CO₂, ↑H⁺ (lactic acid), ↑temperature → marked right shift → greatly enhanced O₂ unloading exactly where needed
- Resting: only ~25% O₂ extracted; during maximal exercise: up to 75–85% extracted
I. Hypothermia
- ↓Temperature → left shift → Hb holds O₂ tightly → ↓tissue O₂ delivery
- Clinically relevant in cardiac surgery with cardiopulmonary bypass, near-drowning
- Also causes: ↓O₂ demand (which partly compensates)
8. Summary Table
| Condition | Shift | P50 | Clinical consequence |
|---|---|---|---|
| Exercise, fever, sepsis | Right | ↑ | ↑ O₂ delivery to active tissues |
| Chronic anaemia, altitude | Right | ↑ | Compensatory ↑ unloading |
| CO poisoning | Left | ↓ | O₂ loads but won't unload |
| Methaemoglobinaemia | Left | ↓ | SpO₂ misleading; tissue hypoxia |
| Stored blood | Left | ↓ | Impaired O₂ release post-transfusion |
| HbF (fetus) | Left | ↓ | Facilitates placental O₂ transfer |
| HbS (sickle cell) | Right | ↑ | Sickling at low PO₂ |
| Hypothermia | Left | ↓ | ↓ O₂ delivery to tissues |
| Hyperventilation / alkalosis | Left | ↓ | ↑ Hb-O₂ affinity |
| Acidosis / hypoventilation | Right | ↑ | ↑ O₂ release (Bohr effect) |
Sources: Fishman's Pulmonary Diseases and Disorders; Murray & Nadel's Respiratory Medicine; Guyton & Hall Medical Physiology; Mulholland & Greenfield's Surgery
Oxygen flux and Bain cicuits
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Loading Image
Searching Images
Bain circuit Mapleson D coaxial anaesthesia breathing system diagram
Reading File
Finding Sources
Loading Image
Loading Image
Finding Sources
I now have all the content and images needed. Let me compose the comprehensive answer.
Oxygen Flux and the Bain Circuit
PART 1: OXYGEN FLUX (Oxygen Delivery — DO₂)
1. Definition
Oxygen flux (also called oxygen delivery, DO₂) is the rate at which oxygen is transported from the lungs to the systemic microcirculation per unit time. It is the product of cardiac output (CO) and arterial oxygen content (CaO₂).
2. The Core Formula
DO₂ = CO × CaO₂ × 10
Where the ×10 converts CaO₂ from mL/dL to mL/L so units are consistent.
Normal DO₂ = ~1000 mL/min (at rest, average adult)
3. Components of DO₂
A. Cardiac Output (CO)
CO = Heart Rate (HR) × Stroke Volume (SV)
Determinants of stroke volume:
| Factor | Description |
|---|---|
| Preload | End-diastolic volume (Frank-Starling law) |
| Contractility | Intrinsic myocardial force independent of load |
| Afterload | Systemic vascular resistance (SVR) — force against which ventricle ejects |
Normal CO = 5 L/min
B. Arterial Oxygen Content (CaO₂)
CaO₂ = (Hb × 1.34 × SaO₂) + (0.003 × PaO₂)
| Component | Contribution | Notes |
|---|---|---|
| Hb-bound O₂ | ~19.8 mL/dL | Dominant — Hb 15 g/dL × 1.34 × 0.98 |
| Dissolved O₂ | ~0.3 mL/dL | Tiny at normal PaO₂ (~100 mmHg) |
Normal CaO₂ = 17–20 mL/dL
So: DO₂ = 5 L/min × 20 mL/dL × 10 = 1000 mL/min ✓
4. Oxygen Consumption (VO₂) and Extraction
VO₂ = CO × (CaO₂ − CvO₂) × 10 (Fick principle)
Normal VO₂ = ~250 mL/min (25% of DO₂ at rest)
Oxygen Extraction Ratio (OER) = VO₂ / DO₂ = ~25%
The myocardium has the highest extraction ratio at ~75% — it has almost no oxygen reserve.
