TETANUS(clostridium tetani) under the following headings; a) Pathogenesis(MOA of Tetanus Toxin b) Clinical Manifestation & Complications c) Lab diagnosis**** (-morphology of C.Tetani d) Add a note on its prophylaxis (tetanus toxoid, dpt vaccine
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Searching Images
Clostridium tetani drumstick spore morphology gram stain
Reading File
Reading File
Loading Image
Loading Image
Loading Image
I now have all the content and images needed to deliver a comprehensive, well-structured answer. Here is the complete write-up:
TETANUS — Clostridium tetani
a) Pathogenesis — Mechanism of Action of Tetanus Toxin
The Organism
C. tetani is a non-invasive organism. It remains strictly localised at the wound site and causes disease almost entirely through toxaemia. Germination of spores and proliferation of vegetative cells is promoted by:
- Necrotic tissue (devitalized wounds, burns, umbilical stumps)
- Calcium salts
- Coexisting pyogenic (aerobic) infections — these deplete local oxygen, creating the requisite low oxidation-reduction potential
The Two Toxins
| Toxin | Nature | Role |
|---|---|---|
| Tetanolysin | Oxygen-labile hemolysin (related to streptolysin O) | Promotes local anaerobic conditions; clinical significance uncertain |
| Tetanospasmin | Plasmid-encoded, heat-labile neurotoxin (150 kDa) | Responsible for all clinical manifestations |
Step-by-Step MOA of Tetanospasmin
Step 1 — Production & Release
Tetanospasmin is synthesised during the stationary phase of bacterial growth and released upon cell lysis. It is produced as a single 150 kDa polypeptide, then cleaved by an endogenous protease into:
- Light chain (A chain, 50 kDa) — the toxic enzymatic unit
- Heavy chain (B chain, 100 kDa) — responsible for binding and transport The two chains remain joined by a disulfide bond.
Step 2 — Binding at the Neuromuscular Junction
The carboxyl-terminal domain of the heavy chain binds to specific polysialoganglioside receptors (sialic acid-containing) and adjacent glycoproteins on the surface of presynaptic membranes of lower motor neurons.
Step 3 — Retrograde Axonal Transport
The intact toxin is internalised into endosomal vesicles and transported retrogradely up the axon — at ~75–250 mm/day — to the motor neuron soma in the anterior horn of the spinal cord and brainstem. Tetanospasmin does not cross the blood-brain barrier; retrograde transport is its route to the CNS.
Step 4 — Translocation into the Cytosol
Once inside the neuron, the endosome becomes acidified, causing a conformational change in the N-terminal domain of the heavy chain. This triggers insertion into the endosomal membrane and passage of the light chain into the cytosol.
Step 5 — Cleavage of Synaptobrevin (VAMP2)
The light chain is a zinc endopeptidase (metalloproteinase) that cleaves synaptobrevin (also called VAMP2), a core SNARE protein required for docking and fusion of neurotransmitter vesicles with the presynaptic membrane.
Step 6 — Block of Inhibitory Neurotransmission
Synaptobrevin cleavage prevents release of the inhibitory neurotransmitters glycine and GABA from inhibitory interneurons (Renshaw cells and GABAergic interneurons) in the spinal cord.
Step 7 — Spastic Paralysis
Without inhibitory tone, motor neurons fire uncontrollably and tonically. Both agonist and antagonist muscles contract simultaneously → spastic paralysis, hyperreflexia, and generalised muscle spasms. This is the opposite of botulinum toxin, which blocks excitatory neurotransmitter (acetylcholine) release → flaccid paralysis.
Key point: Toxin binding to motor neurons is irreversible. Recovery depends on the growth of new axonal terminals.
— Medical Microbiology 9e; Jawetz, Melnick & Adelberg's Medical Microbiology, 28e; Sherris & Ryan's Medical Microbiology, 8e
b) Clinical Manifestations & Complications
Incubation Period
3–21 days (mean ~8 days). Shorter incubation correlates with greater severity — because the wound is closer to the CNS, allowing faster toxin transport.
