Mechanism of production of concentrated urine 10 mark answer bds first yr
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Finding Sources
Reading File
Reading File
Reading File
Loading Image
Searching Images
countercurrent multiplier mechanism loop of Henle urine concentration
Loading Image
Loading Image
I now have comprehensive information from Guyton & Hall and Harrison's to write a detailed 10-mark BDS first-year answer. Here it is:
Mechanism of Production of Concentrated Urine
— Guyton & Hall Textbook of Medical Physiology; Harrison's Principles of Internal Medicine
Introduction
The kidneys can produce urine with an osmolarity as high as 1200–1400 mOsm/L compared to plasma at ~300 mOsm/L. This concentrating ability depends on two fundamental requirements:
- High levels of ADH (Antidiuretic Hormone) — makes distal tubules and collecting ducts permeable to water.
- Hyperosmotic renal medullary interstitium — provides the osmotic gradient to drive water reabsorption.
The hyperosmotic medullary interstitium is created by the countercurrent multiplier mechanism and maintained by the countercurrent exchanger (vasa recta).
Structural Basis
About 25% of nephrons are juxtamedullary nephrons with long loops of Henle and vasa recta that dip deep into the medulla, some reaching the renal papilla. Three structures are essential:
- Loop of Henle — acts as the countercurrent multiplier
- Vasa recta — acts as the countercurrent exchanger
- Collecting ducts — carry urine through the hyperosmotic medulla
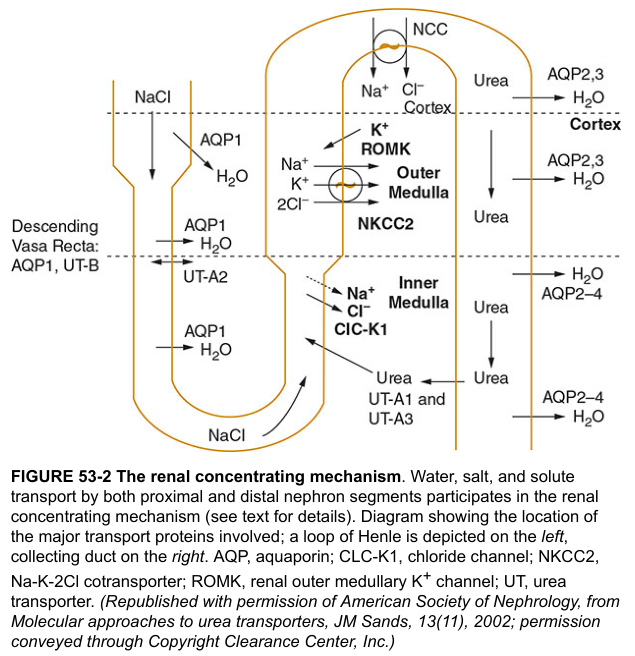
Part 1: Countercurrent Multiplier Mechanism (Loop of Henle)
The loop of Henle establishes medullary hyperosmolarity through differential permeability of its two limbs:
| Segment | Water Permeability | NaCl Permeability | Active Transport |
|---|---|---|---|
| Descending limb | High (AQP1) | Low | None |
| Thin ascending limb | Impermeable | Moderate (passive) | None |
| Thick ascending limb | Impermeable | Low | +++ (NKCC2 pump) |
Step-by-step mechanism:
Step 1 — Active NaCl pumping from thick ascending limb:
The thick ascending limb actively transports Na⁺, K⁺, and Cl⁻ out via the NKCC2 (Na-K-2Cl) cotransporter into the medullary interstitium, creating a ~200 mOsm/L gradient. Because this limb is impermeable to water, water cannot follow — solute accumulates in the interstitium.
Step 2 — Water extraction from descending limb:
The descending limb is highly permeable to water (via AQP1 channels). As fluid flows down, water diffuses OUT into the now-hyperosmotic interstitium, progressively concentrating the tubular fluid. By the tip of the loop, fluid osmolarity reaches ~1200 mOsm/L.
