Thalacimia
Reading File
Finding Sources
Searching PubMed
thalassemia
Reading File
Reading File
Reading File
Searching Images
thalassemia blood smear peripheral smear hemolytic anemia
Reading File
Loading Image
Loading Image
Excellent. I now have all the content needed for a comprehensive answer on thalassemia.
Thalassemia
(Note: "Thalacimia" is a misspelling of thalassemia — from the Greek thalassa, meaning "sea," reflecting its prevalence around the Mediterranean.)
Definition
Thalassemia is a group of genetically heterogeneous inherited disorders caused by mutations that decrease synthesis of either α-globin or β-globin chains, resulting in anemia via two mechanisms:
- Hemoglobin deficiency → hypochromic, microcytic red cells with reduced oxygen-carrying capacity
- Imbalanced globin chain synthesis → excess unpaired chains precipitate, damage red cell membranes, causing hemolysis and ineffective erythropoiesis
— Robbins, Cotran & Kumar Pathologic Basis of Disease
Genetics
| Chain | Gene locus | Copy number |
|---|---|---|
| α-globin | Chromosome 16 | 4 genes (2 per chromosome) |
| β-globin | Chromosome 11 | 2 genes (1 per chromosome) |
Thalassemias are autosomal codominant conditions. Their worldwide prevalence is explained partly by protection against falciparum malaria in heterozygous carriers — the same mechanism proposed for sickle cell disease.
β-Thalassemia
Molecular Pathogenesis
β-thalassemia is caused by point mutations (>100 known) falling into two functional categories:
- β⁰ mutations — no β-globin synthesis at all
- β⁺ mutations — reduced but detectable β-globin synthesis
Three major mutational classes:
| Mutation Type | Effect | Result |
|---|---|---|
| Splicing mutations | Most common β⁺ cause; ectopic splice sites → some normal mRNA | β⁺ |
| Promoter mutations | Reduce transcription 75–80% | β⁺ |
| Chain terminator mutations | Nonsense or frameshift → no functional β-globin | β⁰ (most common cause) |
Unpaired α-chains precipitate within erythroid precursors → membrane damage → apoptosis of 70–85% of red cell precursors (ineffective erythropoiesis) → massive erythroid hyperplasia, extramedullary hematopoiesis, and skeletal deformities. Released RBCs with inclusions undergo extravascular hemolysis in the spleen.
Clinical Syndromes
| Syndrome | Genotype | Features |
|---|---|---|
| β-Thalassemia major (Cooley anemia) | β⁰/β⁰, β⁺/β⁰, β⁺/β⁺ (homozygous) | Severe transfusion-dependent anemia; presents in first year of life |
| β-Thalassemia intermedia | Variable | Moderately severe; regular transfusions not required |
| β-Thalassemia minor (trait) | β⁰/β or β⁺/β (heterozygous) | Asymptomatic; mild or absent anemia; microcytosis on smear |
β-Thalassemia Major: Clinical Features
Children are well at birth (fetal hemoglobin HbF is protective), then develop severe anemia by 6–9 months as HbF → HbA switch occurs.
Key manifestations:
- Severe microcytic, hypochromic anemia — Hb may be as low as 3–4 g/dL
- Hepatosplenomegaly — from extramedullary hematopoiesis and hemolysis
- Skeletal abnormalities — marrow expansion erodes bone cortex:
- "Hair-on-end" or "crew-cut" appearance on skull X-ray
- Chipmunk facies, frontal bossing
- Growth retardation and cachexia — erythroid progenitors steal nutrients
- Secondary hemochromatosis — from increased gut iron absorption (via erythroferrone-mediated hepcidin suppression) + repeated transfusions → cardiac failure, hepatic cirrhosis, endocrinopathies
- Jaundice — hemolysis-driven
Lab findings:
- Severe anemia (microcytic, hypochromic)
- Target cells, poikilocytosis, nucleated RBCs on peripheral smear
- Elevated HbF, elevated HbA2, absent or reduced HbA
- Elevated serum bilirubin, LDH, reticulocytes
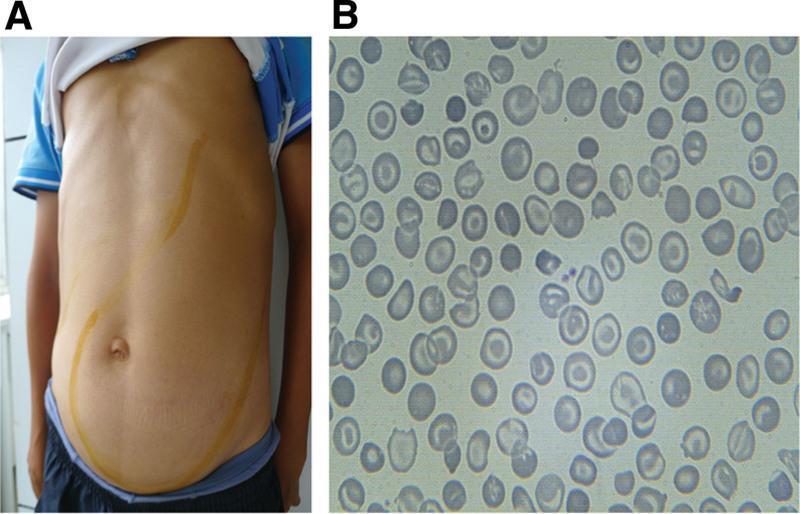
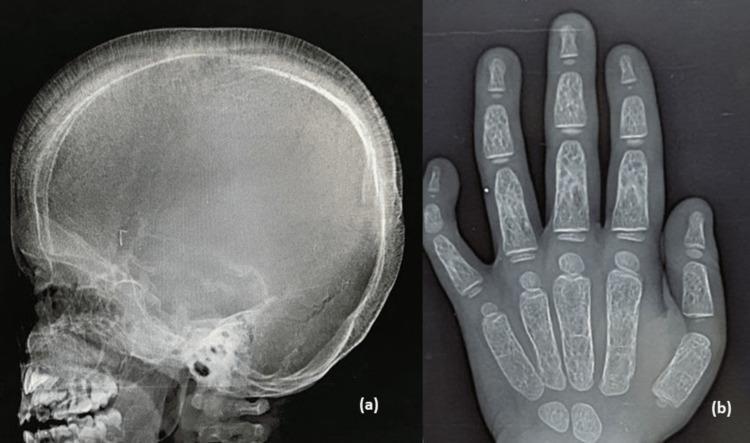
α-Thalassemia
Caused mainly by gene deletions (unlike β-thalassemia). Severity is proportional to the number of deleted α-globin genes.
