Therapeutics 2 : synopsis 1. Basic principles of cancer chemotherapeutic agents 2. General introduction of cancer chemotherapeutic agents ( classification, basic moa , indications and common adrs )
Reading File
Finding Sources
Finding Sources
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
This synopsis covers both topics comprehensively. Here's a quick orientation to what's included:
What this synopsis covers:
Part 1 — Basic Principles:
- Nature of cancer and tumor biology
- Cell cycle phases and CCS vs. CCNS drug classification
- Log-kill hypothesis and Gompertzian tumor growth
- Rationale for combination chemotherapy (with classic regimen examples)
- Mechanisms of drug resistance (including P-glycoprotein, target mutations)
- Goals of therapy: curative, control, palliative; adjuvant vs. neoadjuvant vs. induction
- General toxicity profile (bone marrow, GI, gonads, secondary malignancies)
Part 2 — Drug Classes (with MOA, Indications, ADRs):
- Alkylating agents — nitrogen mustards, nitrosoureas, platinum analogs
- Antimetabolites — folate antagonists, pyrimidine analogs, purine analogs
- Natural products — vinca alkaloids, taxanes, topoisomerase I & II inhibitors, antitumor antibiotics
- Targeted therapy — BCR-ABL TKIs (all generations), EGFR inhibitors, checkpoint inhibitors (PD-1/CTLA-4), ADCs
- Hormonal agents — SERMs, AIs, GnRH agonists, androgen-axis blockade
- Miscellaneous — proteasome inhibitors, PARP inhibitors, mTOR inhibitors, IMiDs
The Quick Reference ADR table at the end is especially useful for exams — it groups the most high-yield toxicities by drug.
Gout
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
I now have comprehensive data from Robbins (pathology), Katzung, and Lippincott (pharmacology). I have all I need to write a thorough synopsis.
Gout — Comprehensive Synopsis
1. DEFINITION
Gout is a metabolic disorder characterized by recurrent episodes of acute arthritis caused by deposition of monosodium urate (MSU) crystals in joints, cartilage, and soft tissues, occurring in the setting of hyperuricemia (plasma urate > 6.8 mg/dL).
— Robbins & Kumar Basic Pathology, p. 4380; Katzung's Basic and Clinical Pharmacology, 16th Ed., p. 1028
2. EPIDEMIOLOGY
- Most common in men (95% of cases); women typically affected postmenopause
- Men typically develop gout in their 4th–6th decades
- Asymptomatic hyperuricemia is common (~19% of the US population), but only 1 in 8 patients with urate levels of 7–8 mg/dL develop clinical gout over 14 years
- Risk rises significantly with urate >9 mg/dL or in the presence of comorbidities (CKD, cardiovascular disease, obesity, diabetes)
3. PATHOPHYSIOLOGY
3.1 Uric Acid Metabolism
