Diuretics(Loop , Thiazide) and Renal Pharmacology. Detailed pharmacolofical notes
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Searching Images
loop diuretics mechanism TAL nephron sodium potassium chloride
Searching Images
thiazide diuretics mechanism distal convoluted tubule NaCl cotransporter
Reading File
Reading File
I now have extensive content from both major pharmacology textbooks. I have enough to write comprehensive notes. Let me now compile the full detailed pharmacological notes.
Diuretics — Loop & Thiazide: Detailed Pharmacological Notes
Sources: Goodman & Gilman's Pharmacological Basis of Therapeutics, 14e; Lippincott Illustrated Reviews: Pharmacology; Brenner & Rector's The Kidney; Harrison's Principles of Internal Medicine
I. Overview & Classification of Diuretics
Diuretics increase urinary excretion of Na⁺ and water, reducing extracellular fluid (ECF) volume. They are classified by their primary site of action in the nephron:
| Class | Site of Action | Efficacy (% filtered Na⁺ excreted) |
|---|---|---|
| Carbonic anhydrase inhibitors | Proximal tubule | ~5% |
| Loop diuretics | Thick ascending limb (TAL) | Up to 25% |
| Thiazides | Distal convoluted tubule (DCT) | ~5–8% |
| K⁺-sparing (amiloride, triamterene) | Collecting duct | ~2–3% |
| Aldosterone antagonists (spironolactone) | Collecting duct | ~2–3% |
| Osmotic (mannitol) | Proximal tubule + TAL | Variable |
II. LOOP DIURETICS (High-Ceiling Diuretics)
A. Drugs in Class
| Drug | Relative Potency | Oral Bioavailability | t½ | Elimination |
|---|---|---|---|---|
| Furosemide | 1× | ~60% (variable 10–100%) | ~1.5 h | ~65% renal, ~35% hepatic metabolism |
| Bumetanide | 40× | ~80% | ~0.8 h | ~62% renal, ~38% metabolic |
| Torsemide | 3× | ~80% | ~3.5 h | ~20% renal, ~80% hepatic |
| Ethacrynic acid | 0.7× | ~100% | ~1 h | ~67% renal |
Key distinction: Furosemide and bumetanide contain a sulfonamide moiety. Ethacrynic acid is a phenoxyacetic acid derivative — the only loop diuretic safe to use in patients with true sulfa allergy. Torsemide is a sulfonylurea.
B. Mechanism of Action
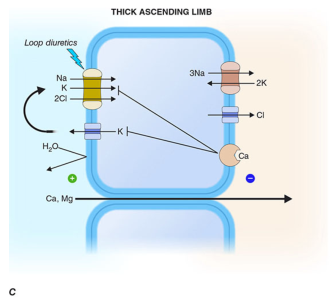
Loop diuretics act primarily on the thick ascending limb (TAL) of the loop of Henle, where the Na⁺-K⁺-2Cl⁻ symporter (NKCC2/BSC1) drives co-transport of Na⁺, K⁺, and 2Cl⁻ from the tubular lumen into the epithelial cell.
Step-by-step:
- Loop diuretics are highly protein-bound → not filtered at the glomerulus
- They reach the tubular lumen via active secretion by the organic acid transport system (OAT1/OAT3 on basolateral membrane → MRP-4 on luminal membrane) in the proximal tubule
- They bind the Cl⁻ binding site of the NKCC2 symporter on the luminal membrane of the TAL
- Salt transport in the TAL is halted; downstream segments lack the capacity to compensate
- This destroys the hypertonic medullary interstitium → loss of urinary concentrating ability
Why TAL is the most powerful diuretic target:
- TAL normally reabsorbs 25–30% of filtered NaCl
- The TAL is water-impermeable → salt reabsorption here creates the concentrated medullary gradient
- Downstream nephron segments cannot rescue the flood of unabsorbed solute
The NKCC2 transporter — two variants:
- Absorptive (NKCC2/BSC1): kidney-specific, apical membrane of TAL — primary drug target
- Secretory (NKCC1/BSC2): ubiquitous, basolateral in epithelia — responsible for ototoxicity at high doses
Bartter syndrome is caused by loss-of-function mutations in the NKCC2 transporter (or associated proteins), mimicking chronic loop diuretic use.
