I now have rich content from multiple textbooks plus images. Let me compile the comprehensive notes:
🎓 PUBERTY — PART 1: DEFINITION & THEORIES
Exam-Focused Deep Notes | OB/GYN | Combined from Standard Textbooks
📌 QUICK RECALL BOX (For Last-Minute Revision)
| Key Point | Value |
|---|
| Puberty definition | Period when endocrine & gametogenic functions mature to allow reproduction |
| First event in girls | Thelarche (breast development) |
| First event in boys | Testicular enlargement |
| Normal age in girls | 8–13 years |
| Normal age in boys | 9–14 years |
| Age of adrenarche | 6–8 yrs (girls), 8–10 yrs (boys) |
| Menarche average age (USA) | 12.5 years |
| Master controller of puberty | GnRH (pulsatile) from hypothalamus |
| The "gatekeeper" neuropeptide | Kisspeptin (encoded by KISS1 gene) |
1. DEFINITION OF PUBERTY
Puberty is the transitional developmental period between childhood (juvenile state) and adulthood during which an individual becomes capable of reproduction. It involves physical, endocrine, and psychological maturation.
"Puberty, strictly defined, is the period when the endocrine and gametogenic functions of the gonads have first developed to the point where reproduction is possible." — Ganong's Review of Medical Physiology
There is an important distinction between:
- Adolescence — the broader period of final maturation of the reproductive system (social/psychological/physical)
- Puberty — the specific endocrine and gametogenic maturation enabling reproduction (narrower definition)
Two Core Physiological Processes of Puberty
Puberty fundamentally involves two parallel but distinct processes:
1. Gonadarche
- Physical and functional maturation of the gonads
- Gonads produce gametes (spermatozoa / ova) and sex steroids (estrogen, testosterone, progesterone)
- Driven by the HPG axis (hypothalamic–pituitary–gonadal)
- In males → leads to spermarche (onset of sperm production)
- In females → leads to folliculogenesis, thelarche, and menarche
2. Adrenarche
- Increased production of androgenic steroids by the adrenal cortex zona reticularis
- DHEA (dehydroepiandrosterone)
- DHEAS (DHEA sulfate)
- Androstenedione (A4)
- Occurs in both sexes during the prepubertal period (ages 6–8 years)
- Induces pubarche — growth of pubic and axillary hair
- Peculiar to humans and great apes only
- Mechanism controlling timing of adrenarche: unknown
- Adrenarche is independent of gonadarche — its absence does NOT prevent gonadarche or reproductive competence
- DHEA levels peak at ~age 25 in women (slightly later in men), then slowly decline in old age
- The rise in DHEA appears due to increased activity of 17α-hydroxylase
2. TERMINOLOGY / VOCABULARY OF PUBERTY
These terms appear frequently in exam questions — know each precisely:
| Term | Definition |
|---|
| Thelarche | Onset of breast development (first sign in girls, ~60% of cases) |
| Adrenarche | Onset of adrenal androgen secretion → pubic/axillary hair |
| Pubarche | Development of pubic hair (result of adrenarche) |
| Menarche | First menstrual period |
| Gonadarche | Activation and maturation of the gonads |
| Spermarche | Onset of sperm production in testes |
| Telarche | Synonymous with thelarche |
| Adolescence | Broader period of maturation (physical + psychological) |
Sequence of events in girls:
Thelarche → Pubarche/Adrenarche → Peak height velocity → Menarche
- Breast development precedes pubic hair in ~60% of girls
- Interval between thelarche and menarche: ~2 years
- Initial menstrual periods are typically anovulatory; regular ovulation appears ~1 year later
Sequence of events in boys:
Testicular enlargement (first sign) → Penile enlargement → Pubic hair → Axillary hair → Voice change → Spermarche → Peak height velocity
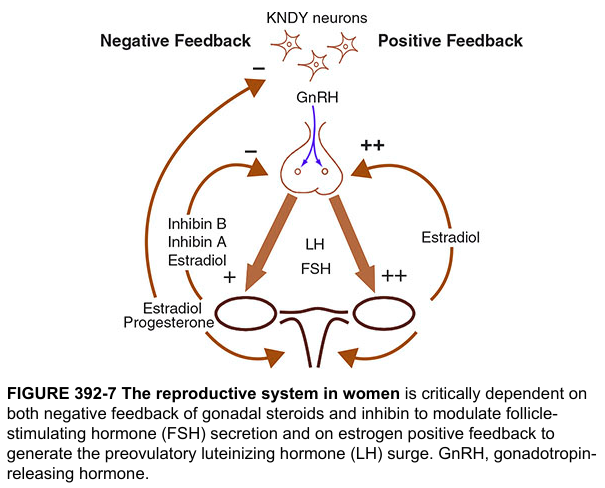
3. PHYSIOLOGY OF THE HPG AXIS — THE HORMONAL BASIS
Before understanding theories, understand the axis:
Hypothalamus
↓ GnRH (pulsatile)
Anterior Pituitary
↓ LH + FSH
Gonads (Ovary/Testis)
↓ Sex steroids (Estrogen / Testosterone / Progesterone)
Target organs + Negative/Positive feedback to Hypothalamus & Pituitary
- GnRH (gonadotropin-releasing hormone): decapeptide secreted by hypothalamic neurons
- Must be pulsatile — continuous GnRH causes downregulation of pituitary receptors → ↓ LH/FSH
- FSH: stimulates follicular development (granulosa cells) in females; Sertoli cells in males
- LH: triggers ovulation, stimulates estrogen/progesterone (theca cells); stimulates testosterone (Leydig cells) in males
- One of the earliest events of puberty = pulsatile GnRH release during REM sleep → gradually extends to waking hours
4. THEORIES / MECHANISMS OF PUBERTY
This is the most exam-important section. Multiple theories have been proposed. Modern understanding integrates all of them.