Uric acid is the end product of purine catabolism in humans (unlike most mammals, which possess uricase to convert uric acid to the more soluble allantoin). Two pathways produce purines:
| Pathway | Description |
|---|---|
| De novo synthesis | Purine nucleotides built from non-purine precursors |
| Salvage pathway | Free purines recycled via HGPRT (hypoxanthine-guanine phosphoribosyltransferase) |
Purines → Hypoxanthine → Xanthine → Uric acid (catalyzed by xanthine oxidase)
3.2 Causes of Hyperuricemia
| Mechanism | Causes |
|---|---|
| Overproduction (10%) | HGPRT deficiency (Lesch-Nyhan syndrome, partial), PRPP synthetase overactivity, tumor lysis syndrome, hemolytic anemia, myeloproliferative disorders |
| Underexcretion (90%) | Idiopathic (most primary gout), chronic kidney disease, drugs (thiazides, loop diuretics, low-dose aspirin, cyclosporine), alcohol, metabolic syndrome |
| Mixed | Alcohol excess, obesity |
Primary gout (90%): Reduced uric acid excretion of unknown cause
Secondary gout: Identifiable cause — drugs, disease states, enzymatic defects
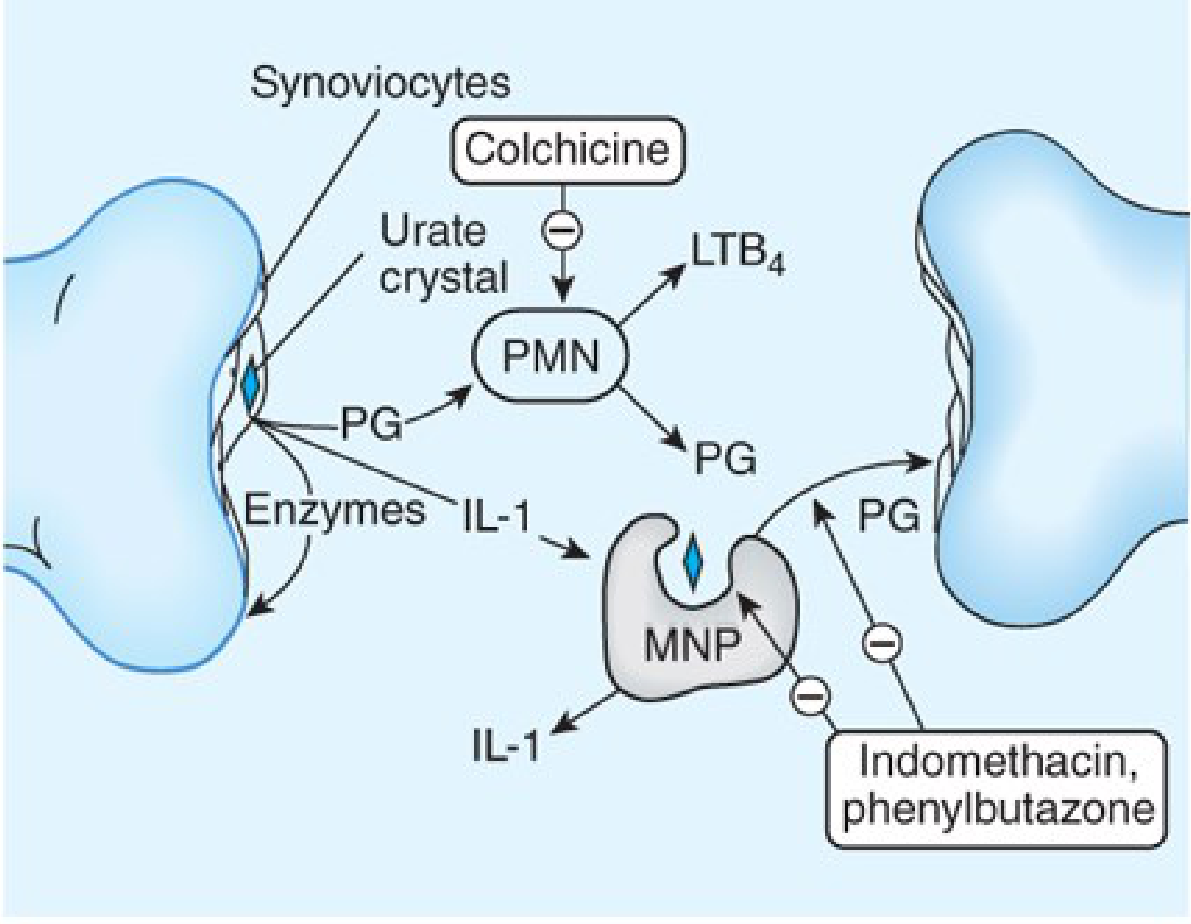
3.3 Crystal-Induced Inflammation
MSU crystals deposited in joints → NLRP3 inflammasome activation → cascade:
- Synoviocytes phagocytose urate crystals → release prostaglandins, IL-1, LTB₂
- Neutrophils recruited into joint space → amplify inflammation (release free radicals, lysosomal enzymes)
- Macrophages appear later → ingest crystals → release further cytokines
- IL-1β is the central mediator: NLRP3 activates caspase-1 → cleaves pro-IL-1β to active IL-1β
- Crystal-induced lysosomal membrane damage → enzyme leakage → tissue injury
— Katzung's Basic and Clinical Pharmacology, 16th Ed., p. 1028
4. CLINICAL STAGES
| Stage | Features |
|---|---|
| Asymptomatic hyperuricemia | Elevated uric acid; no symptoms; most never progress to gout |
| Acute intermittent gout | Sudden, severe monoarticular arthritis; first MTP joint (podagra, ~50%); also ankle, knee, wrist; erythema, warmth, swelling; resolves spontaneously in days–weeks |
| Intercritical gout | Symptom-free periods between attacks |
| Chronic tophaceous gout | Persistent arthritis; tophi (aggregates of MSU crystals + inflammatory tissue) in joints, periarticular tissue, cartilage (helix of ear), tendons; cartilage destruction; joint deformity |
Associations: Uric acid renal calculi, interstitial nephritis, adverse cardiovascular outcomes
5. DIAGNOSIS
5.1 Synovial Fluid Analysis (Gold Standard)
- Needle-shaped MSU crystals that are negatively birefringent under polarized light (yellow when parallel to slow ray of compensator)
- Intracellular crystals in neutrophils during acute attack
5.2 Serum Uric Acid
- Hyperuricemia (>6.8 mg/dL) is necessary but not sufficient
- May be normal during acute attack (crystals precipitate as uric acid drops)
