Management of heavy uterine bleeding
Reading File
Finding Sources
Finding Sources
Searching PubMed
"heavy menstrual bleeding" AND management
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
I now have comprehensive content. Let me compile the full answer.
Management of Heavy Uterine Bleeding (HUB)
Heavy uterine bleeding (HUB), also called heavy menstrual bleeding (HMB) or formerly menorrhagia, is defined as menstrual blood loss that interferes with a woman's physical, emotional, social, or quality-of-life. It is one of the most common gynecologic complaints.
1. Initial Assessment
Before treating, identify the underlying cause using the PALM-COEIN classification (structural vs. non-structural):
Structural (PALM): Polyp, Adenomyosis, Leiomyoma, Malignancy/hyperplasia
Non-structural (COEIN): Coagulopathy, Ovulatory dysfunction, Endometrial, Iatrogenic, Not yet classified
Non-structural (COEIN): Coagulopathy, Ovulatory dysfunction, Endometrial, Iatrogenic, Not yet classified
Key evaluation steps:
- CBC, coagulation studies (PT, aPTT), pregnancy test, thyroid function
- Pelvic ultrasound (first-line imaging)
- Endometrial biopsy for women >45 years or with risk factors (obesity, nulliparity, anovulation history, tamoxifen use, family history of endometrial/colon cancer)
- Screen for bleeding disorders — especially in adolescents with HMB since menarche (von Willebrand disease accounts for up to 20% of acute uterine bleeding in adolescents)
When to suspect a bleeding disorder (Table 9-9, Berek & Novak):
- HMB since menarche
- Family history of bleeding disorder
- Epistaxis, bruising >2 cm without injury, prolonged post-dental bleeding
- Postpartum hemorrhage, especially delayed >24 hrs
2. Acute / Emergency Management (Hemodynamically Unstable)
(Tintinalli's Emergency Medicine, Table 96-4)
| Agent | Dose | Notes |
|---|---|---|
| Conjugated equine estrogen (IV) | 25 mg IV q4–6h until bleeding stops | Emergency GYN consult; give antiemetics. Contraindicated in VTE, thrombophilia, malignancy |
| Tranexamic acid (IV) | 1.0–1.3 g IV | Can transition to PO 3×/day; effective within ~3 h. Contraindicated in VTE/thrombophilia |
| Fluid and blood resuscitation | As needed | Identify and treat coagulopathies; exclude pregnancy, foreign body, laceration |
3. Medical Management (Hemodynamically Stable / Outpatient)
Non-hormonal
| Agent | Dose | Mechanism |
|---|---|---|
| Tranexamic acid (FDA-approved for HMB) | 1.3 g PO 3×/day for 3–5 days during menses | Antifibrinolytic; reduces blood loss ~30–50%. Effective for both idiopathic HMB and fibroid-related bleeding |
| NSAIDs (mefenamic acid, ibuprofen, naproxen) | Mefenamic acid 500 mg TID; ibuprofen 400 mg q6h; naproxen 500 mg BID for 4–5 days | Prostaglandin inhibition; reduces blood loss vs. placebo. Less effective in fibroid-related HMB |
Hormonal
| Agent | Regimen | Notes |
|---|---|---|
| Combined OCP (monophasic, ≤35 µg EE) | TID ×7 days (acute); then OD for maintenance | Bleeding stops in ~3 days. Contraindicated: smokers >35 yrs, HTN, VTE, liver disease, breast cancer |
| Progestin-only (medroxyprogesterone acetate) | 20 mg PO TID ×7 days or OD ×10 days | For patients with contraindications to estrogen; more effective in older/perimenopausal women |
| Levonorgestrel IUS (Mirena) | In-office insertion | ~50% amenorrhea at 1 year; highly effective for anovulatory and fibroid-related HMB |
| Depot medroxyprogesterone (DMPA) | 150 mg IM q3 months | ~50% amenorrhea at 1 year; monitor bone density with long-term use |
| GnRH agonists ± add-back therapy | Monthly injection ×6 months | Reduces fibroid volume 30–35%; used for preoperative downsizing. 95% experience side effects (hot flushes 78%, vaginal dryness 32%). Uterus returns to pretreatment size 4–6 months after stopping |
| GnRH antagonists (elagolix + low-dose add-back) | Oral daily | Newer option, phase III-approved for HMB from uterine fibroids/endometriosis |
4. Special Clinical Scenarios
Anovulatory Bleeding
- Mild (adequate Hgb, minimal disruption): menstrual charting, iron supplementation, close follow-up