Mixed venous saturation (SvO₂) reflects the global balance:
SvO₂ = 1 − (VO₂/DO₂) → Normal: 60–80% Central venous saturation (ScvO₂) normal: 65–85%
5. Normal Haemodynamic Parameters Table
| Parameter | Equation | Normal |
|---|---|---|
| DO₂ | CO × CaO₂ × 10 | 950–1150 mL/min |
| CaO₂ | 1.34 × Hb × SaO₂ + 0.003 × PaO₂ | 17–20 mL/dL |
| VO₂ | CO × (CaO₂ − CvO₂) × 10 | 200–300 mL/min |
| OER | VO₂ / DO₂ | 22–30% |
| SvO₂ | Pulmonary artery (mixed venous) | 60–80% |
6. DO₂ vs VO₂ Relationship: The Critical DO₂ Threshold
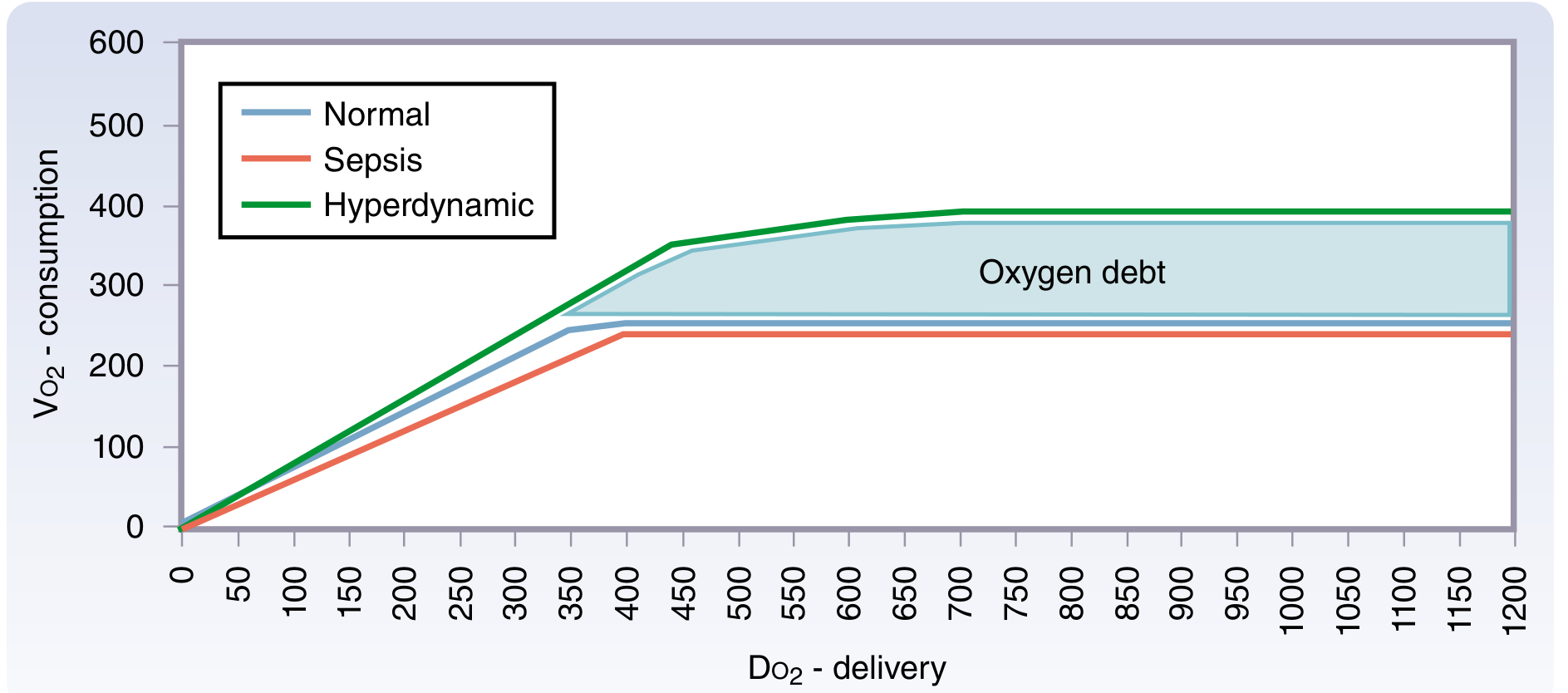
Two physiological zones:
| Zone | DO₂ level | VO₂ behaviour | State |
|---|---|---|---|
| Supply-independent | DO₂ > critical threshold (~400 mL/min) | VO₂ constant despite ↑DO₂ | Normal |
| Supply-dependent | DO₂ < critical threshold | VO₂ falls proportionally with DO₂ | Shock / anaerobic metabolism |
In the supply-dependent zone, cells switch to anaerobic metabolism → lactic acid accumulates → oxygen debt.