Clinical Forms
1. Generalised Tetanus (80–90% of cases)
The most common form. Classic sequence:
- Trismus (lockjaw) — masseter spasm; the presenting complaint in ~75% of cases; often sends patients to a dentist initially
- Risus sardonicus — sardonic smile from sustained contraction of facial muscles
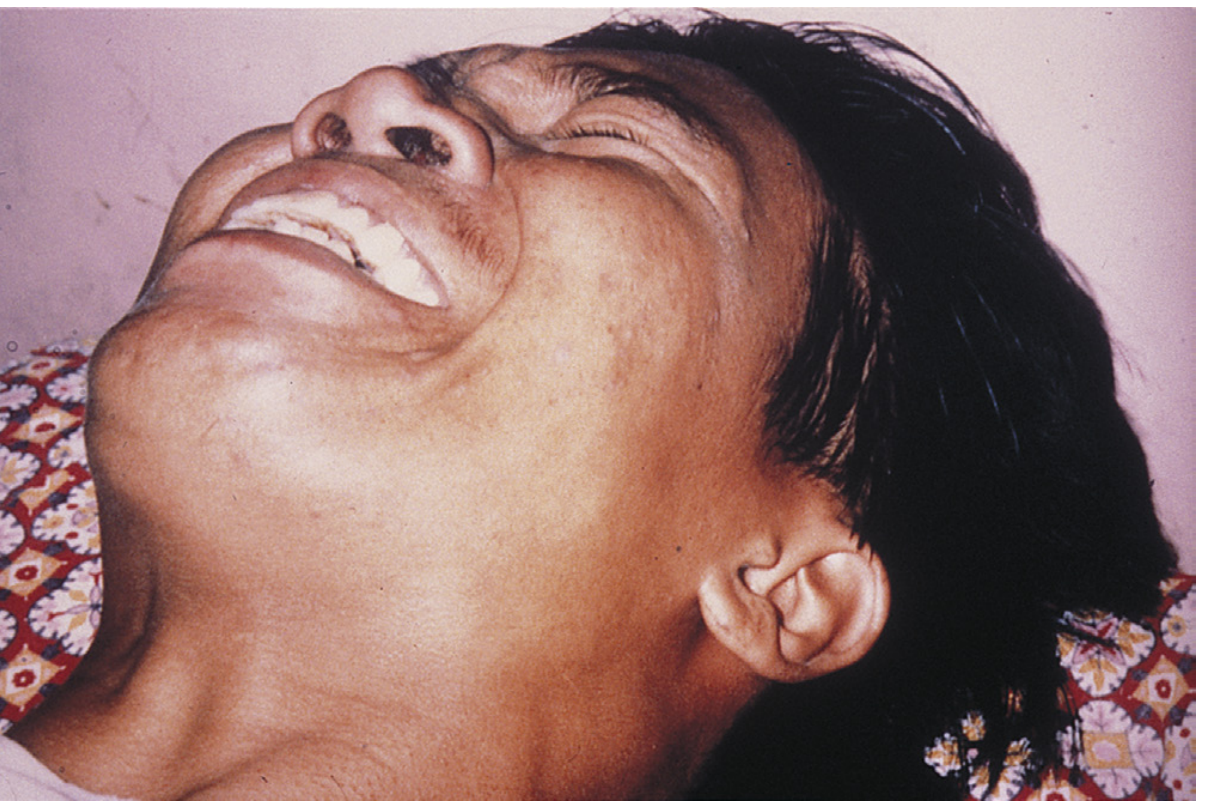
- Dysphagia, drooling, hydrophobia
- Opisthotonos — violent arching of the back from paraspinal muscle spasm
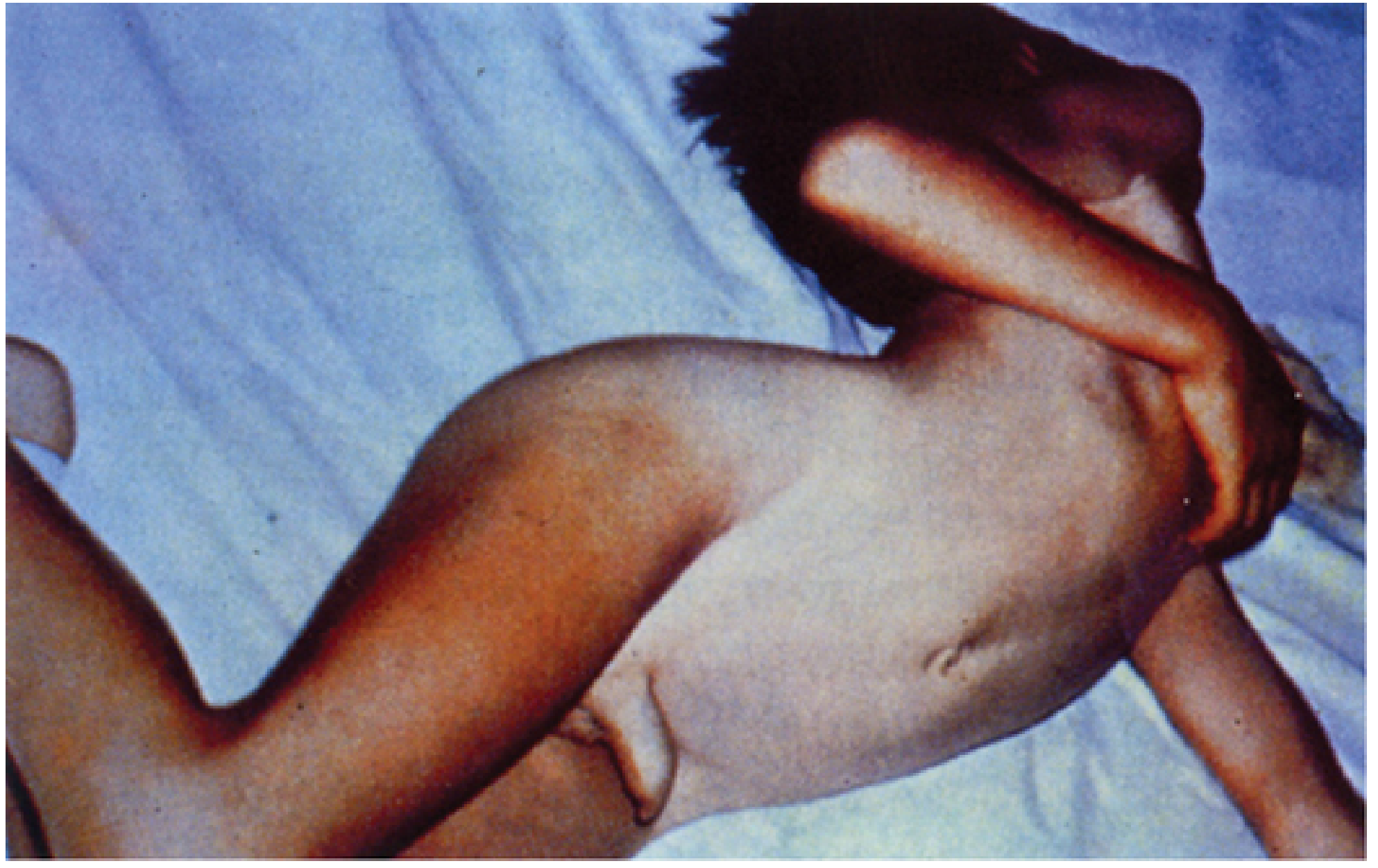
- Generalised tonic-clonic spasms triggered by any external stimulus (sound, light, touch)
- The patient remains fully conscious throughout
- Autonomic involvement (severe disease): labile blood pressure, cardiac arrhythmias, profuse diaphoresis, hyperthermia, tachycardia, urinary retention
2. Local Tetanus
Persistent muscle rigidity confined to the region of the wound. Mild form; may progress to generalised tetanus.
3. Cephalic Tetanus
Follows head injury or otitis media. Cranial motor nerve dysfunction (most commonly CN VII — facial palsy). Very poor prognosis.
4. Neonatal Tetanus (Tetanus Neonatorum)
Generalised tetanus in neonates — typically from infected umbilical stump after non-sterile cord care. Mortality >90% in infants. Almost exclusively a disease of developing countries.
Complications
| System | Complications |
|---|---|
| Respiratory | Laryngeal spasm → asphyxia, respiratory failure (leading cause of death) |
| Cardiovascular | Arrhythmias, labile BP, sympathetic storms |
| Musculoskeletal | Fractures and tendon avulsions from violent spasms; rhabdomyolysis |
| Metabolic | Dehydration, hyperthermia |
| Thromboembolic | Pulmonary embolism |
| Infectious | Secondary bacterial pneumonia, UTI (from urinary retention) |
