How to get normal IVS thickness and LVPW thickness normal
Reading File
Finding Sources
Finding Sources
Searching the Web
normal IVS and LVPW thickness echocardiography values ASE guidelines
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
I now have comprehensive data from both textbook sources and current ASE/EACVI guidelines. Here is a complete reference:
Normal IVS and LVPW Thickness — Echocardiography Reference Values
How They Are Measured
Both IVS (interventricular septal thickness) and LVPW (left ventricular posterior wall thickness) are measured at end-diastole (onset of QRS complex), using the leading-edge to leading-edge technique on M-mode or 2D-guided M-mode from the parasternal long-axis or short-axis view. The M-line must be perpendicular to the long axis of the ventricle, positioned just below the mitral leaflet tips at the level of the chordae.
Normal Reference Values (ASE/EACVI 2015 Guidelines)
These are the most widely used clinical thresholds, stratified by sex:
| Parameter | Women — Normal | Men — Normal |
|---|---|---|
| IVSd (septal thickness, end-diastole) | 0.6 – 1.0 cm | 0.6 – 0.9 cm |
| LVPWd (posterior wall thickness, end-diastole) | 0.6 – 1.0 cm | 0.6 – 0.9 cm |
Grading of Abnormal Thickness (ASE/EACVI)
| Grade | Women (cm) | Men (cm) |
|---|---|---|
| Normal | 0.6 – 1.0 | 0.6 – 0.9 |
| Mild hypertrophy | 1.1 – 1.3 | 1.0 – 1.2 |
| Moderate hypertrophy | 1.4 – 1.6 | 1.3 – 1.5 |
| Severe hypertrophy | > 1.6 | > 1.5 |
Age- and Sex-Stratified Values (JACC Imaging, 2022 — updated normative data)
Mean ± SD at end-diastole in mm:
| Women | Men | |||||||
|---|---|---|---|---|---|---|---|---|
| 20–39 y | 40–59 y | 60–79 y | ≥80 y | 20–39 y | 40–59 y | 60–79 y | ≥80 y | |
| IVSd (mm) | 6.8 ± 1.2 | 7.4 ± 1.3 | 8.1 ± 1.5 | 8.2 ± 1.0 | 7.9 ± 1.3 | 8.7 ± 1.3 | 9.2 ± 1.5 | 9.3 ± 1.7 |
| LVPWd (mm) | 6.6 ± 0.9 | 6.9 ± 1.0 | 7.5 ± 1.1 | 7.7 ± 1.2 | 7.3 ± 1.0 | 8.0 ± 1.2 | 8.3 ± 1.2 | 8.2 ± 0.8 |
Key points: wall thickness increases with age and is greater in men than women at every age group (both p < 0.001). This means an IVS of 9 mm in a 70-year-old man is within normal limits, while the same value would be borderline in a young woman.