- Moderate–severe: combined OCP or cyclic progestins; LNG-IUS for long-term control
- First-line in reproductive age: combined hormonal contraceptives or progestins (Berek & Novak, p. 410)
Fibroid-Related HMB
- Watchful waiting acceptable if mildly symptomatic (77% stable at 1 year)
- Medical: tranexamic acid, GnRH agonists (preoperative use), LNG-IUS
- NSAIDs are not effective for fibroid-related HMB specifically
Coagulopathy (e.g., von Willebrand disease)
- Avoid NSAIDs
- Hormonal agents increase factor VIII and vWF levels
- Desmopressin (DDAVP) stimulates endogenous release of factor VIII/vWF
- Tranexamic acid and recombinant vWF as adjuncts
- Multidisciplinary approach (hematology + gynecology)
Anticoagulated Patients
- Combined contraceptives and tranexamic acid are contraindicated
- Progestin-only with caution and GYN consultation
- Weigh risks/benefits of anticoagulation reversal
PCOS
- Low-dose OCP or progestins for cycle regulation and bleeding control
Perimenopausal Bleeding
- 10% risk of malignancy — prompt endometrial biopsy for all women >45
- GnRH agonists or watchful waiting if approaching menopause
5. Surgical Management
Indicated when medical management fails or is contraindicated:
| Procedure | Indication |
|---|---|
| Dilation & curettage (D&C) | Acute control; diagnostic |
| Hysteroscopy ± directed biopsy | Structural causes (polyps, submucosal fibroids) |
| Endometrial ablation | Completed childbearing; definitive for dysfunctional uterine bleeding |
| Myomectomy (hysteroscopic or laparoscopic) | Fibroid-related HMB; fertility preservation. 25–30% recurrence |
| Uterine artery embolization (UAE) | Fibroids; effective in reducing fibroid volume and symptoms |
| Hysterectomy | Last resort; life-threatening bleeding unresponsive to all other measures |
Stable patients can be discharged with prompt outpatient follow-up after achieving initial hemostasis. (Tintinalli's, p. 653)
6. Key Prescribing Precautions
- Contraindications to estrogen: active VTE, thrombophilia, vascular disease, malignancy, liver disease, smokers >35
- Tranexamic acid contraindications: active VTE, thrombophilia
- DMPA & GnRH agonists: monitor bone mineral density with long-term use
- Always provide antiemetics when using high-dose estrogen
Sources:
- Berek & Novak's Gynecology, pp. 409–414, 515
- Tintinalli's Emergency Medicine, pp. 650–653 (Table 96-4)
- Katzung's Basic and Clinical Pharmacology, 16th ed., p. 288
Recent evidence note: A 2025 systematic review (PMID 39617186) found coagulopathies (vWD, platelet disorders) are significantly under-recognized as a cause of HMB across both adolescents and adults — reinforcing the importance of screening. A 2025 Cochrane review (PMID 40183418) updated evidence on preoperative medical therapy for fibroids.
Endometriosis
Finding Sources
Searching PubMed
"endometriosis"[MeSH Terms] AND management
Finding Sources
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
I now have comprehensive, detailed content from Robbins (pathology), Berek & Novak's Gynecology (management), and Goldman-Cecil Medicine (clinical features and treatment). Let me now compile the full comprehensive answer.
Endometriosis
Definition
Endometriosis is the presence of endometrial glands and stroma at sites outside the uterus. It affects approximately 10% of women of reproductive age and nearly 50% of women with infertility. (Robbins & Kumar Basic Pathology)
Classification / Types
There are three recognized types:
- Superficial peritoneal endometriosis — flat implants on peritoneal surfaces
- Ovarian endometriosis (endometriomas / "chocolate cysts") — ovarian cysts 3–5 cm filled with degenerated blood
- Deep infiltrating endometriosis (DIE) — lesions penetrating >5 mm below peritoneal surface; highest risk of malignant transformation
Common sites: ovaries, pouch of Douglas, uterosacral ligaments, fallopian tubes, bladder, rectum, peritoneum. Distant sites (lung, umbilicus, surgical scars) occur via lymphovascular spread.
