Shockwave the mechanisms how it works infication and contraindications and application
Reading File
Finding Sources
Finding Sources
Searching the Web
shockwave therapy mechanism of action indications contraindications clinical application
Finding Sources
Reading Web Page
shockwave therapy mechanism indications contraindications application
Searching PubMed
extracorporeal shockwave therapy musculoskeletal mechanism
Searching Images
shockwave therapy mechanism radial focused ESWT diagram
Reading Web Page
shockwave therapy types mechanism of action biological effects cellular
I now have comprehensive information to provide a thorough answer. Let me compile it.
Shockwave Therapy (ESWT) — Complete Clinical Overview
What Is Shockwave Therapy?
Extracorporeal Shockwave Therapy (ESWT) delivers high-energy acoustic pressure waves from outside the body into targeted tissue. A "shockwave" is a single pressure pulse characterized by a rapid rise to a high positive peak pressure (up to 100 MPa), followed by a brief tensile (negative) phase — all within microseconds.
Types of Shockwaves
| Feature | Focused ESWT | Radial Pressure Wave (RSWT) |
|---|---|---|
| Generation | Electrohydraulic, electromagnetic, or piezoelectric; focal point away from applicator | Compressed-air ballistic projectile striking a metal applicator |
| Pressure peak | High (at the focal point, deep in tissue) | Maximal at skin surface, diverges with depth |
| Depth of penetration | 3–12 cm (adjustable focus) | Superficial (~3 cm) |
| Energy type | True shockwave | Technically a pressure wave (radial wave, NOT a true shockwave) |
| Best for | Deep structures: tendon insertions, bone, non-union, AVN | Superficial soft tissue, myofascial trigger points |
Mechanism of Action
ESWT acts through mechanotransduction — mechanical stimuli converted into biochemical signals. Multiple parallel mechanisms operate:
1. Mechanical Effects
- Rapid positive pressure causes energy absorption, reflection, refraction, and transmission in tissue
- Destroys calcific deposits in tendons (e.g., calcific tendinopathy of the shoulder) by physical disintegration
- Induces microcavitation: formation and collapse of microbubbles generating microshear forces that stimulate cell membranes
2. Cellular & Molecular Effects
- ATP release → activates extracellular signal-regulated kinase (ERK) pathways → enhances cell proliferation and wound healing
- Nitric oxide (NO) release → downstream analgesic, anti-inflammatory, and angiogenic effects
- Alteration of ion channels in cell membranes
- Upregulation of growth factors:
- TGF-β1 and IGF-1 → increased collagen synthesis
- VEGF → neovascularization
- BMP-2 → bone healing/osteogenesis
- Increased IL-6, IL-8, MMP-2, MMP-9 → matrix remodeling
- Increased glycosaminoglycans and tenocyte proliferation
- Osteoprogenitor cell differentiation → bone remodeling
3. Analgesic Effects
- Stimulation of nociceptive C-fibers → hyperstimulation → temporary nerve block (hyper-stimulation analgesia)
- Gate-control theory: activates large afferent fibers inhibiting pain transmission
- Depletion of Substance P → reduced neurogenic inflammation
- Reduced calcitonin gene-related peptide (CGRP) at nerve endings
4. Angiogenesis & Tissue Repair
- Promotes neovascularization via VEGF and NO
- Activates endogenous stem cells (CD31/CD34-positive endothelial precursors)
- Stimulates collagen remodeling and tendon repair
Indications
Tendon Pathologies
- Calcific tendinopathy of the shoulder (highest evidence)
- Lateral epicondylopathy (tennis elbow)
- Plantar fasciitis / plantar fascopathy (strong evidence)
- Achilles tendinopathy
- Patellar tendinopathy (jumper's knee)
- Greater trochanteric pain syndrome (gluteal tendinopathy)
- Hamstring, adductor, peroneal, distal biceps tendinopathies
- Rotator cuff tendinopathy (non-calcific)
Bone Pathologies
- Delayed fracture healing / non-union
- Stress fractures
- Avascular necrosis (osteonecrosis) of the femoral head
- Osteochondritis dissecans
- Medial tibial stress syndrome
- Osgood-Schlatter disease
Muscle/Soft Tissue
- Myofascial pain syndrome / trigger points
- Muscle strain (without fiber discontinuity)
Urological
- Erectile dysfunction (low-intensity ESWT)
- Peyronie's disease (limited evidence per urology guidelines)
Wound Healing
- Chronic diabetic foot ulcers
- Pressure ulcers
- Burns (2nd degree)
Other Emerging
- Cardiac ischemia (low-intensity, research stage)
- Neurological conditions (post-stroke spasticity)
- Dupuytren's disease
Note: ESWT is typically indicated after failure of conservative treatment (rest, physiotherapy, NSAIDs for 3–6 months).