🔴 THEORY 1 — THE GONADOSTAT THEORY (Classic / Traditional)
Most asked in exams. Know thoroughly.
Proposed by: Grumbach and colleagues (1970s)
Core Concept:
- During childhood, the hypothalamic-pituitary system is exquisitely sensitive to negative feedback from gonadal sex steroids (even very low levels are sufficient to suppress GnRH/gonadotropin secretion)
- At puberty, this sensitivity DECREASES (the "gonadostat" is "reset" to a higher threshold)
- With reduced sensitivity to negative feedback → GnRH secretion increases → LH/FSH rise → gonadal activation
Analogy: Think of the gonadostat like a thermostat. In childhood, the thermostat is set very low (turns off the heater at minimal warmth). At puberty, the thermostat is reset higher — more heat (steroids) is needed to turn off the system.
Evidence for:
- In children, even small doses of sex steroids suppress LH/FSH
- In adults, much higher doses are needed for the same suppression
- ARC kisspeptin (Kiss1) expression is suppressed by even small amounts of estrogen in pre-pubertal animals; this suppression weakens at puberty
Limitation:
- Does NOT explain the juvenile pause (quiescence between infancy and puberty is under central suppression even in gonadectomized animals/children → the gonadostat cannot be the sole explanation)
- Bilateral gonadectomy in infants causes only a small increase in gonadotropins — meaning there must be a CNS-intrinsic inhibitory mechanism beyond just gonadal feedback
🔴 THEORY 2 — CENTRAL DRIVE / CNS INTRINSIC MATURATION THEORY
Now the predominant accepted mechanism
Core Concept:
- The onset of puberty is governed by intrinsic CNS maturation, independent of gonadal feedback
- The HPG axis is actively suppressed by the CNS during childhood
- At puberty, this suppression is lifted due to:
- ↑ Excitatory inputs to GnRH neurons
- ↓ Inhibitory inputs to GnRH neurons
- The balance shifts in favor of excitation, causing pulsatile GnRH secretion
Key evidence:
- Pulsatile GnRH injection in immature monkeys (with no gonads) → full pubertal development occurs and persists as long as pulsatile injections continue
- Gonadotropins are low in pre-pubertal children despite the gonads being capable of responding
- A neural mechanism (not just gonadal feedback) maintains the GnRH pulse generator in a suppressed state
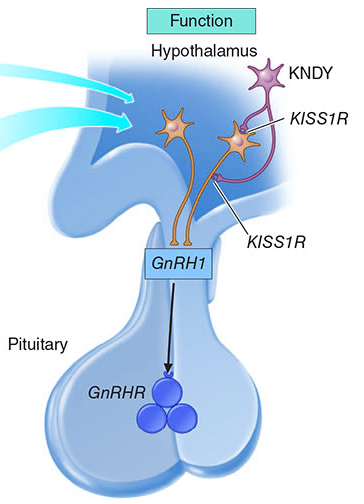
🔴 THEORY 3 — THE KISSPEPTIN / KNDy NEURON THEORY
Most modern, most imp for higher exams. Now integrates all previous theories.
This is the currently accepted master theory of puberty initiation.