Pathogenesis
Several complementary theories exist (Robbins, p. 689):
| Theory | Mechanism |
|---|---|
| Regurgitation (Sampson's) | Retrograde menstruation → implantation of endometrial cells at ectopic pelvic sites (most accepted) |
| Benign metastasis | Lymphovascular spread to distant sites (lung, bone, brain) |
| Coelomic metaplasia | Differentiation of peritoneal mesothelium into endometrial tissue (explains distant/non-pelvic lesions) |
| Stem/progenitor cell | Bone marrow–derived stem cells differentiate into ectopic endometrium |
Molecular sustaining factors:
- Endometriotic implants produce ↑ prostaglandin E2, VEGF, and matrix metalloproteinases (MMPs)
- Recruited macrophages amplify local inflammation
- High aromatase expression → increased local estrogen production → self-sustaining growth cycle
- Disease is estrogen-dependent and progesterone-resistant
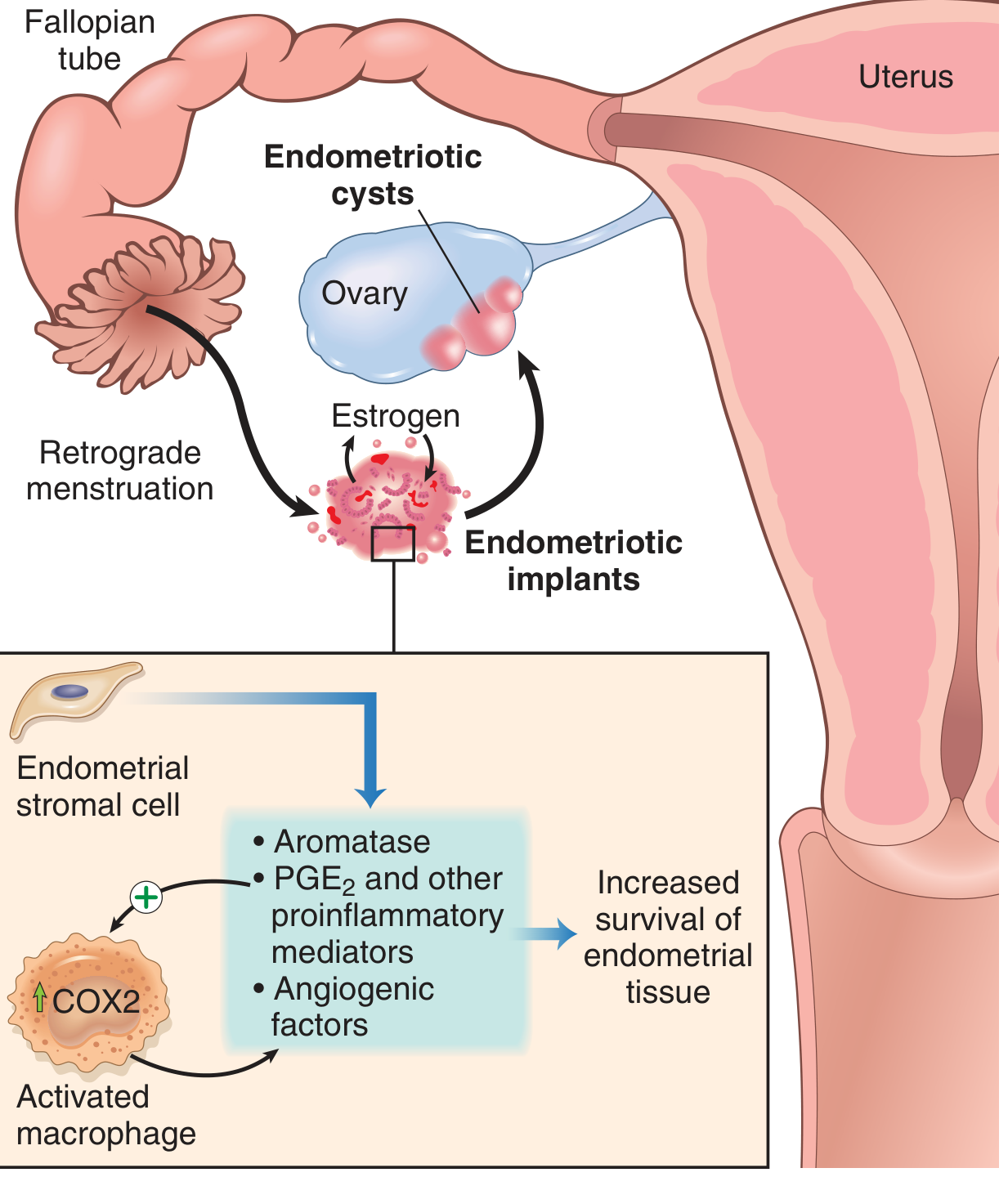
Morphology
Grossly, lesions appear as red-brown nodules (1–2 cm) on serosal surfaces due to cyclic hemorrhage. Organizing hemorrhage → fibrous adhesions → distortion of pelvic anatomy. Ovarian endometriomas contain dark brown, tarry fluid ("chocolate cysts").