Contraindications
Per the International Society for Medical Shockwave Treatment (ISMST, 2023 Guidelines):
Absolute Contraindications (All Energy Levels)
| Contraindication | Rationale |
|---|---|
| Malignant tumor in the shockwave field | Risk of tumor dissemination/stimulation |
| Fetus/pregnancy (fetus in field) | Risk of fetal harm |
| Pacemaker / implantable defibrillator in the shockwave field | Device malfunction |
| Active infection / osteomyelitis at treatment site | Risk of spreading infection |
Additional Absolute Contraindications for High-Energy Focused ESWT
| Contraindication |
|---|
| Lung tissue in the shockwave field |
| Brain/CNS/spinal cord in the shockwave field |
| Epiphyseal plates (growing bone) in children / skeletal immaturity |
| Severe coagulopathy |
Relative / Important Considerations
- Current NSAID use or anticoagulant use (warfarin, heparin) — increased bleeding risk
- Corticosteroid injection within 6 weeks at the same site
- Blood thinning medications
- Skull bones, vertebral bodies, ribs — exercise caution with focused high-energy
Note: Cancer itself in a different/remote area is NOT an absolute contraindication. Similarly, chronic infection in wound care is increasingly showing benefit — guidelines may evolve.
Clinical Application Protocol
Before Treatment
- Confirm indication and failure of prior conservative treatment
- Screen for contraindications (imaging to confirm diagnosis — ultrasound or X-ray)
- Informed consent — explain: onset of effect after 4–12 weeks, potential temporary increase in pain, bruising
- Identify anatomical target by palpation ± ultrasound guidance
Treatment Parameters
| Parameter | Focused ESWT | Radial ESWT |
|---|---|---|
| Energy Flux Density (EFD) | 0.08–0.30 mJ/mm² | 1.5–4 bar (pressure) |
| Impulses per session | 1,000–4,000 | 1,500–3,000 |
| Frequency | 1–4 Hz | 6–15 Hz |
| Sessions | 3–5 (weekly or biweekly) | 3–6 (weekly) |
| Coupling medium | Ultrasound gel (mandatory) | Ultrasound gel |
| Anesthesia | Generally avoided (blocks biofeedback; may mask dose titration) | Not needed |
During Treatment
- Patient positioned comfortably exposing the target area
- Ultrasound gel applied to skin
- Applicator held perpendicular to tissue surface
- Energy titrated to patient tolerance (biofeedback guides dosing)
- For trigger points: "referred pain recognition" confirms correct localization
After Treatment
- Avoid NSAIDs for 1–2 weeks post-treatment (inflammation is part of the healing response)
- Patients should expect temporary soreness for 24–48 hours