During recovery, VO₂ rises above normal (hyperdynamic phase) to repay the oxygen debt — seen classically in post-resuscitation sepsis.
7. Causes of Reduced DO₂
| Mechanism | Examples |
|---|---|
| ↓ Cardiac output | Cardiogenic shock (MI), hypovolemia, cardiac tamponade, PE |
| ↓ Haemoglobin | Haemorrhage, haemolysis, chronic anaemia |
| ↓ SaO₂ | Respiratory failure, V/Q mismatch, shunt |
| ↓ PaO₂ | Hypoventilation, diffusion limitation, altitude |
| CO poisoning | COHb: reduces O₂ carrying capacity AND impairs unloading |
| Sepsis | Maldistribution of flow; microcirculatory failure despite normal global DO₂ |
8. Clinical Monitoring of Oxygen Flux
| Marker | Significance |
|---|---|
| SvO₂ < 60% | ↑ Extraction → supply-demand mismatch |
| ScvO₂ < 65% | Surrogate for SvO₂; used in Surviving Sepsis Bundle |
| Lactate > 2 mmol/L | Anaerobic metabolism → inadequate DO₂ |
| Base deficit | Cumulative oxygen debt |
| Near-infrared spectroscopy (NIRS) | Regional tissue saturation (cerebral, somatic) |
PART 2: THE BAIN CIRCUIT
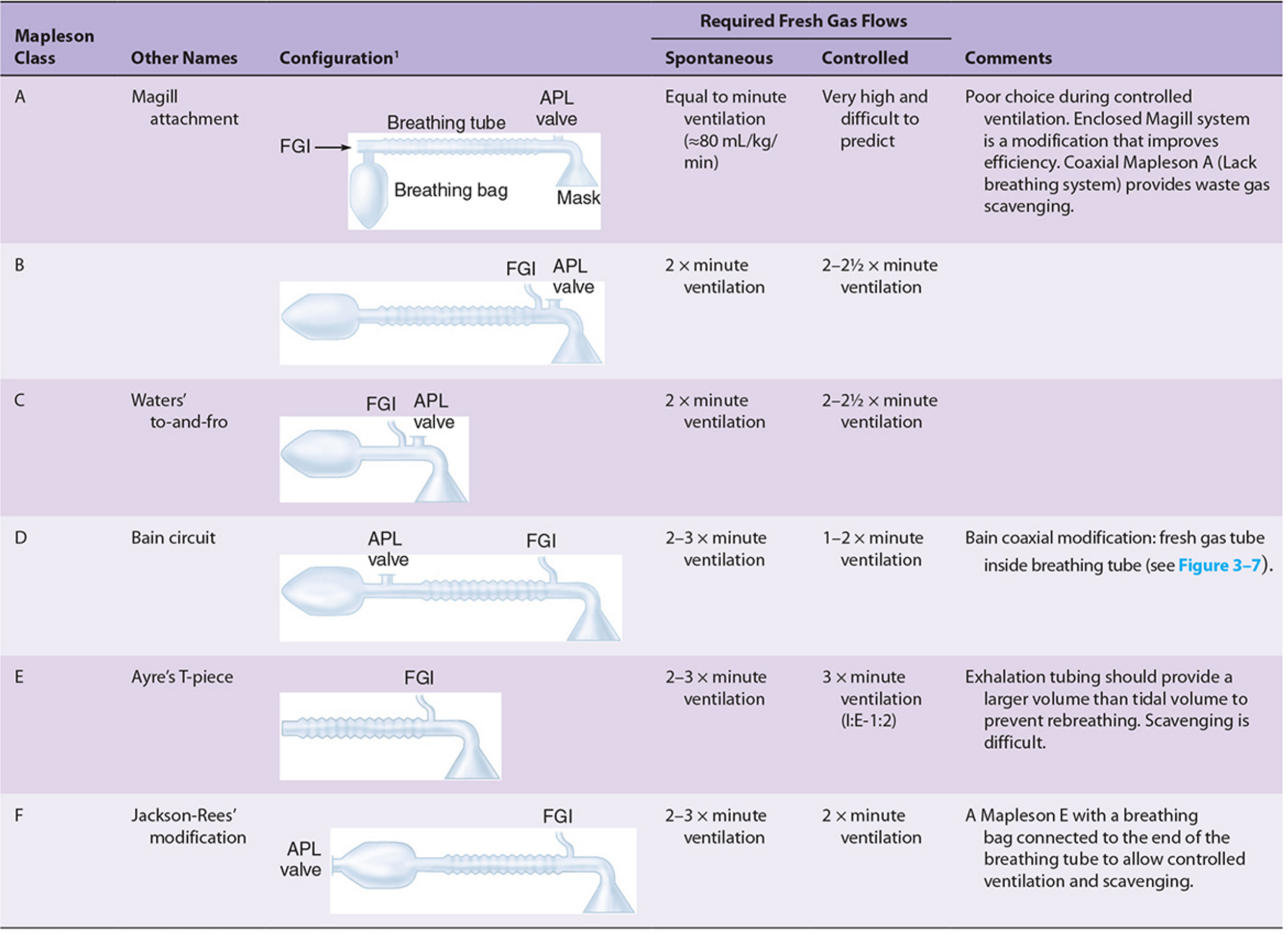
