Research paper on non alcoholic fatty liver disease
Reading File
Searching PubMed
"non-alcoholic fatty liver disease"[MeSH Terms]
Finding Sources
Reading Article
Reading Article
Finding Sources
Reading Article
Reading Article
Reading File
Searching PubMed
"non-alcoholic fatty liver disease"[MeSH Terms] AND "pathogenesis"[MeSH Terms]
Finding Sources
Searching the Web
NAFLD MASLD 2024 2025 pathogenesis treatment guidelines review
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Now I have comprehensive information from textbooks, PubMed meta-analyses, and the latest guidelines. Let me compile a thorough research paper summary.
Non-Alcoholic Fatty Liver Disease (NAFLD): A Comprehensive Review
Nomenclature note (2023): A multi-society consensus renamed NAFLD to Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD) and NASH to MASH (Metabolic dysfunction-Associated SteatoHepatitis). Both terms are used in this review as the literature is transitioning.
1. Definition and Spectrum
NAFLD/MASLD is defined as the presence of hepatic steatosis in ≥5% of hepatocytes in the absence of significant alcohol use, other known causes of steatosis (e.g., hepatitis C, corticosteroids, tamoxifen, methotrexate, iron overload), plus at least one of five cardiometabolic risk factors (abdominal obesity, pre-diabetes/T2DM, hypertension, hypertriglyceridemia, or low HDL).
The disease spans a spectrum:
| Stage | Description |
|---|---|
| Isolated fatty liver (steatosis) | Fat in >5% of hepatocytes; minimal inflammation; >80% of NAFLD cases |
| NASH/MASH | Steatosis + lobular inflammation + hepatocyte ballooning ± Mallory-Denk bodies |
| Fibrosis | Pericellular/perisinusoidal; staged F0–F4 |
| Cirrhosis | End-stage; ~20% of NASH patients over 15 years |
| Hepatocellular carcinoma (HCC) | Can arise even without cirrhosis in NASH |
- Robbins & Kumar Basic Pathology, p. 609–612
- Sleisenger & Fordtran's GI & Liver Disease, p. 1354–1365
2. Epidemiology
- Global prevalence: ~25–30% of the general adult population; 30–40% in Western nations.
- MASLD affects approximately 60–70% of individuals with T2DM and 70–80% of those with obesity.
- A 2023 meta-analysis in Gut (PMID 37491159) of 156 studies (n=1,832,125) found:
- NAFLD prevalence in T2DM: 65%
- NASH prevalence in T2DM: 31.6%
- Clinically significant fibrosis (F2–F4) in T2DM with NAFLD: 35.5%
- Advanced fibrosis (F3–F4): 15%
- Demographics: More common in males; postmenopausal women have disproportionately advanced disease. Hispanics have the highest ethnic prevalence (~45–58%), followed by Caucasians and African Americans.
- Children: Pediatric NAFLD prevalence is 7.6% in the general population, rising to 34.2% in pediatric obesity clinics.
- NAFLD is now the leading indication for liver transplantation in the USA.