Upper Limits of Normal (95th Percentile)
| Women | Men | |||||||
|---|---|---|---|---|---|---|---|---|
| 20–39 y | 40–59 y | 60–79 y | ≥80 y | 20–39 y | 40–59 y | 60–79 y | ≥80 y | |
| IVSd (mm) | 9.3 | 10.0 | 11.0 | 10.2 | 10.5 | 11.3 | 12.1 | 12.5 |
| LVPWd (mm) | 8.4 | 9.0 | 9.7 | 10.0 | 9.2 | 10.3 | 10.7 | 9.8 |
Clinical Significance
- IVSd ≥ 13 mm (anterior septum or posterior wall) in a first-degree relative of an HCM patient = major diagnostic criterion for hypertrophic cardiomyopathy (Goldman-Cecil Medicine)
- Relative Wall Thickness (RWT) = 2 × PWTd / LVIDd
- Normal: < 0.42
- Helps classify LV geometry: concentric remodeling, concentric hypertrophy, or eccentric hypertrophy
- LV mass formula (M-mode): LV mass = 0.80 × [1.04 (STd + PWTd + LVIDd)³ − LVIDd³] + 0.6 g
Practical Summary
For most adults in routine clinical practice:
- Normal IVSd and LVPWd: 6–10 mm (women slightly lower than men)
- Simple upper limit of normal: 11 mm for women, 10–11 mm for men (older adults may go to 12 mm)
- Values are equal for IVS and LVPW — asymmetric hypertrophy (IVS/LVPW ratio > 1.3–1.5) raises concern for HCM
Sources: Textbook of Clinical Echocardiography (Otto, 9780323882088); ASE/EACVI 2015 Chamber Quantification Guidelines; JACC Imaging 2022 normative data; TECHmED ASE reference values
What are treatment of abnormal IVS thickness and abnormal LVPw thickness
Finding Sources
Finding Sources
Searching the Web
treatment abnormal IVS LVPW thickness echocardiography LVH HCM guidelines 2024
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Loading Image
Excellent data gathered. Here is a comprehensive treatment guide:
Treatment of Abnormal IVS and LVPW Thickness
Abnormal IVS/LVPW thickness (i.e., LV wall hypertrophy) is not a single disease — it is a phenotype with multiple underlying causes, each requiring a different treatment approach. The first step is always to identify the etiology.
Step 1 — Identify the Cause
| Cause | Key Features |
|---|---|
| Hypertensive heart disease | Most common; symmetric or mild asymmetric LVH; history of HTN |
| Hypertrophic Cardiomyopathy (HCM) | Often asymmetric; IVS > LVPW; ± LVOTO; younger patients; family history |
| Aortic stenosis | Concentric LVH; systolic murmur; reduced AVA on echo |
| Athlete's heart | Mild LVH (usually < 13 mm); reversible; normal diastolic function |
| Infiltrative disease | Amyloid (granular sparkling echo), Fabry, sarcoid — specific patterns on CMR |
| End-stage renal disease / metabolic | CKD, dialysis, acromegaly, diabetes |
2. Hypertensive LVH (Most Common Cause)
Goal: BP control to regress LV mass
Non-pharmacologic (first-line for all stages):
- Weight reduction (most effective single intervention)
- DASH diet / low sodium (< 2.4 g/day)
- Regular aerobic exercise
- Limit alcohol
- Smoking cessation
Antihypertensive Drugs — LVH Regression (ranked by evidence):
| Drug Class | Effect on LVH | Notes |
|---|---|---|
| ARBs (losartan, valsartan) | ✅✅✅ Best regression | LIFE trial: superior to atenolol for LVH regression |
| ACE inhibitors (ramipril, enalapril) | ✅✅✅ Excellent | Also prevent LVH development; renoprotective |
| Calcium channel blockers (amlodipine) | ✅✅ Good | Effective in elderly and isolated systolic HTN |
| Diuretics (chlorthalidone) | ✅✅ Good | Add-on in resistant HTN |
| Beta-blockers (atenolol) | ✅ Modest | Less effective for LVH regression than ARBs/ACEi |
Key point from Harrison's: In RCTs, antihypertensive therapy achieves regression of LV mass and prevention of LVH — ARBs and ACEi are preferred agents (Harrison's Principles of Internal Medicine 22E).
3. Hypertrophic Cardiomyopathy (HCM) — Asymmetric IVS > LVPW
Treatment depends on whether LVOTO (LV outflow tract obstruction) is present.