| Renal | Acute kidney injury (from rhabdomyolysis) |
Untreated generalised tetanus carries a mortality rate of 15–60%; highest in neonates and the elderly.
— Goldman-Cecil Medicine International Edition; Tintinalli's Emergency Medicine, 8e; Medical Microbiology 9e
c) Laboratory Diagnosis — Including Morphology of C. tetani
Morphology of Clostridium tetani
Microscopy
| Feature | Description |
|---|---|
| Size | Large rod: 0.5–2 × 2–18 μm |
| Gram stain | Gram-positive (early growth); may appear gram-variable or gram-negative in older cultures or tissue specimens |
| Shape | Slender, straight rods with rounded ends |
| Spores | Round, terminal spores that bulge beyond the cell diameter → classic "drumstick" or "tennis racket" appearance |
| Motility | Motile via peritrichous flagella |
| Capsule | None |
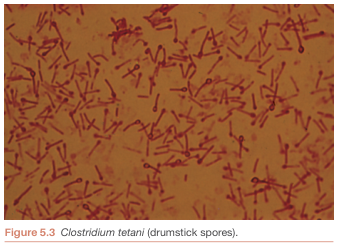
Culture Characteristics
- Strictly anaerobic — extremely oxygen-sensitive (obligate anaerobe)
- Growth on blood agar appears as a spreading film (swarming) rather than discrete colonies — due to peritrichous flagella
- Growth is slow and inhibited even by brief oxygen exposure
- Proteolytic but cannot ferment carbohydrates
- Spores are highly resistant: survive in soil for many years; resistant to many disinfectants and to boiling for several minutes
Antigenic Structure
- Multiple serotypes distinguished by specific flagellar (H) antigens
- All share a common O (somatic) antigen
- All produce the same antigenic type of neurotoxin (tetanospasmin)
Laboratory Diagnosis
The diagnosis of tetanus is primarily CLINICAL. Laboratory isolation of C. tetani confirms but is not required — and treatment must never be withheld awaiting culture results.