C. Effects on Urinary Excretion
| Electrolyte/Parameter | Effect |
|---|---|
| Na⁺, Cl⁻ | ↑↑ (up to 25% of filtered load) |
| K⁺ | ↑↑ (increased distal delivery → enhanced K⁺/H⁺ secretion) |
| Mg²⁺ | ↑ (loss of paracellular driving force) |
| Ca²⁺ | ↑↑ (loss of lumen-positive potential abolishes paracellular Ca²⁺ reabsorption) |
| H⁺ | ↑ (hypochloremic metabolic alkalosis) |
| Uric acid | Acute: ↑; Chronic: ↓ (competition with OAT in proximal tubule) |
| Urinary concentrating ability | ↓ (destroys medullary gradient) |
| Urinary diluting ability | ↓ (TAL is part of the diluting segment) |
| HCO₃⁻ | ↑ (furosemide has weak carbonic anhydrase-inhibiting activity) |
Urine volume: Profound (hence "high-ceiling"). Loop diuretics display a sigmoidal dose-response curve:
- Below threshold → no diuresis
- Steep portion → dose-dependent diuresis
- Ceiling effect → no further diuresis above maximum
D. Renal Hemodynamic Effects
- Increase RBF (if volume depletion prevented) — mechanism may involve prostaglandins; NSAIDs blunt this
- Block tubuloglomerular feedback (TGF): By inhibiting NaCl transport into the macula densa, the macula densa cannot detect tubular NaCl → GFR maintained (unlike carbonic anhydrase inhibitors)
- Powerful stimulators of renin release: via macula densa pathway + sympathetic activation + baroreceptor mechanism
- Acute venodilation: Furosemide acutely increases systemic venous capacitance, reducing left ventricular filling pressure before diuresis begins — mediated by prostaglandins
E. Pharmacokinetics (ADME)
- Protein binding: Extensive → minimal filtration; access to tubule via proximal secretion
- Furosemide: ~65% excreted unchanged in urine; remainder glucuronidated in kidney → prolonged t½ in renal disease
- Bumetanide & torsemide: Significant hepatic metabolism → prolonged t½ in liver disease
- Oral bioavailability: Furosemide is notorious for erratic absorption (10–100%). Bumetanide and torsemide are reliably ~80%
- Short t½ → postdiuretic Na⁺ retention: As loop diuretic levels fall in tubule, Na⁺ is avidly reclaimed — overcome by sodium restriction or more frequent dosing
- IV dosing in hospitalized patients: Initial IV dose = 2–2.5× the chronic oral dose
F. Therapeutic Uses
- Acute pulmonary edema — drug of choice; venodilation + brisk natriuresis rapidly reduces LV filling pressure
- Chronic heart failure — reduce pulmonary/peripheral congestion; torsemide preferred over furosemide (more reliable absorption, fewer hospitalizations)
- Hypertension — not first-line in normal renal function (less antihypertensive efficacy vs. thiazides); drugs of choice when GFR <30 mL/min or resistant hypertension
- Nephrotic syndrome — often the only drugs capable of mobilizing massive edema
- Cirrhosis with ascites — useful but risk of volume depletion and hepatic encephalopathy; spironolactone often combined
- Hypercalcemia — saline + furosemide promotes calciuresis
- Chronic kidney disease edema — dose-response curve is right-shifted; higher doses required
- Hyponatremia (life-threatening) — forced diuresis
- Drug overdose — forced diuresis to enhance renal elimination
- Hyperkalemia — promotes kaliuresis
G. Adverse Effects
| Adverse Effect | Mechanism |
|---|---|
| Hypokalemia | ↑ distal Na⁺ delivery → enhanced K⁺/H⁺ secretion |
| Hypochloremic metabolic alkalosis | Loss of Cl⁻ and H⁺ |
| Hyponatremia | Volume depletion → non-osmotic ADH release |
| Hypomagnesemia | ↑ urinary Mg²⁺ excretion → cardiac arrhythmia risk |
| Hypocalcemia (rarely tetany) | ↑ urinary Ca²⁺ excretion |
| Hyperuricemia (→ gout) | Competition with uric acid in proximal OAT |
| Hyperglycemia | Hypokalemia impairs insulin secretion |
| Ototoxicity | NKCC1 inhibition in inner ear (endolymph) — tinnitus, hearing loss, vertigo; ethacrynic acid most ototoxic |
| Volume depletion | Overzealous use → hypotension, ↓ GFR, thromboembolic events |
| Skin rashes, photosensitivity | Sulfonamide moiety |
| Bone marrow depression | Rare |
Ototoxicity precaution: Furosemide infusion rate must not exceed 4 mg/min. Synergistic ototoxicity with aminoglycosides, cisplatin, carboplatin.