The KISS1 System
- Kisspeptins = family of structurally related peptides encoded by the KISS1 gene (located on long arm of chromosome 1)
- Act via G protein-coupled receptor GPR54 (also called Kiss1R or KISS1 receptor)
- Produced by two populations of neurons in the hypothalamus:
- Arcuate nucleus (ARC) — also called infundibular nucleus in humans
- Anteroventral periventricular nucleus (AVPV) — in rodents; preoptic area in humans
KNDy Neurons (Key Concept)
KNDy = Kisspeptin + Neurokinin B + Dynorphin neurons
Located in the arcuate nucleus, these neurons:
- Neurokinin B (NKB): acts via NK3R receptor → stimulatory (excitatory) — causes synchronized discharge of KNDy neurons
- Dynorphin (Dyn): acts via KOR (kappa-opioid receptor) → inhibitory — terminates the burst
- Kisspeptin: released onto GnRH neurons → potently stimulates GnRH secretion
- Together, NKB and dynorphin create the oscillating/pulsatile pattern of GnRH release (the GnRH pulse generator)
How kisspeptin triggers puberty:
- During childhood: GnRH neurons are inhibited (via CNS mechanisms, partially via dynorphin, partially via other inhibitory neuropeptides)
- At puberty: Kisspeptin neurons mature → begin firing more robustly
- Kisspeptin binds GPR54 on GnRH neurons → ↑ GnRH pulse amplitude and frequency
- GnRH → ↑ LH and FSH → gonadal activation → sex steroid production → secondary sexual characteristics
Why kisspeptin matters clinically:
| Mutation | Effect |
|---|
| Loss-of-function mutation in KISS1 or KISS1R (GPR54) | Hypogonadotropic hypogonadism → absent pubertal development |
| Gain-of-function (activating) mutation in KISS1R or KISS1 | Central precocious puberty |
Kisspeptin as the bridge between gonadal feedback and GnRH:
- GnRH neurons lack estrogen and progesterone receptors (ER, PR)
- Kisspeptin neurons possess ER and PR
- Therefore: All sex steroid feedback (both negative and positive) to GnRH neurons is mediated THROUGH kisspeptin neurons
- This elegantly explains both negative feedback (suppression of GnRH) and positive feedback (LH surge) through kisspeptin intermediaries
🔴 THEORY 4 — CRITICAL BODY WEIGHT / FRISCH HYPOTHESIS
Simple but important — frequently asked
Proposed by: Rose Frisch (1970s)
Core Concept:
- A critical body weight (~47–48 kg) or more accurately a critical body fat percentage (~17% body fat) is required to trigger puberty
- Increased body fat → earlier puberty
- Body fat of ~22% needed for maintenance of menstrual cycles
Mechanism:
- Adipose tissue → peripheral conversion of androgens to estrogens (aromatase activity)
- Adipose tissue secretes leptin → acts on hypothalamus
- Leptin receptors are present on kisspeptin neurons and GnRH neurons
- ↑ Leptin → ↑ kisspeptin → ↑ GnRH pulsatility → puberty initiation
Clinical evidence:
- Girls with anorexia nervosa, athletes with very low body fat → delayed puberty / secondary amenorrhea
- Obese girls → earlier puberty (global secular trend)
- Children with leptin deficiency → fail to enter puberty → treat with leptin → puberty initiates
Limitation: Body weight is a permissive factor, not a direct trigger.
🔴 THEORY 5 — SKELETAL AGE / BONE AGE THEORY
- Bone age (skeletal maturation) correlates better with pubertal timing than chronological age
- Puberty typically initiates at a bone age of ~11 years in girls and ~12 years in boys
- Used clinically to assess pubertal status and to predict adult height
🔴 THEORY 6 — PINEAL GLAND / MELATONIN THEORY
Core Concept:
- The pineal gland secretes melatonin (inhibits GnRH)
- In children: long nights → high melatonin → suppresses HPG axis
- At puberty: decreasing melatonin levels → disinhibition of HPG axis
- Supported by: blind children (no light-dark signal) have altered pubertal timing; pineal tumors can cause precocious puberty (by destroying melatonin-producing cells)
Status: A contributing/permissive theory, not the primary trigger.