1. Background — Mapleson Classification
Mapleson (1954) classified breathing systems (A–F) based on the relative positions of the Fresh Gas Inlet (FGI) and the APL (Adjustable Pressure-Limiting) valve. The position of these components determines efficiency and the required fresh gas flow (FGF) to prevent CO₂ rebreathing.
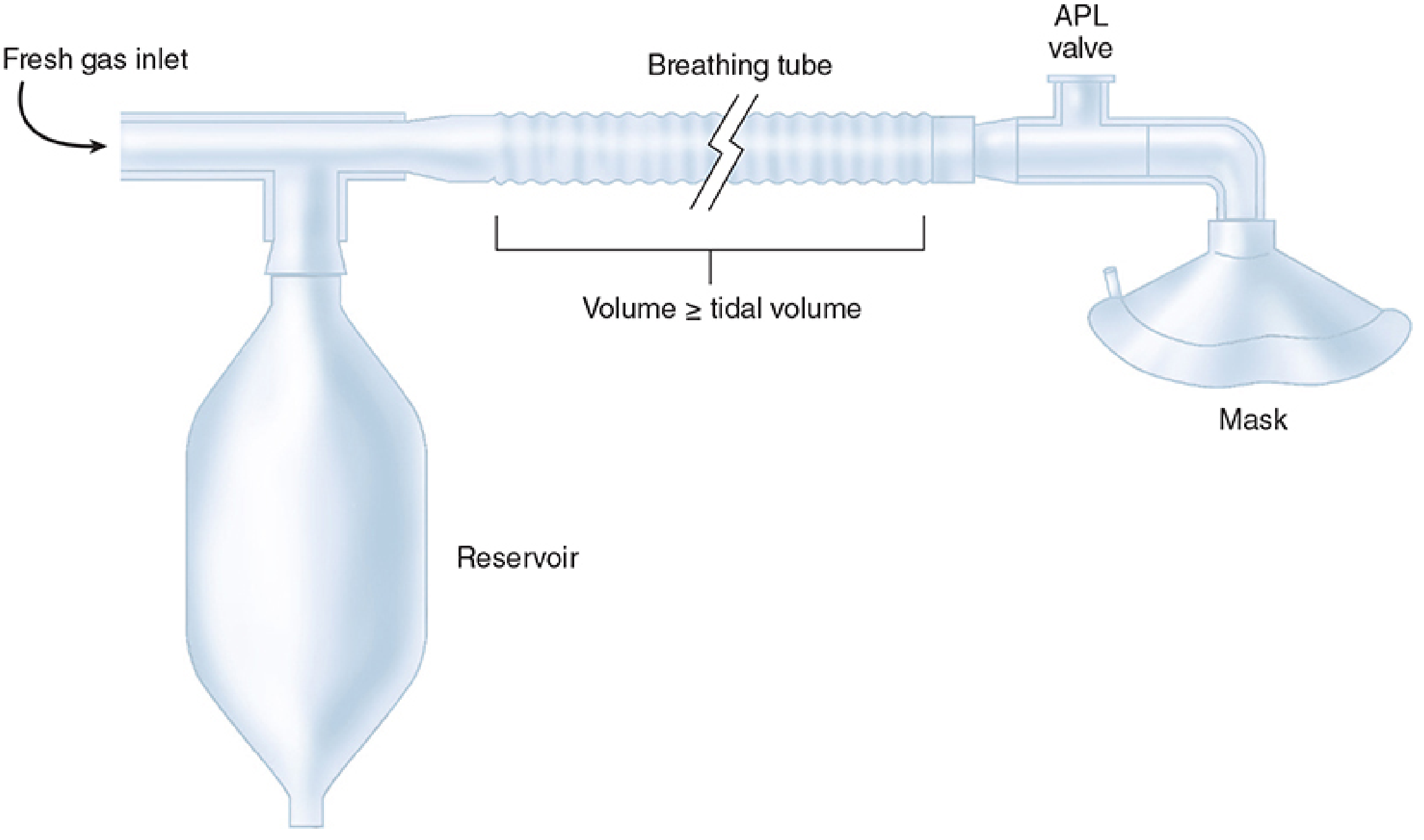
2. The Bain Circuit — Description
The Bain circuit (1972, Bain & Spoerel) is a coaxial modification of the Mapleson D system:
- Inner tube (narrow): carries fresh gas from the machine → empties at the patient end
- Outer tube (wide corrugated): carries exhaled gases away from the patient → vented via the APL (pop-off) valve near the reservoir bag (machine end)
[Reservoir bag] — [APL valve] ←←←←← EXHALED gas (outer tube) ←←←←← [Patient]
←←←←← FRESH gas (inner tube) ←←←←←←←←
The fresh gas enters the inner tube at the machine end but exits at the patient end — the opposite direction to exhalation.
3. Components
| Component | Description |
|---|---|
| Outer corrugated tube | ~22 mm diameter; exhaled gas reservoir; transparent for inner tube inspection |
| Inner narrow tube | ~7 mm diameter; carries fresh gas to patient end |
| Reservoir bag | 2–3 L; acts as gas reservoir and visible ventilation monitor |
| APL valve (pop-off) | Located at machine/bag end; vents excess gas; set open during spontaneous breathing |
| Patient connector (Y-piece) | Connects to mask, ETT, or LMA |
4. Fresh Gas Flow Requirements
No CO₂ absorber → rebreathing is prevented entirely by adequate fresh gas flow:
| Mode | Required FGF | Notes |
|---|---|---|
| Spontaneous ventilation | 2–3 × minute ventilation | Fresh gas pushes exhaled CO₂ toward APL valve |
| Controlled (IPPV) ventilation | 70 mL/kg/min or 1.5–2 × minute ventilation | Fresh gas sweeps CO₂ away; more efficient during IPPV |
For an average 70 kg adult (minute ventilation = 70 mL/kg/min × 70 = 4.9 L/min):
- Spontaneous: FGF ~10–12 L/min
- Controlled: FGF ~4.9–7 L/min
The Bain circuit is more efficient during controlled ventilation (like all Mapleson D systems) because positive pressure during inspiration flushes alveolar gas toward the APL valve.