H. Drug Interactions
| Interacting Drug | Effect |
|---|---|
| Aminoglycosides, cisplatin | Additive ototoxicity |
| Digitalis glycosides | Hypokalemia → increased arrhythmia risk |
| Lithium | ↑ plasma lithium (↓ renal clearance) |
| NSAIDs | Blunted diuretic response (inhibit prostaglandins + ↓ RBF) |
| Thiazides | Synergistic profound diuresis (sequential nephron blockade) |
| Probenecid | Competes for proximal tubule secretion → blunted response |
| Sulfonylureas | Hyperglycemia |
| Amphotericin B | ↑ nephrotoxicity and electrolyte imbalance |
I. Contraindications
- Severe Na⁺ depletion / volume depletion
- Anuria unresponsive to a trial dose
- Sulfa allergy (for sulfonamide-based agents — use ethacrynic acid instead)
- Caution in post-menopausal osteopenic women (↑ Ca²⁺ excretion affects bone)
- Hepatic encephalopathy risk in cirrhosis
III. THIAZIDE DIURETICS
A. Drugs in Class
True thiazides (benzothiadiazine structure):
- Chlorothiazide (first orally active)
- Hydrochlorothiazide (HCTZ) — most widely used
Thiazide-like diuretics (similar mechanism, different structure):
- Chlorthalidone (~2× more potent than HCTZ; longer t½ ~45–60 h)
- Indapamide — additional direct vasodilatory effect
- Metolazone — effective even with low GFR; particularly useful for diuretic resistance
All members: equal maximum diuretic efficacy (differ only in potency) — "low ceiling diuretics" because dose escalation beyond the therapeutic range does not produce additional diuresis.
B. Mechanism of Action
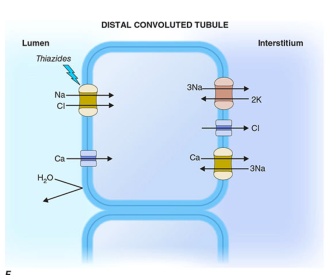
Thiazides act on the distal convoluted tubule (DCT) to inhibit the Na⁺/Cl⁻ cotransporter (NCC) on the apical (luminal) membrane.
Step-by-step:
- Thiazides are secreted into the tubular lumen via the proximal tubule organic acid secretory system — therefore impaired renal function (↓ GFR) reduces diuretic efficacy
- They bind the NCC on the apical membrane of DCT cells, blocking Na⁺ and Cl⁻ reabsorption
- ↑ Na⁺ in tubular fluid → ↑ Na⁺/K⁺ exchange in collecting duct → K⁺ wasting
- Unique calcium effect: By lowering intracellular Na⁺, the basolateral Na⁺/Ca²⁺ exchanger is enhanced → increased Ca²⁺ reabsorption (opposite to loop diuretics)
- Long-term: reduce peripheral vascular resistance (mechanism independent of diuresis — possibly related to vascular smooth muscle effects)
NSAIDs reduce thiazide efficacy by inhibiting renal prostaglandin synthesis and reducing RBF.
C. Effects on Urinary Excretion
| Electrolyte/Parameter | Effect |
|---|---|
| Na⁺, Cl⁻ | ↑ |
| K⁺ | ↑ (hypokalemia most frequent side effect) |
| Mg²⁺ | ↑ (hypomagnesemia) |
| Ca²⁺ | ↓ (unique — Ca²⁺ retention; hypercalcemia) |
| Uric acid | ↓ excretion → hyperuricemia |
| HCO₃⁻ | ↑ slight |
| Urine volume | Initial: ↑; Chronic: ↓ (compensatory mechanisms) |
| Ability to produce dilute urine | ↓ (DCT is part of diluting segment) |
| Concentrated urine | ✓ Can still produce concentrated urine (unique among diuretics) |
D. Pharmacokinetics
| Drug | t½ | Notes |
|---|---|---|
| HCTZ | 6–15 h | Renal elimination |
| Chlorthalidone | ~45–60 h | Much longer; preferred for hypertension |
| Indapamide | ~15–25 h | Largely hepatic metabolism |
| Metolazone | ~8 h | Works at GFR <30 mL/min |
| Chlorothiazide | ~1.5 h | Oral, IV available |
All thiazides require secretion into the proximal tubule to reach the DCT. Decreasing renal function reduces diuretic efficacy (though antihypertensive effects may persist even below GFR 30 mL/min/1.73 m²).