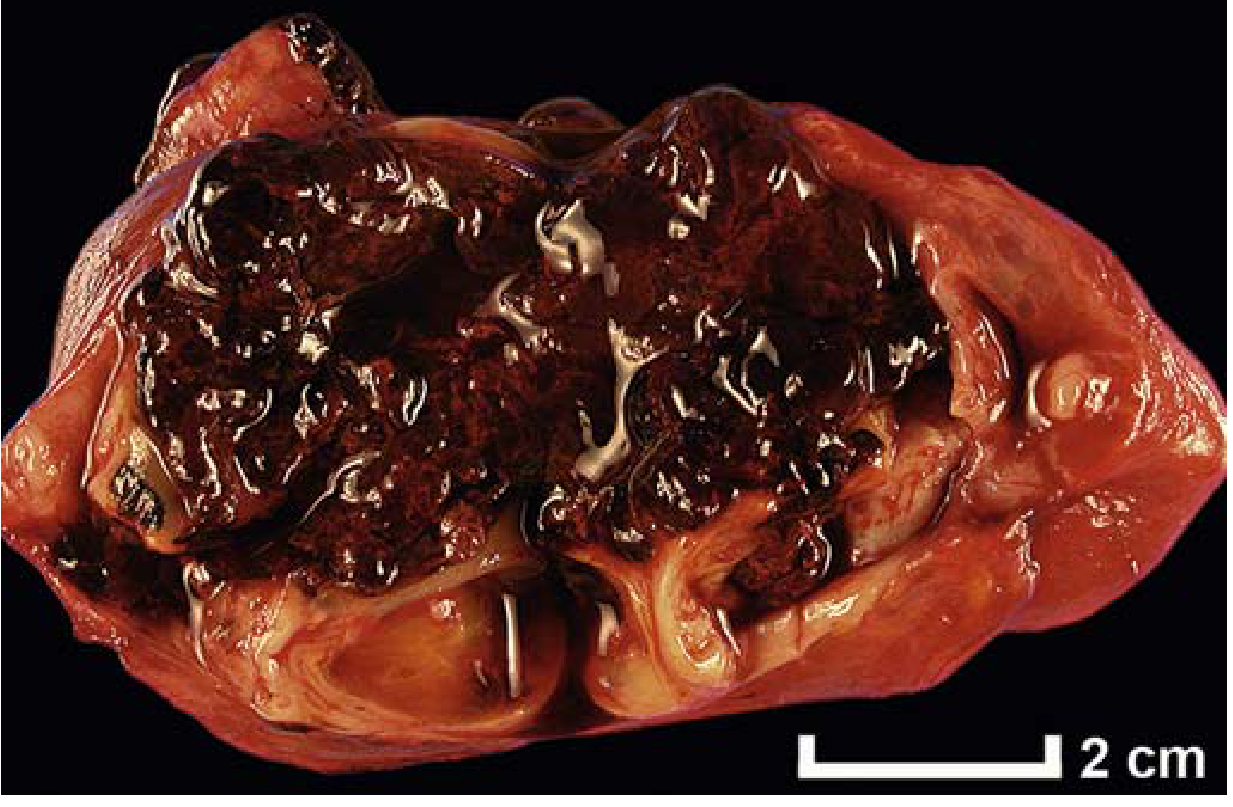
Histology: requires both endometrial glands AND stroma at ectopic site for definitive diagnosis.