Step 3 — Dilution in ascending limb:
As concentrated fluid ascends through the thick ascending limb, continued active NaCl pumping (without water) dilutes the tubular fluid to ~100–140 mOsm/L before it reaches the distal tubule.
Step 4 — Multiplication:
Continuous flow of new fluid from the proximal tubule + continuous pumping in the ascending limb creates a multiplier effect — the single-effect gradient of 200 mOsm/L is multiplied longitudinally to produce an overall cortex-to-papilla gradient of up to 1200 mOsm/L.
Part 2: Role of Urea in Medullary Hyperosmolarity
Urea contributes significantly (~500 mOsm/L at the papillary tip) to medullary osmolarity through urea recycling:
- In the presence of ADH, water is absorbed from the inner medullary collecting duct, concentrating urea in the tubular lumen.
- ADH increases UT-A1 and UT-A3 urea transporters in the inner medullary collecting duct → urea diffuses into the medullary interstitium.
- Some urea re-enters the thin loop of Henle (via UT-A2) and recirculates — each cycle further increases medullary urea concentration.
This recirculation is vital for concentrating nitrogenous waste products when water is scarce.
Part 3: Countercurrent Exchange by Vasa Recta (Preservation of Gradient)
Without a specialized blood supply, the osmotic gradient built by the loop of Henle would be washed away. The vasa recta (hairpin-shaped capillaries) prevent this:
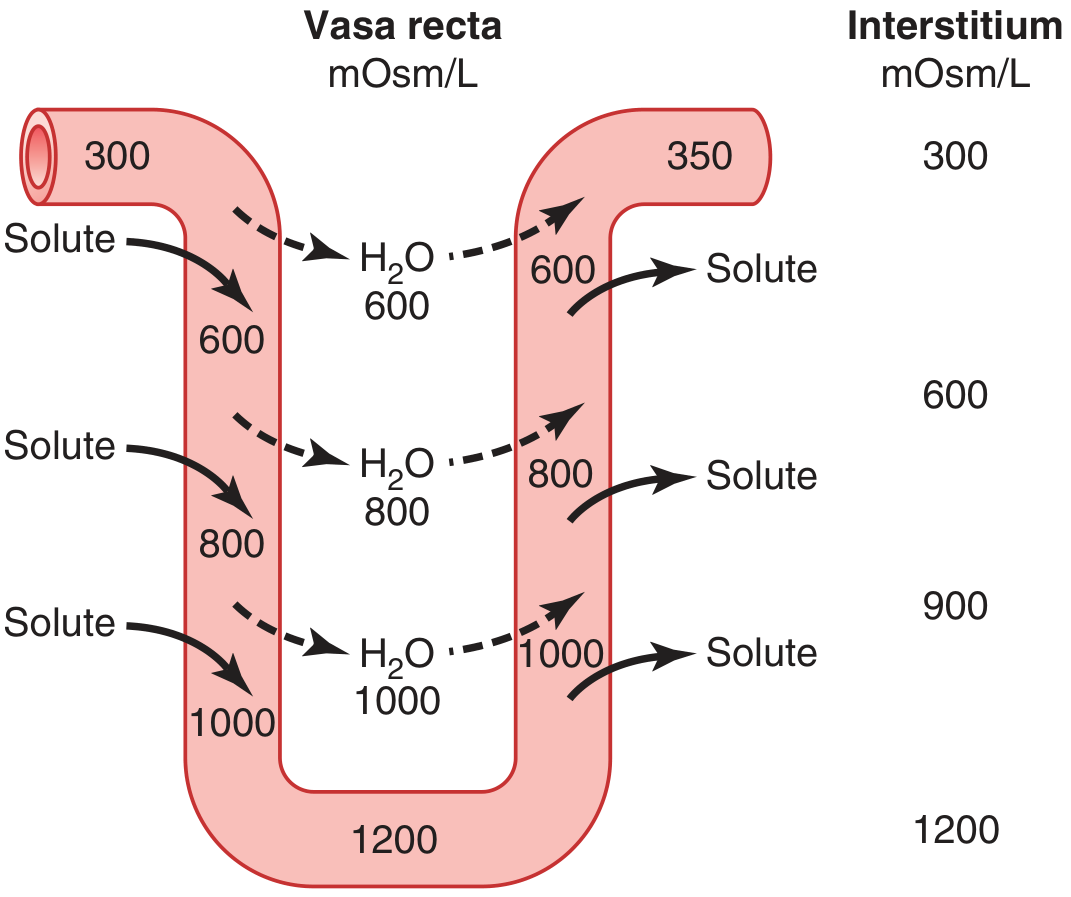
- Descending vasa recta: As blood flows down, it equilibrates with the increasingly hyperosmotic interstitium — solute enters, water leaves → blood reaches ~1200 mOsm/L at the papilla.