3. Pathogenesis
The original "2-hit" hypothesis (steatosis → oxidative stress → injury) has been replaced by a "multiple parallel hits" model integrating:
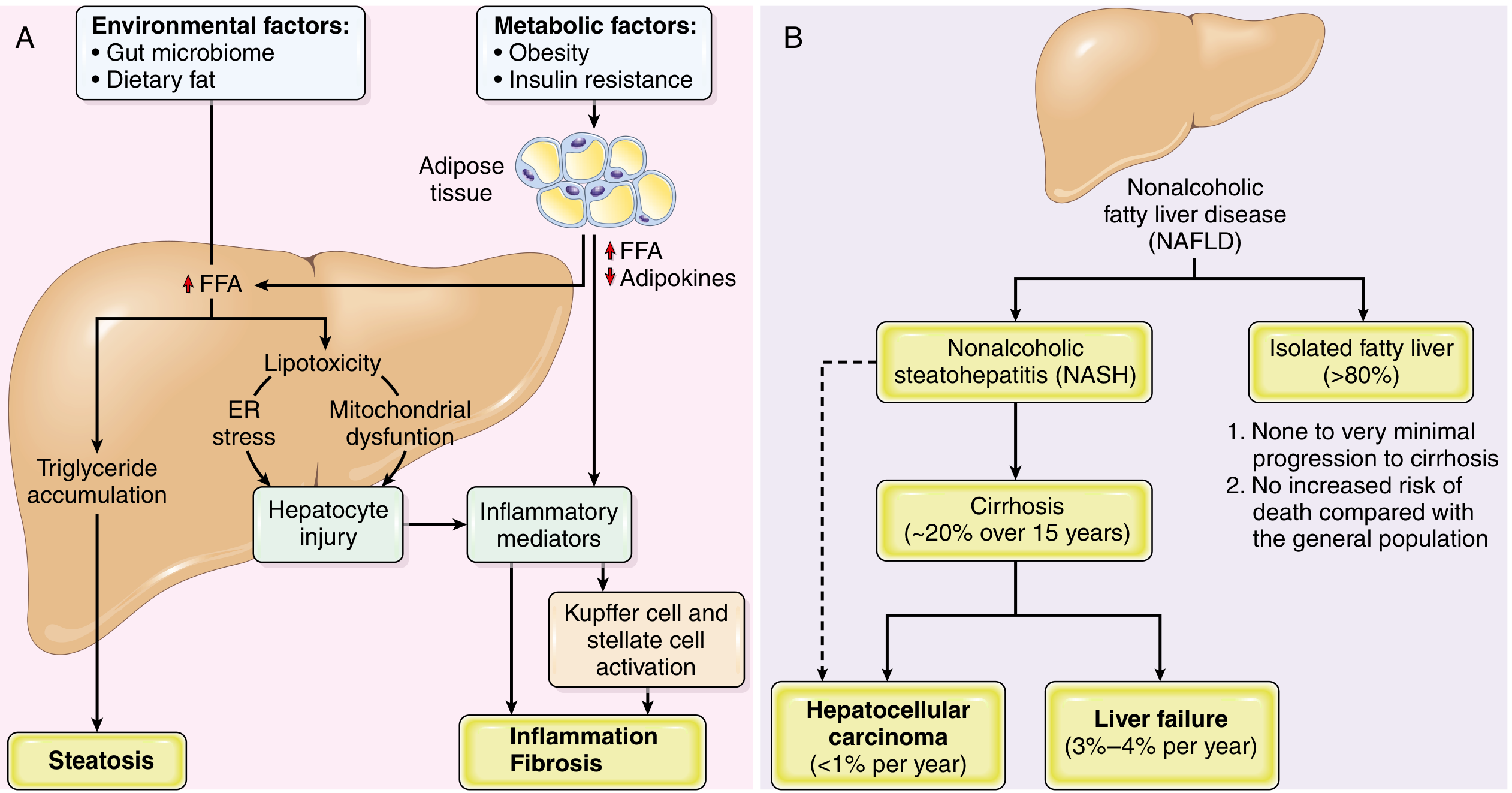
3a. Hepatic Steatosis
- Insulin resistance → impaired suppression of adipose tissue lipolysis → increased free fatty acid (FFA) delivery to the liver
- Increased de novo lipogenesis (DNL) driven by hyperinsulinemia
- FFA esterification → triglyceride accumulation → macrovesicular steatosis
- Impaired VLDL export further traps lipids
3b. Progression to Steatohepatitis (NASH)
- Lipotoxicity: accumulation of toxic lipid species (diacylglycerols, ceramides, free cholesterol) beyond triglycerides
- Mitochondrial dysfunction: impaired β-oxidation, reactive oxygen species (ROS) overproduction
- ER stress: unfolded protein response activation → hepatocyte apoptosis
- Gut-liver axis: dysbiosis → increased intestinal permeability → portal delivery of LPS → Toll-like receptor 4 activation in Kupffer cells
- Kupffer cell activation → TNF-α, IL-1β, IL-6 → stellate cell activation → fibrogenesis
- Dietary fat and gut microbiome composition modulate disease severity
3c. Genetic Susceptibility
A 2023 GWAS meta-analysis in Nature Genetics (PMID 37709864) identified 17 loci associated with NAFLD, including well-established variants in PNPLA3 (rs738409), TM6SF2, MBOAT7, and HSD17B13. These variants alter lipid metabolism, hepatocyte function, and inflammatory signaling.