| Syndrome | Deleted genes | Genotype | Features |
|---|---|---|---|
| Silent carrier | 1 of 4 | −/α, α/α | Asymptomatic; slight microcytosis |
| α-Thalassemia trait | 2 of 4 | −/−, α/α (Asian) or −/α, −/α (African) | Resembles β-thal minor; microcytosis, minimal anemia; HbA2 normal/low |
| HbH disease | 3 of 4 | −/−, −/α | Moderately severe anemia; β-globin tetramers (HbH) form — high O₂ affinity, poor O₂ delivery; HbH precipitates → splenomegaly |
| α-Thalassemia major (Hydrops fetalis) | 4 of 4 | −/−, −/− | γ-chain tetramers (Hb Bart's) — lethal without intrauterine transfusion; massive fetal edema, hepatosplenomegaly |
Ethnic note: Two-gene deletion on the same chromosome (−/−, α/α) is common in Asian populations and creates risk of HbH disease or hydrops fetalis in offspring. Two-gene deletion on different chromosomes (−/α, −/α) is common in African populations and rarely produces severely affected children.
Diagnosis
| Test | Findings |
|---|---|
| CBC | Microcytic (low MCV), hypochromic (low MCH), often low Hb |
| Peripheral smear | Target cells, poikilocytosis, nucleated RBCs (severe cases) |
| Hemoglobin electrophoresis | ↑HbF, ↑HbA2, absent/low HbA (β-thal major); HbH, Hb Bart's (α-thal) |
| Genetic testing | Confirms specific mutations/deletions |
| Serum iron studies | Elevated (iron overload), distinguish from iron deficiency |
Management
| Approach | Indication |
|---|---|
| Regular blood transfusions (every 3–4 weeks) | β-thalassemia major; target Hb 9–10 g/dL |
| Iron chelation (deferoxamine, deferasirox, deferiprone) | Prevent secondary hemochromatosis |
| Splenectomy | Hypersplenism causing increased transfusion needs |
| Hematopoietic stem cell transplantation (HSCT) | Curative in eligible patients; best outcomes in young patients without organ damage |
| Gene therapy / CRISPR-Cas9 | Emerging; aims to reactivate γ-globin (HbF) or correct the β-globin gene |
| Luspatercept | Approved for β-thalassemia; reduces ineffective erythropoiesis (TGF-β ligand trap) |
| Folic acid supplementation | All patients with hemolytic anemia |
| Intrauterine transfusion | α-thalassemia major (hydrops fetalis) |
Recent Evidence
A 2026 systematic review (PMID 39794549) on CRISPR/Cas9 for thalassemia and sickle cell disease confirms clinical trials are yielding promising results, with gene editing of BCL11A (to reactivate HbF) showing durable responses. A 2024 meta-analysis (PMID 39459358) highlighted that adrenal insufficiency is a significant endocrine complication in β-thalassemia patients, warranting routine screening.
Epidemiology & Malarial Protection
Thalassemia is most prevalent in:
- Mediterranean basin (Italy, Greece, Cyprus)
- Middle East and Indian subcontinent
- Sub-Saharan Africa
- Southeast Asia
Heterozygous carriers have a selective advantage against Plasmodium falciparum malaria, explaining the high allele frequencies in endemic regions.
Sources: Robbins, Cotran & Kumar Pathologic Basis of Disease; Robbins & Kumar Basic Pathology (9e)
Thanks 😊👍🏻
You're welcome! 😊 Feel free to ask anytime you have more questions — whether it's more on thalassemia or any other topic. Good luck with your studies! 📚
This is a shared conversation. Sign in to Orris to start your own chat.