Clinical Features
Classic triad:
- Dysmenorrhea (severe, often progressive) — most common presenting symptom
- Dyspareunia (deep)
- Chronic pelvic pain
Additional features:
- Infertility — 30–40% present with infertility as primary complaint
- Menstrual irregularities, heavy periods
- Dyschezia (painful defecation) — DIE involving rectum
- Dysuria — bladder involvement
- Cyclical haematuria, haemoptysis (rare thoracic endometriosis)
Natural history: Progressive in 30–60% within 1 year without treatment. Deterioration seen in 47%, improvement in 30%, resolution in 23% over 6 months in observational studies. (Berek & Novak, p. 651)
Staging (rASRM / ESHRE)
The revised American Society for Reproductive Medicine (rASRM) classification scores implants, adhesions, and endometrioma involvement:
| Stage | Score | Description |
|---|---|---|
| I | 1–5 | Minimal |
| II | 6–15 | Mild |
| III | 16–40 | Moderate |
| IV | >40 | Severe |
Limitation: rASRM staging correlates poorly with pain severity or fertility prognosis. The Endometriosis Fertility Index (EFI) better predicts pregnancy rates post-surgery.
Diagnosis
- Definitive diagnosis: laparoscopy with histological confirmation (biopsy showing glands + stroma)
- Pelvic ultrasound: first-line imaging; identifies endometriomas ("ground-glass" appearance); limited for peritoneal and DIE lesions
- MRI pelvis: gold standard for mapping DIE; used in surgical planning for complex disease
- CA-125: may be elevated but non-specific; not used for diagnosis alone
- No reliable non-invasive biomarker exists to date
Empirical treatment (without laparoscopy) is acceptable in adolescents and women with classic symptoms when imaging is unremarkable — NSAIDS + hormonal therapy trial.
Management
Treatment must be individualized based on: symptom severity, desire for fertility, age, extent of disease, and patient preferences. Endometriosis is a chronic, relapsing condition — recurrence is common after both medical and surgical treatment. (Berek & Novak, p. 655)
1. Analgesics (First-Line for Pain)
- NSAIDs (ibuprofen 400–800 mg q4–6h; naproxen 500 mg BD; mefenamic acid 500 mg TDS) — inhibit prostaglandin synthesis; effective for dysmenorrhea
- Start at onset of menses or cramping, continue up to 3 days
- COX-2 inhibitors — effective; reduce risk of GI side effects
- Opioids: avoid long-term; consider pain clinic referral for refractory chronic pelvic pain
2. Hormonal Medical Therapy
All hormonal therapies aim to suppress ovulation and reduce estrogen stimulation of implants. None are curative — symptoms typically recur after stopping.
| Agent | Dose | Notes |
|---|---|---|
| Combined OCP (continuous or cyclic) | Standard dose OCP continuously | First-line; reduces dysmenorrhea; prevents endometrioma recurrence post-surgery (94% free at 36 months vs. 51% without OCP). Cyclic and continuous use both effective |
| Progestins — norethindrone acetate | 5 mg/day orally | Effective for pain; fewer estrogen-related side effects; well-tolerated long-term |
| DMPA (depot MPA) | 150 mg IM q3 months | Induces amenorrhea; effective for pain; monitor bone density |
| LNG-IUS (Mirena) | In-office insertion | Reduces dysmenorrhea and chronic pelvic pain; local delivery; fertility returns after removal |
| GnRH agonists (leuprorelin, triptorelin, nafarelin) | Leuprorelin 3.75 mg SC monthly or 11.25 mg q3 months | Creates medical "pseudomenopause"; highly effective. Side effects: hot flushes (78%), vaginal dryness, bone loss. Add-back therapy (low-dose estrogen ± progestin) recommended for use >6 months |
| GnRH antagonists (elagolix, relugolix) | Elagolix 150 mg orally daily (up to 24 months) | Oral; rapid onset; dose-dependent hypoestrogenism. Elagolix FDA-approved for endometriosis pain |
| Danazol | 200–400 mg BD–TDS | Androgenic/antigonadotropic; effective but poorly tolerated (acne, hirsutism, hepatotoxicity); largely replaced by GnRH agents |
| Aromatase inhibitors (letrozole, anastrozole) ± add-back | Letrozole 2.5 mg/day | For refractory/recurrent disease; targets aromatase overexpression in implants; always combine with OCP or GnRH agonist to prevent ovarian stimulation |
Post-surgical hormonal suppression:
- Routine post-op GnRH agonist does not significantly reduce pain recurrence at 12 months vs. placebo alone