E. Therapeutic Uses
- Hypertension — first-line therapy in most patients; antihypertensive effect is only partially dependent on diuresis; long-term reduction of peripheral vascular resistance is important
- Edema — mild-to-moderate fluid retention (heart failure, hepatic cirrhosis, corticosteroid/estrogen therapy)
- Chronic heart failure — adjunct for volume management; often combined with loop diuretics
- Hypercalciuria / Calcium nephrolithiasis — Ca²⁺-retaining effect prevents stone formation
- Nephrogenic diabetes insipidus (DI) — paradoxical antidiuretic effect: volume depletion → ↑ proximal Na⁺ reabsorption → ↓ water delivery to collecting duct → reduced urine volume
- Osteoporosis — Ca²⁺ retention may reduce bone loss (secondary benefit)
Thiazides are ineffective when GFR <30 mL/min for diuretic purposes — switch to loop diuretics (exception: metolazone remains effective).
F. Adverse Effects

| Adverse Effect | Mechanism | Notes |
|---|---|---|
| Hypokalemia | ↑ distal Na⁺ delivery → K⁺/Na⁺ exchange | Most common adverse effect; monitor K⁺ periodically |
| Hypomagnesemia | ↑ urinary Mg²⁺ loss | Can worsen hypokalemia (Mg²⁺ deficiency impairs K⁺ repletion) |
| Hyponatremia | ↑ ADH (volume-mediated) + ↓ diluting capacity + ↑ thirst | Serious risk in elderly |
| Hyperuricemia | Competition with uric acid in OAT → ↓ uric acid excretion | Can precipitate gout |
| Hyperglycemia | Hypokalemia → impaired insulin release | Monitor glucose in diabetics |
| Hypercalcemia | ↑ Ca²⁺ reabsorption in DCT | Useful in hypercalciuria; harmful in hypercalcemia states |
| Hyperlipidemia | ↑ LDL, triglycerides, ↓ HDL | Minor, reversible |
| Orthostatic hypotension | Volume depletion | |
| Photosensitivity | Sulfonamide moiety | Phototoxic/photoallergic skin reaction |
| Sexual dysfunction | — | Impotence in males (dose-dependent) |
Mnemonic for thiazide side effects — "GLHK" or "HyperGLUC":
- Hyperglycemia
- Hyperlipidemia
- Hyperuricemia
- Hypercalcemia
- HypoKalemia, HypoMagnesemia, HypoNatremia
G. Drug Interactions
| Interacting Drug | Effect |
|---|---|
| Loop diuretics | Sequential nephron blockade → synergistic profound diuresis |
| Digitalis glycosides | Hypokalemia → digoxin toxicity |
| Lithium | ↓ renal Li⁺ clearance → toxicity |
| NSAIDs | ↓ diuretic and antihypertensive efficacy |
| Antidiabetics | Hyperglycemia may require dose adjustment |
| Corticosteroids | Additive hypokalemia |
| Calcium supplements / Vitamin D | Risk of hypercalcemia |
H. Contraindications
- Sulfa allergy (relative — cross-reactivity rare but possible)
- Anuria
- Severe hypokalemia/hyponatremia
- Gout (caution — hyperuricemia)
- GFR <30 mL/min for diuretic purposes (except metolazone)
- Pregnancy: avoid (associated with neonatal thrombocytopenia, electrolyte disturbances)
IV. KEY COMPARATIVE TABLE: Loop vs. Thiazide
| Feature | Loop Diuretics | Thiazide Diuretics |
|---|---|---|
| Site of action | TAL of loop of Henle | DCT |
| Transporter blocked | NKCC2 (Na⁺-K⁺-2Cl⁻) | NCC (Na⁺-Cl⁻) |
| Efficacy | HIGH (25–30% filtered Na⁺) | LOW–MODERATE (5–8%) |
| Ceiling | High ceiling | Low ceiling |
| Ca²⁺ effect | ↑ excretion (hypocalcemia) | ↓ excretion (hypercalcemia) |