HCM Management Flowchart (Braunwald's Heart Disease / Harrison's 22E)
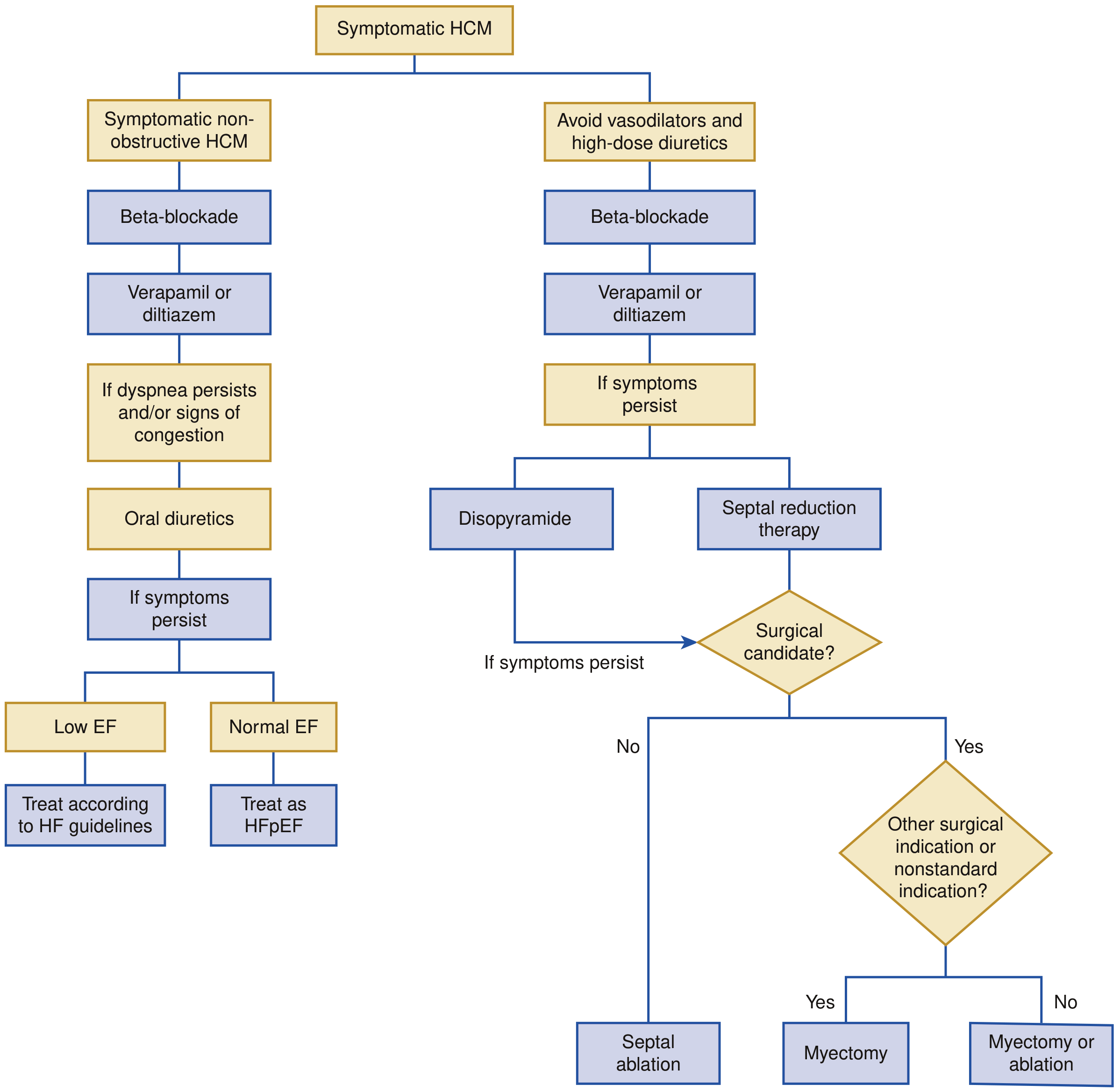
A. Obstructive HCM (LVOTO gradient ≥ 30 mmHg)
Avoid: vasodilators (nitrates, dihydropyridine CCBs), high-dose diuretics, digoxin — all worsen obstruction.