🔴 THEORY 7 — GENETIC / EPIGENETIC THEORY
High-yield for MCQs about precocious puberty genetics
Puberty timing is highly heritable (twin studies: heritability 50–80%)
Key genes involved:
| Gene | Role |
|---|
| KISS1 | Encodes kisspeptin; activating mutations → precocious puberty |
| KISS1R (GPR54) | Kisspeptin receptor; activating mutations → precocious puberty; loss-of-function → HH |
| MKRN3 | Maternally imprinted gene; loss-of-function → familial precocious puberty (most common genetic cause of CPP) |
| TACR3 | Encodes NK3R; mutations → IHH |
| TAC3 | Encodes neurokinin B; mutations → IHH |
| LIN28B | Sentinel puberty gene; potent regulator of microRNA processing; regulates timing |
| POU1F1 | Pituitary development/function |
| ESR1 | Estrogen receptor α; gonadal feedback |
| STARD4, RXRG | Hormone synthesis/bioactivity |
| INHBA | Inhibin B; gonadal feedback |
MKRN3 (Makorin RING Finger Protein 3):
- Maternally imprinted → only paternally expressed allele is active
- Acts as a repressor of puberty (like a "brake")
- Loss-of-function mutation → brake is removed → precocious puberty
- Explains why family history of precocious puberty is often paternal
Epigenetic mechanisms:
- Epigenetic remodeling of genes (DNA methylation, histone modification) regulates the timing of kisspeptin neuron maturation
- This may explain the secular trend toward earlier puberty despite unchanged genetic code
5. PHASES OF HPG AXIS ACTIVATION (Reactivation Theory)
The HPG axis goes through multiple phases of activation and quiescence from fetal life to adulthood:
| Phase | Timing | HPG Activity | Notes |
|---|
| Phase 1 (Fetal) | Fetal life (mid-gestation) | HIGH | GnRH pulse generator active; LH/FSH detectable in cord blood |
| Phase 2 (Neonatal/Mini-puberty) | Birth to ~6 months | HIGH | Second surge of LH/FSH; estrogen in females; testosterone in males; called "mini-puberty of infancy"; function unclear |
| Phase 3 (Juvenile pause) | 6 months to ~8–10 years | LOW (quiescent) | HPG axis dormant; FSH > LH but both low; GnRH erratic, low amplitude; CNS active suppression |
| Phase 4 (Puberty / Reactivation) | 8–13 yrs girls, 9–14 yrs boys | RISING | Pulsatile GnRH resumes during REM sleep → extends to daytime → full activation |
| Phase 5 (Adult) | Post-puberty | FULL cyclical | Regular cycles in females; sustained testosterone in males |
Important exam point: The juvenile pause is under CNS intrinsic suppression — this is proven by the fact that gonadectomy (removing gonads) during childhood causes only a small increase in gonadotropins. If the gonadostat were the only mechanism, gonadectomy would cause massive gonadotropin rise. This disproves a pure gonadostat theory as the sole explanation for childhood quiescence.
6. FACTORS INFLUENCING TIMING OF PUBERTY
Factors Causing Earlier Puberty:
- Obesity (↑ body fat → ↑ leptin → ↑ kisspeptin)
- Better nutrition (secular trend — 1–3 months earlier per decade for >175 years)
- Female sex (girls enter puberty 1–2 years earlier than boys)
- African American ethnicity (adrenarche and thelarche ~1 year earlier vs. white girls)
- Endocrine disruptors (environmental estrogens, bisphenol A, phthalates)
- Stress (can activate HPG axis via CRH/catecholamines)
- Blindness (disrupted melatonin → earlier puberty)
- Gain-of-function mutations in KISS1/KISS1R
- MKRN3 loss-of-function (paternal inheritance)
- CNS lesions (hypothalamic hamartoma — most common cause of CPP)
Factors Causing Later/Delayed Puberty:
- Malnutrition / underweight / anorexia
- Excessive exercise (athletes, ballet dancers)
- Chronic illness (Crohn's, celiac, renal failure)
- Leptin deficiency (congenital)
- Loss-of-function mutations in KISS1/KISS1R/TACR3/TAC3
- Kallmann syndrome (GnRH deficiency + anosmia)
- Constitutional delay of growth and puberty (CDGP) — most common cause
7. HORMONAL CHANGES DURING PUBERTY — DETAILED
In Females:
| Hormone | Change | Effect |
|---|
| GnRH | ↑ pulsatile (starts during REM sleep) | Drives whole axis |
