Pluripotent
Reading File
Finding Sources
Finding Sources
Searching the Web
pluripotent stem cells biology definition types
Reading File
Reading File
Finding Sources
Reading File
Reading File
Loading Image
Reading File
Here is a comprehensive overview of pluripotency drawn from authoritative medical textbooks.
Pluripotency
Pluripotent stem cells (PSCs) are undifferentiated cells defined by two core properties:
- Self-renewal — the capacity to divide indefinitely while remaining undifferentiated
- Multilineage differentiation — the ability to give rise to any cell type derived from all three primary germ layers: ectoderm, mesoderm, and endoderm
They cannot, however, form extra-embryonic tissues (e.g., the placenta) — that broader capacity belongs to totipotent cells (the zygote and early blastomeres).
Potency Hierarchy
| Term | Potential | Example |
|---|---|---|
| Totipotent | All cell types including placenta | Fertilized egg, morula cells |
| Pluripotent | All three germ layers; no placenta | ESCs, iPSCs |
| Multipotent | Multiple but restricted lineages | Hematopoietic stem cells |
| Unipotent | Single lineage | Skin basal cells |
Types of Pluripotent Stem Cells
1. Embryonic Stem Cells (ESCs)
ESCs are a transient population arising shortly after fertilization and disappearing after gastrulation, when the three germ layers are established. They are derived from the inner cell mass (ICM) of the blastocyst.
- Mouse ESCs resemble the ICM of the blastocyst-stage embryo
- Human ESCs resemble the early epiblast — a disc-like columnar epithelium derived from the ICM, and thus slightly further along in development
- ESCs can be propagated indefinitely in culture under special conditions while retaining pluripotency
- Key transcription factors maintaining pluripotency: OCT4 (POU5F1), SOX2, NANOG, KLF4
- Using combinations of growth factors, ESCs can be coaxed into neurons, glia, cardiomyocytes, vascular cells, pancreatic β-cells, hepatocytes, and blood cells
— Brenner and Rector's The Kidney; Goldman-Cecil Medicine
2. Induced Pluripotent Stem Cells (iPSCs)
Introduced by Takahashi & Yamanaka (2006), iPSCs are somatic (adult) cells — typically fibroblasts or blood cells — reprogrammed back to a pluripotent state by forced expression of master transcription factors, without requiring eggs or embryos.
Yamanaka factors used for reprogramming:
- Oct4, Sox2, Klf4, c-Myc
Key features:
- Once reprogrammed, iPSCs are stable in the undifferentiated state without continued forced gene expression