- No immobilization required; gradual return to activity
- Reassess at 4–6 weeks; repeat treatment cycle if partial response
Side Effects (Generally Mild, Transient)
- Local pain during/after treatment
- Skin redness, petechiae, or bruising
- Transient swelling
- Rare: hematoma, nerve irritation
Clinical Images
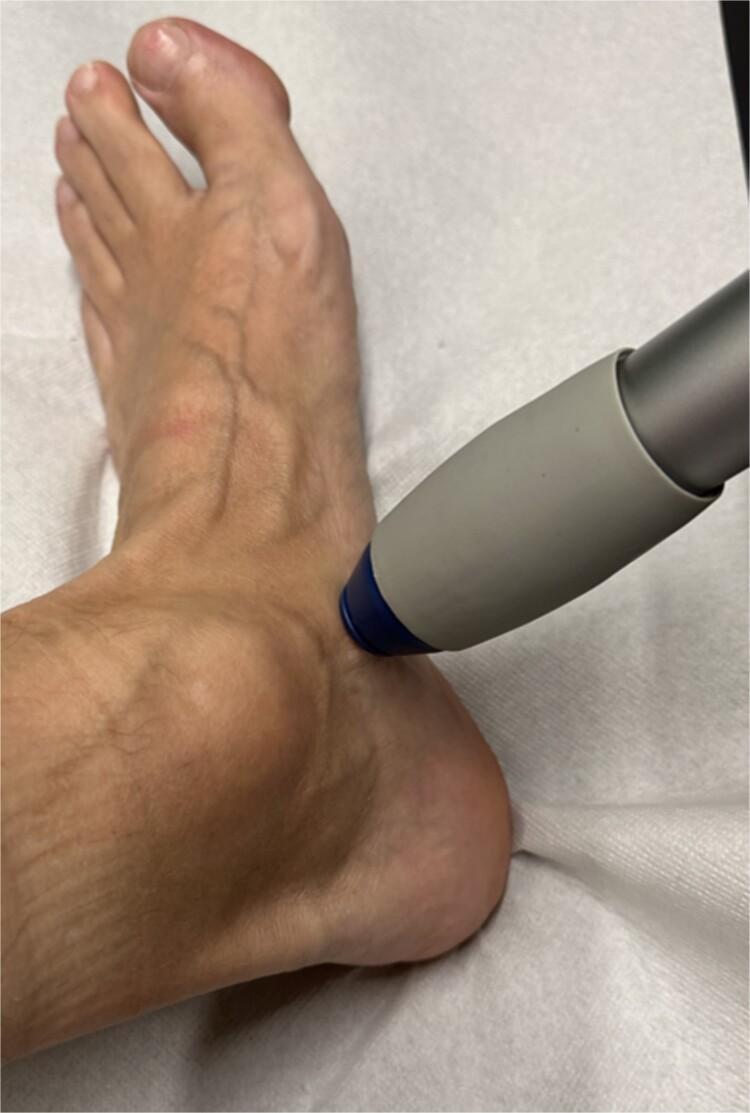
Radial ESWT applied to the medial ankle targeting the tibialis posterior tendon — note the cylindrical applicator with coupling gel and precise anatomical placement.
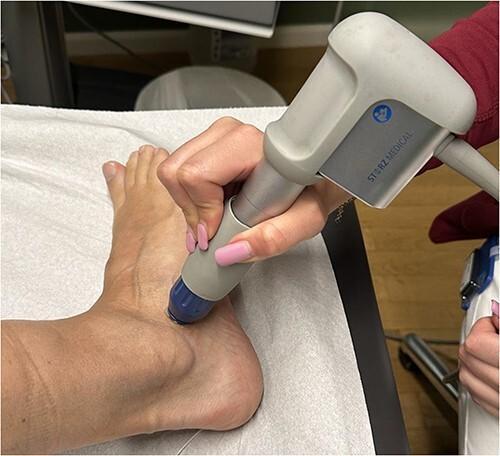
STORZ MEDICAL radial ESWT applied to the lateral malleolus/peroneal tendon region.
Key Takeaway Summary
| Domain | Key Point |
|---|---|
| Physics | Acoustic pressure pulse — rapid rise, brief tensile tail |
| Core mechanism | Mechanotransduction → angiogenesis, collagen synthesis, pain modulation |
| Top indication | Calcific shoulder tendinopathy, plantar fasciitis, lateral epicondylopathy |
| Strongest contraindication | Tumor/pacemaker/fetus in shockwave field |
| Energy level matters | High-energy focused ESWT carries more contraindications than low/radial |
| Analgesia during Tx | Avoid local anesthesia — it removes biofeedback and may reduce efficacy |
| Onset of effect | Weeks to months — not immediate |
Sources: ISMST ESWT Guidelines (2023) · Best Practices in Musculoskeletal ESWT (PMC9321712) · Mayo Clinic / Sports Medicine ESWT Review · Physiotherapy Canada Safety Guidelines (2020)
How it is helpful in avn
Finding Sources
Finding Sources
Searching PubMed
shockwave therapy avascular necrosis femoral head
Searching the Web
shockwave therapy AVN avascular necrosis femoral head mechanism efficacy clinical outcomes
Reading Article
Reading Article
Reading Article
Searching Images
avascular necrosis femoral head MRI shockwave treatment stages
Excellent data. Here is a comprehensive evidence-based answer:
Shockwave Therapy in Avascular Necrosis (AVN) of the Femoral Head
Understanding the Problem — Why AVN Needs ESWT
AVN (osteonecrosis of the femoral head, ONFH) results from interruption of blood supply to the femoral head → ischemia → bone cell death → subchondral collapse → joint destruction. The core pathological drivers are:
- Impaired angiogenesis (no new vessels reach the necrotic zone)
- Osteoblast-osteoclast imbalance (resorption exceeds formation)
- Bone marrow edema (BME) → increased intraosseous pressure → further ischemia
- Mesenchymal stem cell (MSC) dysfunction → impaired bone regeneration
ESWT directly targets all four of these mechanisms.