5. Advantages of the Bain Circuit
| Advantage | Explanation |
|---|---|
| Lightweight and portable | Compact coaxial design |
| Convenient for remote/shared airway (ENT, dental) | APL valve at machine end, away from surgical field |
| Easy scavenging | Pop-off valve remote from patient — simple scavenger attachment |
| Heat and humidity conservation | Expired gases in outer tube warm incoming fresh gas by countercurrent exchange |
| Disposable | Single-use versions available; reduces cross-infection |
| Suitable for both spontaneous and controlled ventilation | Versatile use |
| Minimal apparatus dead space | Fresh gas delivered right at the patient end |
6. Disadvantages and Hazards
| Hazard | Consequence | Prevention |
|---|---|---|
| Kinking of inner tube | Fresh gas not delivered → hypercapnia (CO₂ rebreathing from outer tube) | Inspect outer transparent tube before use |
| Disconnection of inner tube | Same — outer tube becomes dead space | Pethick test |
| High FGF needed | Wasteful; drying/cooling if humidification lost | Ensure FGF formula followed |
| Obstructed filter | ↑ Respiratory resistance mimicking bronchospasm | Check filter |
| Awareness if FGF inadequate | Insufficient anaesthetic agent delivery | Monitor ETCO₂ and volatile agent concentration |
7. Pethick Test (Integrity Test of Inner Tube)
Used to confirm the inner fresh gas tube is intact and patent:
- Occlude the patient end of the circuit
- Apply high-flow oxygen via the machine until the reservoir bag is fully inflated
- Open the patient end — flush oxygen at high flow into the circuit (O₂ flush button)
- Observe the reservoir bag:
- Bag deflates → inner tube intact (Venturi effect at patient end reduces circuit pressure)
- Bag stays inflated → inner tube has a leak (fresh gas escaping into expiratory limb)
8. Comparison: Bain vs Other Systems
| Feature | Mapleson A | Bain (Mapleson D) | Circle system |
|---|---|---|---|
| Best for | Spontaneous ventilation | Controlled ventilation | Long procedures, low flow |
| FGF (spontaneous) | = MV | 2–3 × MV | 0.5–1 L/min |
| FGF (controlled) | >3 × MV (inefficient) | 1.5–2 × MV | 0.5–1 L/min |
| CO₂ absorber | No | No | Yes (soda lime) |
| Heat/humidity | Minimal | Partial (countercurrent) | Good |
| Portability | Moderate | High | Low (heavy machine) |
| Scavenging | Moderate | Easy | Good |
| Paediatric use | Mapleson F preferred | Yes | Yes (with modifications) |
9. Clinical Uses of the Bain Circuit
- ENT, head and neck, dental surgery — surgical field at airway; APL valve at machine end is out of the way
- Short procedures requiring controlled ventilation (e.g., day surgery, minor procedures)
- Transfer/transport anaesthesia — portable and lightweight
- Spontaneous breathing with higher FGF (less common — Mapleson A more efficient for this)
- Paediatrics (Mapleson F/Jackson-Rees preferred but Bain used in older children)
Summary
| Topic | Key Point |
|---|---|
| DO₂ formula | CO × CaO₂ × 10; normal ~1000 mL/min |
| Normal VO₂ | ~250 mL/min (25% extraction) |
| Critical DO₂ | ~400 mL/min; below this → supply-dependent VO₂ → anaerobic metabolism |
| Bain = | Coaxial Mapleson D modification |
| Fresh gas direction | Inner tube → patient end |
| Exhaled gas direction | Outer tube → APL valve (machine end) |
| FGF (controlled) | 70 mL/kg/min or 1.5–2× MV |
| FGF (spontaneous) | 2–3× MV |
| Key hazard | Inner tube kink/disconnect → rebreathing |
| Safety test | Pethick test (reservoir bag deflates = tube intact) |
Sources: Harrison's Principles of Internal Medicine 22E; Sabiston Textbook of Surgery; Mulholland & Greenfield's Surgery 7e; Morgan & Mikhail's Clinical Anesthesiology 7e; Barash Clinical Anesthesia 9e; Miller's Anesthesia 10e
This is a shared conversation. Sign in to Orris to start your own chat.