| Use in low GFR | ✓ Yes (preferred <30) | ✗ No (except metolazone) |
| Uric acid | ↓ excretion (hyperuricemia) | ↓ excretion (hyperuricemia) |
| K⁺ | Wasting | Wasting |
| 1st-line hypertension | No | Yes |
| Pulmonary edema | 1st choice | Not used |
| Nephrogenic DI | Not effective | ✓ Paradoxical antidiuresis |
| Calcium stones | Not used | ✓ Reduces stone recurrence |
| Ototoxicity | Yes (esp. ethacrynic acid) | No |
| Concentrating urine | Cannot | Can (preserves medullary gradient) |
| Oral bioavailability | Variable (furosemide 10–100%) | Generally good |
V. DIURETIC RESISTANCE
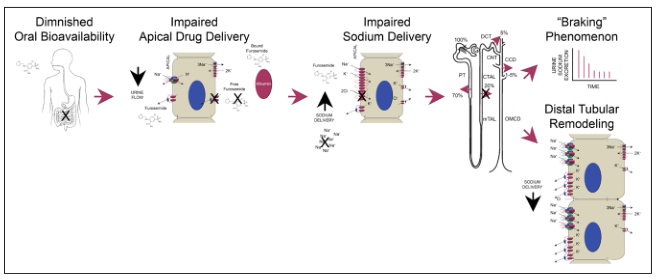
Diuretic resistance (inadequate response despite adequate doses) occurs via several mechanisms:
- Diminished oral bioavailability — especially furosemide (gut edema in heart failure)
- Impaired proximal tubular secretion — hypoalbuminemia reduces drug delivery to lumen; NSAIDs block OAT
- "Braking" phenomenon — chronic neurohormonal activation (RAAS, SNS, ADH) causes compensatory Na⁺ retention in distal nephron
- Distal tubular remodeling — chronic loop diuretic use → hypertrophy of DCT cells → increased NCC expression → more downstream compensation
Management of diuretic resistance:
- Switch to IV administration (bypasses gut absorption problem)
- Combine loop + thiazide (sequential nephron blockade — adds metolazone or HCTZ to loop diuretic = profound synergistic diuresis)
- Restrict dietary sodium (<2 g/day)
- Switch to torsemide (more reliable absorption) or bumetanide
- Consider IV albumin (in hypoalbuminemia/nephrotic syndrome)
- Ultrafiltration or dialysis if refractory
VI. RENAL PHARMACOLOGY: CONTEXT
Tubular Secretion of Diuretics
Both loop and thiazide diuretics reach their luminal targets through active secretion, not filtration:
- Basolateral uptake via OAT1/OAT3
- Luminal efflux via MRP-4
- Any condition reducing proximal tubule secretion blunts diuretic efficacy (renal failure, probenecid competition, hypoalbuminemia)
Renin-Angiotensin System Activation
Both loop and thiazide diuretics activate RAAS by:
- Volume depletion → baroreceptor-mediated sympathetic activation
- Macula densa NaCl sensing → direct renin stimulation (loop diuretics, via NKCC2 block)
- This is why combining diuretics with ACE inhibitors/ARBs is rational — it blocks the compensatory RAAS activation
Electrolyte Monitoring
All patients on loop or thiazide diuretics require monitoring of:
- Serum Na⁺, K⁺, Cl⁻, HCO₃⁻ (electrolytes)
- Mg²⁺ (often overlooked; hypomagnesemia perpetuates hypokalemia)
- Serum creatinine / BUN (volume depletion, pre-renal AKI)
- Uric acid (hyperuricemia)
- Glucose (especially in diabetics on thiazides)
- Blood pressure (orthostatic measurements)
Sources:
- Goodman & Gilman's Pharmacological Basis of Therapeutics, 14e, Chapter 29 (Diuretics)
- Lippincott Illustrated Reviews: Pharmacology, 8e, Chapter 9 (Diuretics)
- Brenner & Rector's The Kidney, 10e
- Harrison's Principles of Internal Medicine, 21e
Add katzung too
Your credits are exhausted. Your daily credits reset tomorrow, or you can purchase a top-up for more.
This is a shared conversation. Sign in to Orris to start your own chat.