- Post-op OCP use significantly reduces endometrioma recurrence (36-month recurrence-free rate: 94% OCP users vs. 51% non-users) — recommended (Berek & Novak, p. 661–662)
3. Surgical Treatment
Indications:
- Failure of medical therapy
- Desire for fertility (infertility as primary complaint)
- Endometrioma >3 cm
- Severe/deep infiltrating endometriosis
- Diagnostic uncertainty
Laparoscopy is the gold standard (preferred over laparotomy — reduces adhesion formation):
| Procedure | Details |
|---|---|
| Fulguration/ablation of implants | Laser or diathermy; effective for superficial disease |
| Excision (laparoscopic) | Complete excision preferred over ablation for deep/DIE lesions; better long-term pain outcomes |
| Cystectomy (endometrioma) | Excision of cyst wall preferred over drainage alone — lower recurrence |
| Adhesiolysis | Restores pelvic anatomy; barrier agents (oxidised cellulose) may reduce re-adhesion |
| Presacral neurectomy (PSN) | Effective additional procedure for midline pain; requires high surgical skill; LUNA adds no benefit |
Laparoscopic surgery vs. diagnostic laparoscopy alone: OR 5.72 (95% CI 3.09–10.60) at 6 months; OR 7.72 (95% CI 2.97–20.06) at 12 months favouring surgical treatment. (Berek & Novak, p. 660)
Definitive surgery (hysterectomy ± bilateral salpingo-oophorectomy):
- Reserved for women who have completed childbearing with severe, refractory disease
- BSO reduces recurrence risk but causes surgical menopause — HRT decisions require individualisation
4. Management of Infertility
- Mild–moderate endometriosis: laparoscopic ablation/excision improves spontaneous pregnancy rates (NNT ~12)
- Severe/DIE: surgical resection before ART improves IVF outcomes
- Hormonal therapy does not improve fertility — suppress ovulation and fertility during treatment
- IVF/ICSI: recommended for infertility with advanced disease or failed conservative surgery
- Use EFI (Endometriosis Fertility Index) to counsel patients on expected pregnancy rates post-surgery
5. Special Populations
Adolescents:
- Empirical treatment (NSAIDs + OCP) first
- Laparoscopy if no response to 3–6 months of medical therapy (endometriosis found in up to 70% of adolescents with refractory pelvic pain)
- Subtle atypical lesions (red, clear, white) are more common than classic black lesions in this age group
Perimenopausal women:
- Symptoms usually improve after menopause (estrogen withdrawal)
- HRT after surgical menopause: add progestin to protect against stimulation of residual implants
Extragenital endometriosis:
- Thoracic (catamenial pneumothorax, haemoptysis): hormonal suppression ± surgical pleurodesis
- Umbilical/scar endometriosis: excision
- Bowel/bladder DIE: multidisciplinary surgical approach (colorectal + gynecology)
Malignant Transformation
- Endometriosis-associated ovarian cancer (primarily clear cell carcinoma and endometrioid adenocarcinoma) occurs in ~1% of cases
- Risk mainly confined to deep infiltrating and ovarian endometriosis
- Surveillance: ultrasound monitoring for endometriomas; rising CA-125 warrants reassessment
Summary Algorithm
Suspected endometriosis (dysmenorrhea ± pelvic pain ± infertility)
↓
First-line: NSAIDs + combined OCP (3–6 months)
↓ if inadequate response
Add or switch: GnRH agonist + add-back / GnRH antagonist / progestins
↓ if inadequate response or infertility/diagnostic uncertainty
Laparoscopy: confirm diagnosis + surgical treatment (excision preferred)
↓ post-surgery
Continuous OCP or LNG-IUS (reduces recurrence)
↓ if severe refractory disease + childbearing complete
Definitive surgery (hysterectomy ± BSO) + individualised HRT
Sources:
- Robbins & Kumar Basic Pathology, pp. 688–689 (pathogenesis, morphology)
- Berek & Novak's Gynecology, pp. 651–665 (management, surgical outcomes, post-op hormonal therapy)
- Goldman-Cecil Medicine, pp. 2554–2555 (clinical features, treatment options)
Recent evidence notes:
- A 2026 systematic review (PMID 40705433) found physiotherapy significantly reduces endometriosis-associated pelvic pain — consider as adjunct.
- A 2025 meta-analysis (PMID 39946383) supports physical activity/exercise as effective for reducing pain and improving quality of life in endometriosis.
- A 2025 meta-analysis (PMID 39358652) on dietary interventions (anti-inflammatory diet, omega-3) showed modest benefit for pain reduction.
This is a shared conversation. Sign in to Orris to start your own chat.