5.3 Imaging
| Modality | Finding |
|---|---|
| X-ray (plain) | "Punched-out" juxta-articular erosions with overhanging edges (late); periarticular soft-tissue swelling (tophi) |
| Ultrasound | "Double contour sign" (urate deposition on cartilage); hyperechoic foci in effusion; tophi as irregular soft-tissue thickening; cortical erosions |
| CT / DECT | Dual-energy CT can directly identify urate deposits |
— Grainger & Allison's Diagnostic Radiology, p. 1125
6. TREATMENT
6.1 Treatment Goals
- Relieve acute gouty attacks rapidly
- Prevent recurrent attacks with prophylactic therapy
- Prevent complications — tophi, nephrolithiasis, nephropathy, joint damage
- Lower serum uric acid to target <6 mg/dL (symptomatic patients)
Indications for urate-lowering therapy:
- ≥2 gouty attacks/year
- Presence of tophi
- Chronic kidney disease
- Uric acid nephrolithiasis
6.2 TREATMENT OF ACUTE GOUT
Three first-line options; choose based on patient comorbidities:
A. NSAIDs
- Indomethacin is the classic choice (50 mg TID × 5–7 days), though all NSAIDs are likely effective
- MOA: Inhibit COX-1/COX-2 → ↓ prostaglandin synthesis → reduce inflammation
- Avoid in: Peptic ulcer, renal impairment, anticoagulation, heart failure
- Start within 24–48 hours of onset for best effect
B. Colchicine
- Plant alkaloid from Colchicum autumnale
- MOA: Binds tubulin → depolymerizes microtubules → impairs neutrophil motility and migration into the inflamed joint; also blocks mitotic spindle (cell division)
- Must be given within 36 hours of attack onset to be effective
- Relieves pain typically within 12 hours
- Dose: Low-dose colchicine (1.2 mg then 0.6 mg one hour later) is as effective as high-dose with fewer GI side effects
- ADRs:
- Acute: Nausea, vomiting, abdominal pain, diarrhea (most common; dose-dependent)
- Chronic: Myopathy, neutropenia, aplastic anemia, alopecia
- Avoid in: Pregnancy; caution with hepatic/renal/cardiovascular disease
- Drug interactions: Metabolized by CYP3A4 and is a P-gp substrate → increased toxicity with CYP3A4 inhibitors (clarithromycin, itraconazole) and P-gp inhibitors (amiodarone, verapamil)
C. Corticosteroids
- Used when NSAIDs and colchicine are contraindicated (e.g., renal impairment)
- Intra-articular corticosteroids when 1–2 joints involved
- Systemic (oral prednisone, IV methylprednisolone) for polyarticular or severe gout
- ADR: Rebound flare on tapering → taper slowly over 2–3 weeks
6.3 URATE-LOWERING THERAPY (CHRONIC GOUT MANAGEMENT)
Important note: Initiating urate-lowering therapy can precipitate an acute attack due to rapid changes in serum urate. Always co-prescribe prophylactic low-dose colchicine or NSAID for ≥6 months when starting these agents.