How ESWT Works in AVN — Specific Mechanisms
1. Angiogenesis (New Blood Vessel Formation)
This is the most critical mechanism in AVN. ESWT:
- Upregulates VEGF (Vascular Endothelial Growth Factor) → stimulates new capillary sprouting into the necrotic zone
- Releases nitric oxide (NO) → vasodilation + angiogenic signaling
- Mobilizes CD31/CD34+ endothelial progenitor cells from bone marrow → they migrate to the necrotic area and form new vessels
- High-energy ESWT specifically upregulates localized NO to promote angiogenesis in bone, which is why high energy is required for bone lesions
2. Osteogenesis (New Bone Formation)
- Stimulates BMP-2 (Bone Morphogenetic Protein-2) → drives osteoprogenitor differentiation into osteoblasts
- Upregulates TGF-β1 → bone matrix synthesis and collagen deposition
- Promotes MSC proliferation and osteoblast differentiation → direct bone repair in necrotic zone
- Studies of femoral heads removed at hip replacement show histological evidence of new bone formation and neovascularization after ESWT
3. Bone Marrow Edema (BME) Reduction
- Mechanical effect reduces intraosseous pressure
- Anti-inflammatory signaling reduces local edema
- Meta-analysis (Tan 2025, PMID 38896858): ESWT significantly improves BME in early ONFH (OR 4.35, 95% CI 1.32–14.37, P=0.02)
4. Pain Modulation
- C-fiber hyperstimulation → analgesia
- Substance P depletion → reduced neurogenic pain
- Gate-control mechanism
MRI Evidence of ESWT Response in AVN
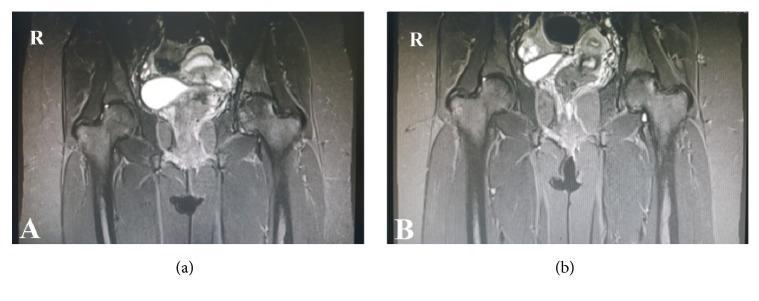
Coronal T1 MRI: (a) baseline Grade II ONFH with bone marrow edema; (b) 2-year post-ESWT showing regression to Grade I with marked reduction in edema — demonstrating radiological improvement after ESWT.
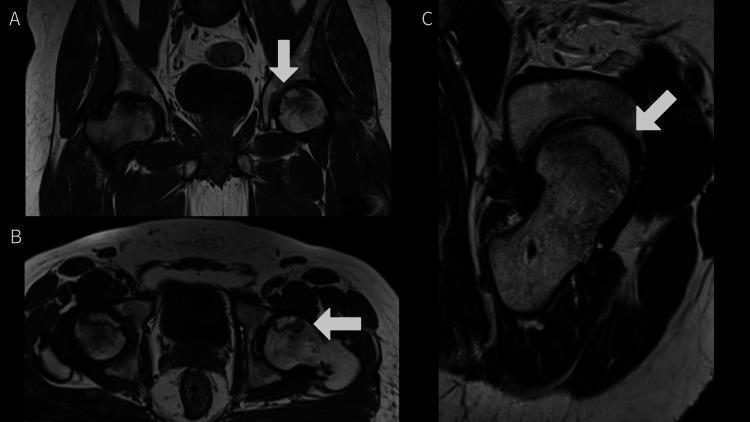
Multi-planar MRI showing bilateral ONFH at different stages — ESWT is most effective when applied before femoral head collapse (ARCO Stage I–II).