4. Clinical Features & Diagnosis
Symptoms
Most patients are asymptomatic. Some experience fatigue, malaise, or right upper quadrant discomfort. Advanced disease presents with signs of chronic liver disease (jaundice, ascites, variceal bleeding, encephalopathy).
Laboratory Findings
- Elevated ALT > AST (AST:ALT ratio typically <1, distinguishing it from alcohol-related liver disease where ratio >2)
- Elevated triglycerides, glucose, HbA1c
- Normal or mildly elevated bilirubin
Imaging
| Modality | Role |
|---|---|
| Ultrasound | First-line; detects steatosis >30%; operator-dependent |
| MRI/MR spectroscopy | Gold standard for quantifying hepatic fat |
| Transient elastography (FibroScan) | Noninvasive fibrosis staging (vibration-controlled = VCTE) |
| CT | Less sensitive; radiation concern |
Noninvasive Fibrosis Markers
- FIB-4 index (age × AST / [platelets × √ALT]) — widely used screening tool
- NAFLD Fibrosis Score (NFS)
- Enhanced Liver Fibrosis (ELF) score
Liver Biopsy
Required to confirm NASH and grade/stage disease. Histologic scoring uses the NAS (NAFLD Activity Score): steatosis (0–3) + lobular inflammation (0–3) + hepatocyte ballooning (0–2). NAS ≥5 is consistent with NASH.
5. Natural History & Complications
A 2024 systematic review and meta-analysis in Clinical Molecular Hepatology (PMID 38281814, n=1,377,466 persons) reported incidence rates per 1,000 person-years in NAFLD:
| Event | Incidence (per 1,000 PY) |
|---|---|
| All-cause mortality | 14.6 |
| Cardiovascular mortality | 4.53 |
| Liver-related mortality | 3.10 |
| Fibrosis progression | 49.0 |
| Cirrhosis | 10.9 |
| HCC | 3.39 |
| Cardiovascular disease | 24.77 |
| Type 2 Diabetes (new onset) | 19.0 |
| Renal impairment | 30.3 |
Key findings:
- Cardiovascular disease is the #1 cause of death in NAFLD
- NASH is associated with higher HCC risk than simple steatosis
- HCC incidence is highest in the Western Pacific/Southeast Asia
- Decompensated cirrhosis rates are increasing over time
Isolated fatty liver (without NASH): minimal risk of progression to cirrhosis; mortality comparable to general population.
6. Treatment
6a. Lifestyle Modification (First-Line)
- Weight loss ≥7–10% of body weight improves steatosis; ≥10% can resolve NASH and reduce fibrosis
- Low-carbohydrate or Mediterranean diet: A 2023 systematic review in Nutrients (PMID 37242133) showed Mediterranean diet significantly reduces liver enzymes, central obesity, and lipid levels in NAFLD
- Physical exercise: Both aerobic and resistance training reduce hepatic fat independent of weight loss
- Alcohol abstinence strongly recommended
6b. Bariatric Surgery
- Indicated for BMI >35 with NAFLD
- Consistently improves/resolves NASH and fibrosis in multiple studies
6c. Pharmacotherapy
FDA-Approved (2024)
Resmetirom (Rezdiffra) — thyroid hormone receptor β-selective agonist:
- Phase III MAESTRO-NASH trial (2024): significant NASH resolution and fibrosis improvement vs. placebo
- First FDA-approved drug specifically for MASH with moderate-to-advanced fibrosis (March 2024)
Semaglutide (GLP-1 receptor agonist):
- A 2023 meta-analysis in Diabetes & Metabolic Syndrome (PMID 37717295, n=2,413) showed:
- ↓ ALT by 14.07 U/L (p<0.001)
- ↓ Liver fat content by 4.97% (p<0.001)
- ↓ Liver stiffness by 0.96 kPa (p=0.04)
- ↑ GI adverse events (nausea, vomiting, diarrhea)