A. Xanthine Oxidase Inhibitors (First-line)
1. Allopurinol
| Parameter | Detail |
|---|---|
| MOA | Purine analogue (isomer of hypoxanthine); inhibits xanthine oxidase → ↓ conversion of xanthine/hypoxanthine to uric acid → ↓ urate synthesis; substrates (xanthine, hypoxanthine) accumulate but are more soluble |
| Dose | Start 100 mg/day; titrate by 50–100 mg every 2–4 weeks to target uric acid <6 mg/dL; max 800 mg/day |
| Renal dosing | Required — dose-reduce with CKD (oxypurinol metabolite accumulates) |
| ADRs | Rash (common), allopurinol hypersensitivity syndrome (rare but severe: fever, eosinophilia, hepatitis, exfoliative dermatitis/TEN), GI upset |
| Drug interaction | Azathioprine + allopurinol = life-threatening myelosuppression (xanthine oxidase metabolizes azathioprine → inhibition → ↑ azathioprine levels; reduce azathioprine dose by 75%) |
| Also interaction | 6-Mercaptopurine (same interaction as azathioprine) |
2. Febuxostat
| Parameter | Detail |
|---|---|
| MOA | Non-purine xanthine oxidase inhibitor — inhibits both oxidized and reduced forms of the enzyme |
| Dose | 40 or 80 mg/day; no renal dose adjustment needed (unlike allopurinol) |
| Use | Allopurinol-intolerant patients (e.g., allergy, intolerance) |
| ADRs | GI upset, liver function abnormalities, gout flares |
| Cardiovascular warning | A large RCT (CARES trial) showed higher all-cause and CV mortality with febuxostat vs. allopurinol in patients with pre-existing major CVD → avoid in high-CV-risk patients |
| Drug interaction | Same azathioprine/6-MP interaction as allopurinol |
B. Uricosuric Agents
MOA: Inhibit renal tubular reabsorption of urate → ↑ urinary excretion of uric acid
| Drug | Details |
|---|---|
| Probenecid | Inhibits URAT1 transporter in proximal tubule; ensure adequate hydration (prevents uric acid stones); contraindicated in nephrolithiasis; avoid with low-dose aspirin (which competes for excretion); initially may precipitate acute attack |
| Lesinurad | Selective URAT1/OAT4 inhibitor; used as add-on to allopurinol when goal uric acid not achieved |
| Benzbromarone | Used in Europe; potent uricosuric; risk of hepatotoxicity |
C. Recombinant Uricase (Pegloticase)
| Parameter | Detail |
|---|---|
| MOA | Recombinant pegylated uricase → converts uric acid to allantoin (more soluble, readily excreted) — the reaction humans cannot perform |
| Use | Refractory chronic gout unresponsive to conventional therapy |
| FDA approval | 2010 |
| ADRs | Infusion reactions, gout flares (esp. first 3–6 months; prophylaxis with NSAIDs/colchicine required), anti-pegloticase antibodies → loss of efficacy |
| Monitoring | Check serum uric acid before each infusion; if >6 mg/dL, antibodies likely → discontinue |
6.4 IL-1 INHIBITORS (Emerging/Refractory Cases)
- Anakinra (IL-1Ra), Canakinumab (anti-IL-1β monoclonal Ab), Rilonacept (IL-1 trap)
- Used in patients with contraindications to all standard therapies or refractory gout
- MOA: Block IL-1β, the central inflammatory mediator in the gouty cascade
7. DRUG SUMMARY TABLE
| Drug | Class | MOA | Use | Key ADRs |
|---|---|---|---|---|
| Indomethacin | NSAID | COX-1/2 inhibition | Acute gout | GI ulceration, renal impairment |
| Colchicine | Alkaloid | Tubulin depolymerization → ↓ neutrophil migration | Acute + prophylaxis | Diarrhea, N&V, myopathy (chronic) |
| Prednisone | Corticosteroid | Anti-inflammatory | Acute (when NSAID/colchicine contraindicated) | Hyperglycemia, rebound flare |
| Allopurinol | XO inhibitor | ↓ uric acid synthesis | Chronic (1st line) | Hypersensitivity syndrome, azathioprine interaction |
| Febuxostat | XO inhibitor (non-purine) | ↓ uric acid synthesis | Chronic (allopurinol-intolerant) | ↑ CV mortality in high-risk patients |
| Probenecid | Uricosuric | URAT1 inhibition → ↑ urate excretion | Chronic (underexcreters) | Nephrolithiasis, drug interactions |