Clinical Evidence — What the Data Shows
Meta-Analysis: Mei et al. 2022 (PMID 34058957) — 9 studies, 409 patients
| Outcome | Result | Significance |
|---|---|---|
| Harris Hip Score (HHS) | Improved by 19.95 points | P < 0.01 ✓ |
| VAS pain score | Reduced by 2.77 points | P < 0.01 ✓ |
| MRI lesion area | Reduced (SMD 1.03) | P < 0.01 ✓ |
| Disease progression | Cannot reliably stop progression | Limited evidence |
Systematic Review: Tan et al. 2025 (PMID 38896858) — 9 studies, 716 patients
| Outcome | Result | Significance |
|---|---|---|
| HHS improvement | +33.38 points | P < 0.00001 ✓ |
| VAS | Dropped from >5 to 1.2 (reduced 4.64 pts) | P < 0.00001 ✓ |
| Necrosis area reduction | Trend toward reduction | P = 0.06 (NS) |
| ARCO stage change | No significant stage improvement | P = 0.60 (NS) |
| Bone marrow edema | Significantly improved | P = 0.02 ✓ |
Network Meta-Analysis: Zhai et al. 2024 (PMID 39234503) — 11 RCTs
- ESWT significantly reduced VAS vs. core decompression + bone grafting (MD -2.84, P significant)
- SUCRA ranking for HHS: ESWT alone ranked 3rd (58.3%), ESWT + injectable medication ranked 2nd (72.2%)
- Recommendation: ESWT alone for long-term/chronic patients; ESWT + injectable medication for acute/short-term onset
Long-Term Study (8–9 year follow-up)
ESWT was superior to core decompression + bone grafting in long-term clinical outcomes — advantages include non-invasiveness, no surgical risk, and comparable or better functional recovery.
ESWT vs. Other Treatments in AVN
| Treatment | Invasiveness | Best Stage | Outcome |
|---|---|---|---|
| ESWT | Non-invasive | ARCO I–II (early) | Good pain relief, functional improvement, BME reduction |
| Core decompression | Surgical | ARCO I–II | Similar or inferior to ESWT in long-term studies |
| Core decompression + bone graft | Surgical | ARCO I–III | More invasive, variable results |
| Cell therapy (MSC injection) | Minimally invasive | ARCO I–II | Best HHS (SUCRA 77%) but requires injection |
| Total hip arthroplasty | Major surgery | ARCO III–IV | Definitive but irreversible |
Stage-Specific Guidance
| ARCO Stage | Description | Role of ESWT |
|---|---|---|
| Stage I | MRI positive, X-ray normal, no collapse | Best response — BME resolves, pain relief excellent |
| Stage II | Sclerosis/cysts on X-ray, no collapse | Good response — functional improvement, may stabilize |
| Stage III | Subchondral fracture / "crescent sign" | Moderate benefit — may delay THA, pain relief still meaningful |
| Stage IV | Femoral head collapse + joint space narrowing | Limited benefit — THA likely needed |
Key principle: ESWT cannot reverse established collapse (Stage III–IV). It is most effective as early intervention to halt progression before structural failure.
Treatment Protocol for AVN
- Device: High-energy focused ESWT (not radial) — required to penetrate deep to the femoral head (6–10 cm)
- Energy density: 0.25–0.40 mJ/mm²
- Impulses: 2,000–4,000 per session
- Sessions: 3–5 sessions, 1 week apart
- Localization: Fluoroscopy or ultrasound guidance to target the necrotic zone precisely
- Coupling: Ultrasound gel over the hip
- Avoid NSAIDs for 2 weeks post-treatment
- Follow-up MRI at 3–6 months to assess BME response
Summary
ESWT works in AVN by restoring blood supply and promoting bone regeneration through angiogenesis (VEGF, NO), osteogenesis (BMP-2, TGF-β1, MSC activation), and BME reduction. It is a non-invasive, effective option for early-stage AVN (ARCO I–II) with strong evidence for pain relief and functional improvement, and emerging evidence for radiological stabilization. It does not reverse collapse in advanced disease but can delay or avoid total hip arthroplasty in appropriately selected patients.
Sources: Mei et al. 2022 (PMID 34058957) · Tan et al. 2025 (PMID 38896858) · Zhai et al. 2024 (PMID 39234503) · ISMST Guidelines 2023
This is a shared conversation. Sign in to Orris to start your own chat.