- Conditionally FDA-approved (subcutaneous 2.4 mg weekly) for MASH with moderate-to-advanced fibrosis (2024)
Other Agents Under Investigation
| Drug | Class | Status |
|---|---|---|
| Lanifibranor | Pan-PPAR agonist | Phase III |
| Obeticholic acid | FXR agonist | Phase III (concerns re: pruritus, LDL↑) |
| Pioglitazone | Thiazolidinedione | Guideline-supported (off-label) in T2DM with NASH |
| Vitamin E | Antioxidant | Recommended in non-diabetic NASH (AGA) |
| Silymarin | Natural flavonoid | 2024 meta-analysis (PMID 38579127) showed modest ALT/AST reduction |
6d. Liver Transplantation
- NAFLD/NASH is a leading and growing indication for LT in the USA
- Posttransplant survival is comparable to other indications
- Higher waitlist mortality due to cardiovascular and renal comorbidities
- Donor livers with >30% macrovesicular steatosis carry increased graft failure risk
- Yamada's Textbook of Gastroenterology, p. 2139
7. Special Populations
- Pediatric NAFLD: Histologically distinct — portal-predominant inflammation, mononuclear rather than neutrophilic infiltrates; managed with lifestyle modification primarily
- Lean NAFLD: Occurs in ~10–20% of NAFLD patients with BMI <25; may have worse metabolic profile and fibrosis despite normal weight
- Post-transplant NAFLD recurrence: Risk factors include weight gain, metabolic syndrome, immunosuppressants (especially corticosteroids and calcineurin inhibitors)
8. Monitoring & Surveillance
- Periodic FIB-4/NFS to track fibrosis progression
- Liver ultrasound + AFP every 6 months for HCC surveillance in those with NASH cirrhosis
- Cardiology referral — cardiovascular risk assessment is essential
- Screen for T2DM, hypertension, dyslipidemia, CKD, and depression (incidence rate 29.1/1,000 PY)
9. Recent Advances (2024–2026)
A January 2026 JAMA review (PMID 41212550, Tilg et al.) summarizes the landmark year of 2024 for MASLD:
- Resmetirom FDA approval (March 2024) — first-ever disease-specific pharmacotherapy
- Semaglutide conditional approval for MASH
- EASL-EASD-EASO Clinical Practice Guidelines 2024 — updated screening, diagnosis, and management recommendations for MASLD
- Liver stiffness measurement (FibroScan) validated as a surrogate endpoint for liver-related outcomes
- APASL 2025 guidelines providing specific guidance on resmetirom patient selection, monitoring, and emerging drug combinations
- GWAS studies identifying 17+ genetic loci, advancing precision medicine in NAFLD
Key References
| PMID | Study | Key Finding |
|---|---|---|
| 41212550 | Tilg et al., JAMA 2026 | Comprehensive MASLD review; resmetirom & semaglutide approved |
| 38281814 | Le et al., Clin Mol Hepatol 2024 | Global adverse event incidence in NAFLD (n=1.37M) |
| 37491159 | En Li Cho et al., Gut 2023 | NAFLD prevalence 65% in T2DM (n=1.83M) |
| 37717295 | Bandyopadhyay et al., 2023 | Semaglutide reduces liver fat & stiffness in NAFLD/NASH |
| 37709864 | Chen et al., Nat Genet 2023 | 17 GWAS loci for NAFLD identified |
| 37242133 | Del Bo' et al., Nutrients 2023 | Mediterranean diet improves lipids & enzymes in NAFLD |
Textbook sources: Robbins & Kumar Basic Pathology, p. 609–612 | Sleisenger & Fordtran's GI & Liver Disease, p. 1354–1365 | Yamada's Textbook of Gastroenterology, p. 2139 | Harrison's Principles of Internal Medicine 22E
This is a shared conversation. Sign in to Orris to start your own chat.