| Pegloticase | Recombinant uricase | Converts uric acid → allantoin | Refractory gout | Infusion reactions, antibody formation |
| Canakinumab | IL-1β monoclonal Ab | Blocks IL-1β | Refractory/contraindications | Infections, cost |
8. KEY HIGH-YIELD FACTS
| Fact | Detail |
|---|---|
| Crystal appearance | Needle-shaped; negatively birefringent (yellow parallel to slow ray) |
| Classic first joint | First metatarsophalangeal joint (podagra) — 50% of acute attacks |
| Normal urate level | <6.8 mg/dL (treatment target: <6 mg/dL) |
| Primary gout mechanism | Underexcretion (90% of cases) |
| HGPRT deficiency | Partial → secondary gout; Complete → Lesch-Nyhan syndrome (gout + neurologic disease) |
| Colchicine window | Must give within 36 hours of attack onset |
| Allopurinol + azathioprine | Reduce azathioprine dose by 75% or switch to mycophenolate |
| Febuxostat warning | Avoid in patients with major pre-existing cardiovascular disease |
| Diuretics and gout | Thiazides and loop diuretics → ↑ urate reabsorption → precipitate gout |
| Aspirin (low-dose) | Paradoxically raises uric acid (competes with urate excretion) |
| Beer/alcohol | Purine-rich + inhibits renal urate excretion → both mechanisms |
| Starting ULT | Always co-prescribe colchicine/NSAID prophylaxis for ≥6 months to prevent flares |
Sources: Robbins & Kumar Basic Pathology; Katzung's Basic and Clinical Pharmacology, 16th Ed., Chapter 36; Lippincott Illustrated Reviews: Pharmacology; Grainger & Allison's Diagnostic Radiology; Comprehensive Clinical Nephrology, 7th Ed.
Etiology
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
In short
Based on the sources already in context, here is the concise answer:
Etiology of Gout — In Short
Gout results from hyperuricemia (serum urate >6.8 mg/dL), caused by either underexcretion or overproduction of uric acid.
1. Underexcretion (>90% of cases)
Primary: Idiopathic — defective renal tubular handling of urate (most common)
Secondary:
- Chronic kidney disease (↓ GFR)
- Hypertension, obesity, metabolic syndrome
- Lead nephropathy (saturnine gout)
- Hyperparathyroidism, hypothyroidism
- Lactic acidosis / ketoacidosis (lactate/ketones compete with urate at URAT1 transporter)
Drugs that reduce urate excretion:
| Drug | Mechanism |
|---|---|
| Thiazide & loop diuretics | ↑ tubular reabsorption |
| Low-dose aspirin (<3 g/day) | Competes with urate secretion |
| Cyclosporine / Tacrolimus | Impair tubular secretion |
| Nicotinic acid (niacin) | ↓ renal urate clearance |
| Ethambutol, Pyrazinamide | Compete with urate secretion |
| Levodopa, ACE inhibitors, β-blockers | ↓ renal urate clearance |
| Alcohol (ethanol) | ↑ lactate → ↑ urate reabsorption |
2. Overproduction (<10% of cases)
Primary (genetic enzyme defects):
| Defect | Effect |
|---|---|
| HGPRT deficiency (partial) | ↓ purine salvage → ↑ de novo synthesis → ↑ uric acid |
| HGPRT deficiency (complete) | Lesch-Nyhan syndrome — gout + intellectual disability + self-mutilation |
| PRPP synthetase overactivity | ↑ PRPP → ↑ purine synthesis → ↑ uric acid |
| Glucose-6-phosphatase deficiency (von Gierke disease) | Both overproduction + underexcretion |
Secondary (increased cell turnover → ↑ nucleotide catabolism):
- Myeloproliferative / lymphoproliferative disorders
- Haemolytic anaemia
- Psoriasis
- Tumour lysis syndrome (chemotherapy)
Dietary:
- Red meat, organ meat, shellfish (purine-rich)
- Beer and alcohol (purine content + ↑ lactate)
- High-fructose corn syrup (↑ purine nucleotide catabolism)
Summary
| Mechanism | Proportion | Key Causes |
|---|---|---|
| Underexcretion | ~90% | CKD, diuretics, cyclosporine, aspirin, alcohol, idiopathic |
| Overproduction | ~10% | HGPRT/PRPP defects, haematologic malignancies, tumour lysis, diet |
— Goldman-Cecil Medicine; Fitzpatrick's Dermatology; Lippincott Biochemistry, 8th Ed.; Washington Manual of Medical Therapeutics
This is a shared conversation. Sign in to Orris to start your own chat.