| LH | ↑ (especially night pulses initially) | Ovarian theca cells → androgen production; triggers ovulation |
| FSH | ↑ | Granulosa cells → estrogen; folliculogenesis |
| Estradiol (E2) | ↑ | Thelarche, uterine growth, menarche, epiphyseal fusion |
| Progesterone | ↑ (post-ovulatory) | Luteal phase; pregnancy maintenance |
| DHEA / DHEAS | ↑ (adrenarche) | Pubarche |
| GH | ↑ | Linear growth, IGF-1 ↑ |
| IGF-1 | ↑ | Bone growth (growth spurt) |
| Insulin | Mild resistance | Physiological insulin resistance of puberty |
In Males:
| Hormone | Change | Effect |
|---|
| LH | ↑ | Leydig cells → testosterone |
| FSH | ↑ | Sertoli cells → spermatogenesis; inhibin B |
| Testosterone | ↑ | Genital growth, muscle mass, voice, libido, spermarche |
| GH | ↑ | Linear growth (more pronounced than girls) |
| IGF-1 | ↑ | Growth spurt (boys gain ~28 cm during pubertal growth) |
| DHEA | ↑ (adrenarche) | Pubarche |
8. SECULAR TREND IN PUBERTY
- There has been a gradual decline in the age of puberty in Europe and USA at a rate of 1–3 months per decade for >175 years
- In the USA:
- Puberty in girls: 8–13 years
- Puberty in boys: 9–14 years
- Menarche: average age 12.5 years
- Recent data: ↓ in age of thelarche but NOT menarche, associated with obesity
- Adrenarche in girls: ~1 year earlier in Black girls vs. White girls
9. EXAM ONE-LINERS & HIGH-YIELD FACTS
🔑 "The master key of puberty is KISSPEPTIN"
- Kisspeptin is encoded by KISS1 gene on chromosome 1 (long arm)
- KNDy = Kisspeptin + Neurokinin B + Dynorphin (all in arcuate nucleus)
- NKB = stimulatory; Dynorphin = inhibitory → together generate the GnRH pulse generator
- GnRH neurons lack estrogen receptors → kisspeptin neurons are the bridge for sex steroid feedback
- Loss of KISS1/KISS1R → hypogonadotropic hypogonadism (no puberty)
- Gain of KISS1/KISS1R → central precocious puberty
- MKRN3 is a puberty repressor — its loss = familial precocious puberty (paternal inheritance)
- Leptin is a permissive metabolic signal for puberty — links nutrition to HPG axis via kisspeptin
- Gonadostat theory: childhood HPG suppressed by high sensitivity to negative feedback → reset at puberty
- Gonadostat alone is insufficient: gonadectomy in children → only minimal ↑ in gonadotropins
- Pulsatile GnRH is essential — continuous GnRH paradoxically suppresses (used therapeutically in precocious puberty — GnRH analogues)
- Growth spurt: girls peak ~7 cm/year; boys gain ~28 cm total during puberty
- First sign of puberty in girls = thelarche (breast development)
- First sign of puberty in boys = testicular enlargement (>4 mL or >2.5 cm long)
- Mini-puberty of infancy = neonatal LH/FSH surge; important for gonadal development
- Puberty associated with mild physiological insulin resistance
10. INTEGRATION DIAGRAM (Conceptual Summary)
GENETIC PROGRAMMING (KISS1, MKRN3, LIN28B)
↓
ENVIRONMENTAL / METABOLIC SIGNALS
(Leptin, Nutrition, Body fat, Melatonin, Stress, Endocrine disruptors)
↓
↑ KISSPEPTIN (from KNDy neurons, ARC nucleus)
↓
→ Stimulates GnRH neurons (pulsatile)
↓ [NKB stimulates; Dynorphin terminates each pulse]
PULSATILE GnRH (starts during REM sleep)
↓
ANTERIOR PITUITARY → ↑ LH + FSH
↓
GONADS → ↑ Sex steroids (Estrogen / Testosterone)
↓
SECONDARY SEXUAL CHARACTERISTICS
+ GROWTH SPURT + MENARCHE/SPERMARCHE
📚 Sources Used
- Ganong's Review of Medical Physiology, 26th Edition — Control of Onset of Puberty, Ch. 22
- Medical Physiology (Boron & Boulpaep) — Gonadarche, Adrenarche, Kisspeptin system, Ch. 53
- Harrison's Principles of Internal Medicine, 22nd Edition (2025) — Normal Pubertal Development, Genetic Factors, Ch. 404
- Berek & Novak's Gynecology — Puberty chapter (Taubel & Rebar)
- KNDy neuron diagrams from Harrison's 21st Edition
➡ Ready for Part 2? We can now cover:
- Tanner Stages (Marshall & Tanner staging for breast, pubic hair, genitalia — with age ranges and clinical significance)
- Pubertal growth spurt in detail
- Hormonal changes across each Tanner stage
Just say "continue" and we'll dive into Tanner staging and pubertal changes in detail!