- Genome-wide gene expression is nearly indistinguishable from ESCs
- Can be generated in a patient-specific manner → autologous cell replacement, avoiding immunologic rejection
- Used to create "disease-in-a-dish" models of genetic disorders
- Enable drug screening and drug discovery platforms
— Brenner and Rector's The Kidney; The Developing Human (Clinically Oriented Embryology)
Reprogramming Mechanisms
A critical step in iPSC reprogramming is the rewriting of the epigenetic code of the donor cell. The nucleus of an adult cell can also be reprogrammed via:
- Somatic cell nuclear transfer (SCNT): implanting a somatic nucleus into an enucleated egg — this produces a totipotent-like reconstructed cell (used in reproductive cloning; Dolly the sheep, 1996)
- Therapeutic cloning: SCNT-derived ESCs intended for cell therapy — limited by egg availability
Applications of Pluripotent Stem Cells
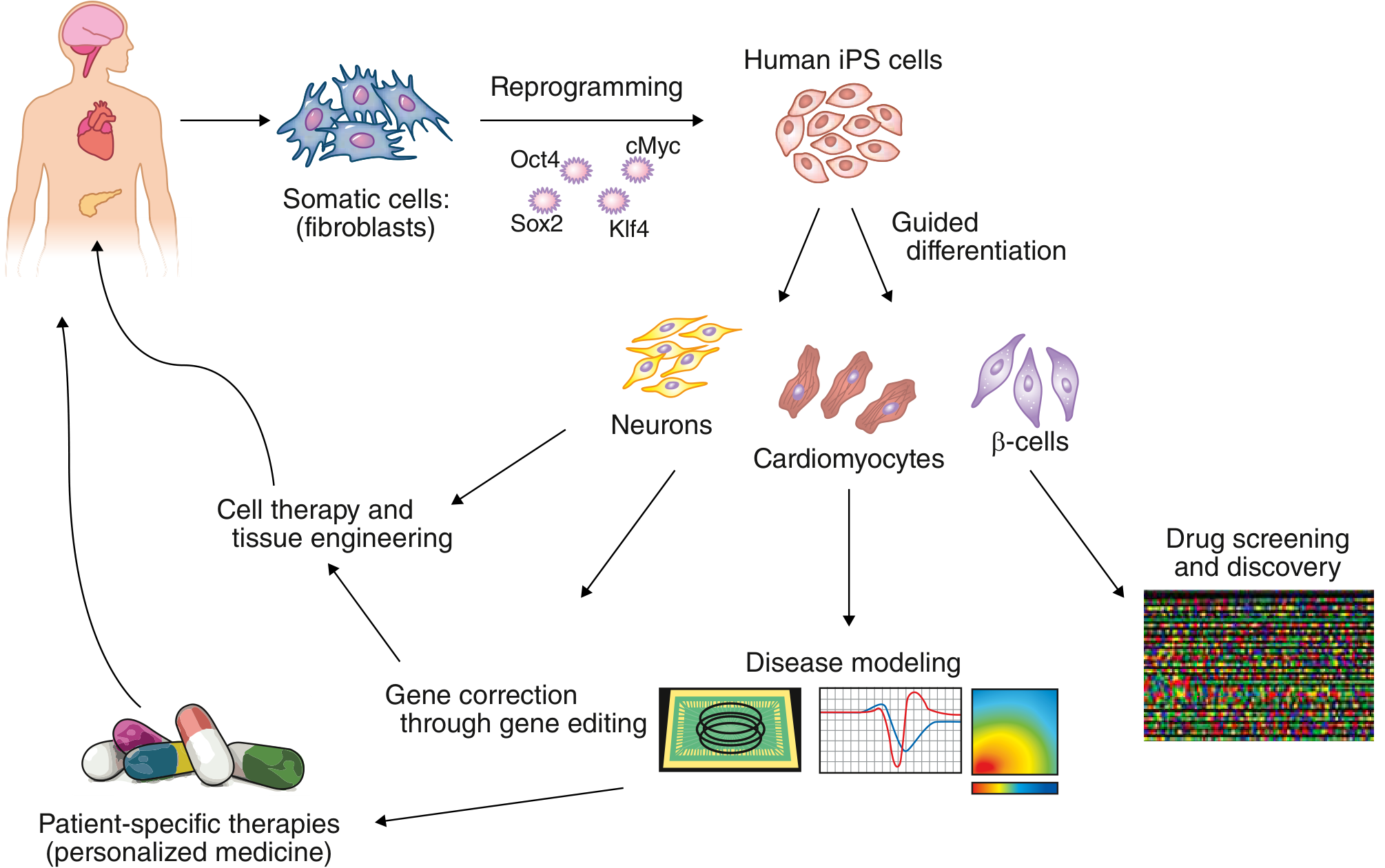
FIGURE: Potential applications of human iPSCs — Goldman-Cecil Medicine, 15th Ed.
| Application | Description |
|---|---|
| Regenerative medicine | Autologous cell transplantation (e.g., cardiomyocytes for heart failure, β-cells for diabetes) |
| Disease modeling | Patient-specific iPSCs differentiated to model cystic fibrosis, cardiomyopathies, neurological diseases |
| Drug discovery | High-throughput screening platforms using disease-specific cell lines |
| Gene therapy | CRISPR/Cas9 correction of mutations in iPSCs → differentiation → return to patient |
| Kidney organoids | iPSC-derived nephron structures for kidney disease research |
| Lung epithelial cells | Modeling pulmonary diseases |
— Goldman-Cecil Medicine; Emery's Elements of Medical Genetics; The Developing Human
Cancer Stem Cells (CSCs)
A related concept: cancer stem cells are a subpopulation within tumors (identified by markers like CD133 in solid tumors) that retain self-renewal capacity, resist chemotherapy and radiation, and drive tumor recurrence. Eradicating CSCs alongside standard therapy is an active area of oncology research.
— The Developing Human, Clinically Oriented Embryology
Clinical Caution
Despite enormous promise in animal models, translation to humans has shown more limited success. Patients are advised that stem cell therapy remains largely experimental outside of regulated trials. "Stem cell tourism" — seeking unregulated treatments abroad — carries real risk of harm with no proven benefit. (Emery's Elements of Medical Genetics)
This is a shared conversation. Sign in to Orris to start your own chat.