- Ascending vasa recta: As blood returns toward the cortex, solute diffuses back into the interstitium, and water re-enters the blood → blood leaves at ~350 mOsm/L.
- Net effect: solute is recycled back to the interstitium rather than being carried away. Medullary blood flow is also slow (<5% of total renal blood flow), minimizing washout.
Part 4: Role of ADH (Antidiuretic Hormone)
ADH (synthesized in hypothalamic supraoptic and paraventricular nuclei; released from posterior pituitary) is the key determinant of final urine concentration:

- ADH binds V2 receptors on collecting duct principal cells → activates adenylate cyclase → ↑ cAMP → PKA activation → aquaporin-2 (AQP2) vesicles fuse with apical membrane → water channel insertion.
- Water exits the cell via basolateral AQP3/AQP4 into the peritubular capillaries.
- Result: tubular fluid equilibrates with the 1200–1400 mOsm/L medullary interstitium → small volume of highly concentrated urine is excreted.
Part 5: Tubular Events — Segment by Segment
| Tubular Segment | Osmolarity of fluid | Key event |
|---|---|---|
| Proximal tubule (leaving) | 300 mOsm/L | Isosmotic reabsorption (80% water + solute) |
| Tip of descending limb | ~1200 mOsm/L | Water exits via AQP1 |
| End of thick ascending limb | ~100–140 mOsm/L | NaCl pumped out; water stays |
| Early distal tubule | ~100 mOsm/L | Further dilution |
| Collecting duct (high ADH) | ~1200–1400 mOsm/L | Water reabsorbed; urine concentrated |
| Final urine | Up to 1400 mOsm/L | Minimal volume excreted |
Summary
Concentrated urine is produced by a four-component system:
- Countercurrent multiplier (loop of Henle) — builds the corticomedullary osmotic gradient (300 → 1200 mOsm/L) by active NaCl transport from the impermeable thick ascending limb while water exits the permeable descending limb.
- Urea recycling — contributes ~500 mOsm/L to inner medullary osmolarity via ADH-dependent UT-A1/A3 transporters and recirculation via UT-A2.
- Countercurrent exchange (vasa recta) — preserves the medullary gradient by returning solute back to the interstitium rather than carrying it into systemic circulation.
- ADH action on collecting ducts — inserts AQP2 water channels, allowing water to move osmotically from the tubular lumen into the hyperosmotic interstitium, producing the final concentrated urine.
The obligatory minimum urine volume = Total solute load (mOsm/day) ÷ Maximum urine osmolarity. At maximum concentrating ability (1200 mOsm/L) and a daily solute load of 600 mOsm, the minimum urine output is ~500 mL/day.
Sources: Guyton and Hall Textbook of Medical Physiology, 14e, pp. 373–379; Harrison's Principles of Internal Medicine, 21e, p. 1386
This is a shared conversation. Sign in to Orris to start your own chat.