Step 1 — Beta-blockers (e.g., metoprolol, propranolol)
- Reduce heart rate → prolong diastolic filling → reduce obstruction
- Negative inotropic effect reduces dynamic obstruction
Step 2 — Verapamil or Diltiazem (if beta-blockers not tolerated or insufficient)
- Non-dihydropyridine CCBs; negative chronotropy + inotropy
- ⚠️ Use with caution if severe obstruction or elevated filling pressures
Step 3 — Add Disopyramide (to beta-blocker or CCB)
- Potent negative inotrope; reduces LVOTO gradient
- Monitor QTc prolongation
Step 4 — Mavacamten (FDA-approved, 2022)
- Cardiac myosin ATPase inhibitor → reduces actin-myosin cross-bridge formation
- Directly reduces contractility and LVOTO
- High efficacy including in beta-blocker-refractory patients
- Aficamten — under investigation, similar mechanism
Step 5 — Septal Reduction Therapy (for refractory severe symptoms, ~5% of patients):
- Surgical septal myectomy: Gold standard; excision of proximal septal myocardium via aortotomy; durable results; very low mortality in experienced centers
- Alcohol septal ablation: Controlled infarction of proximal septum via catheter-injected ethanol; for poor surgical candidates or patient preference
- Both carry risk of complete heart block requiring permanent pacemaker
B. Non-Obstructive HCM
- Beta-blockers (first-line for symptoms)
- Verapamil/diltiazem (alternative)
- Diuretics (for fluid congestion — use cautiously)
- If low EF develops: treat per heart failure with reduced EF (HFrEF) guidelines
- If preserved EF with congestion: treat as HFpEF
C. Sudden Death Prevention in HCM
| Major Risk Factor | Notes |
|---|---|
| Prior cardiac arrest or sustained VT | Strongest indication for ICD |
| Family history of HCM-related SCD | |
| Massive LVH (wall thickness ≥ 30 mm) | |
| Unexplained syncope | |
| Abnormal BP response to exercise | |
| NSVT on Holter |
- ICD implantation is recommended for patients with ≥1 major risk factor
- ESC risk calculator (HCM Risk-SCD) uses continuous variables (LVOTO gradient, LA size, etc.)
- Shared decision-making is essential given low positive predictive value of individual risk factors
4. Aortic Stenosis (Pressure Overload LVH)
- Definitive treatment is aortic valve replacement (AVR) — surgical (SAVR) or transcatheter (TAVR)
- Medical therapy does not slow AS progression
- After AVR, LVH regresses over months to years
- ACEi/ARBs may help manage BP but do not treat the stenosis
5. Infiltrative Causes (Amyloid, Fabry, Sarcoid)
| Cause | Specific Treatment |
|---|---|
| Cardiac amyloidosis (ATTR) | Tafamidis (stabilizes transthyretin); patisiran/inotersen (hATTR with neuropathy) |
| Fabry disease | Enzyme replacement therapy (agalsidase alfa/beta); migalastat (amenable variants) |
| Cardiac sarcoidosis | Systemic corticosteroids; ICD if complete heart block or sustained VT |
Summary Table
| Etiology | Primary Treatment | Additional |
|---|---|---|
| HTN-LVH | BP control: ARB/ACEi + lifestyle | Target BP < 130/80 |
| Obstructive HCM | Beta-blocker → CCB → disopyramide → mavacamten → septal reduction | Avoid vasodilators; ICD if SCD risk |
| Non-obstructive HCM | Beta-blocker/CCB; diuretics for congestion | ICD if SCD risk |
| Aortic stenosis | AVR (SAVR or TAVR) | |
| ATTR amyloid | Tafamidis | |
| Fabry | Enzyme replacement |
Sources: Braunwald's Heart Disease, 10e (Fig. 54.10); Harrison's Principles of Internal Medicine, 22E (2025); Goldman-Cecil Medicine; 2024 AHA/ACC HCM Guideline
Time duration of ARB and ACE inhibhitors taken by 45 years old female old thyroid patient
Asking for Preferences
This is a shared conversation. Sign in to Orris to start your own chat.