| Method | Details |
|---|---|
| Specimen | Wound swab/tissue from depth of wound (anaerobic transport) |
| Gram stain | Gram-positive rods with terminal drumstick spores |
| Anaerobic culture | Blood agar, Robertson's cooked meat medium; incubate anaerobically at 37°C |
| Culture positivity | Only ~30% — organism is killed rapidly by air, and disease can arise from very few organisms |
| Toxin detection | Gold standard: mouse neutralisation test — inject culture filtrate ± specific antitoxin into mice; toxin-injected mice develop spasms; antitoxin-protected mice are unaffected. Performed only in reference public health laboratories. |
| Serology | Neither toxin nor anti-toxin antibody is detectable in patient serum — toxin is rapidly sequestered in neurons |
| CSF | Normal |
| Differential diagnosis | Strychnine poisoning (most important), dystonic drug reactions, hypocalcaemic tetany, meningitis, rabies, alcohol withdrawal |
Key principle: Anaerobic culture and toxin neutralisation test in mice is the definitive microbiological confirmation; however, isolation is unsuccessful in the majority of cases and has no bearing on clinical management.
— Jawetz, Melnick & Adelberg's Medical Microbiology, 28e; Medical Microbiology 9e; Sherris & Ryan's Medical Microbiology, 8e; Goldman-Cecil Medicine
d) Prophylaxis — Tetanus Toxoid & DPT Vaccine
Principle
Tetanus is entirely preventable. Natural infection does not confer immunity (toxin dose is too small to stimulate an adequate immune response). Active immunisation with tetanus toxoid is the cornerstone of prevention.
Tetanus Toxoid
Preparation:
- Tetanospasmin is treated with formalin (formaldehyde) → destroys toxicity while retaining full antigenicity → produces toxoid
- Adsorbed onto aluminium salts (aluminium hydroxide or phosphate) to enhance immunogenicity and prolong antigen release (depot effect)
- The formaldehyde-treated toxoid stimulates production of neutralising antitoxin (anti-tetanospasmin IgG)
Primary Immunisation Schedule (Adults):
- Three doses of tetanus toxoid (Td)
- First two doses: 4–8 weeks apart
- Third dose: ~6–12 months after the second
- Booster: every 10 years thereafter to maintain protective antitoxin levels (≥0.01 IU/mL serum)
DPT Vaccine (Diphtheria–Pertussis–Tetanus)
The combined DPT/DTwP vaccine (or its acellular variant DTaP) is the standard childhood immunisation that includes tetanus toxoid alongside:
- D — Diphtheria toxoid
- P — Pertussis (whole-cell DTwP or acellular DTaP)
- T — Tetanus toxoid
Childhood Schedule (WHO/EPI standard):
| Dose | Age |
|---|---|
| DPT-1 | 6 weeks |
| DPT-2 | 10 weeks |
| DPT-3 | 14 weeks |
| Booster (DPT-4) | 15–18 months |
| Pre-school booster (DT/Td) | 4–6 years |
| Adolescent/adult Tdap | ~11–12 years, then Td every 10 years |
For adults and adolescents: the Td formulation (reduced diphtheria toxoid) is used instead of the full paediatric DPT dose.
Post-Exposure Prophylaxis (Wound Management)
| Immunisation history | Clean minor wound | Dirty/tetanus-prone wound |
|---|---|---|
| ≥3 doses, last booster <5 years ago | No action needed | No action needed |
| ≥3 doses, last booster 5–10 years ago | No action needed | Td booster |
| ≥3 doses, last booster >10 years ago | Td booster | Td booster |
| Unknown or <3 doses | Td + TIG | Td + TIG |
TIG = Tetanus Immune Globulin (Human)
- Dose: 250–500 IU IM (prophylaxis); 3,000–10,000 IU (treatment)
- Provides passive immunity for 2–4 weeks
- Neutralises only unbound toxin — toxin already fixed to neurons is unaffected
- Should be administered at a separate site from the toxoid injection
Wound Care (Non-Immunological Prophylaxis)
- Surgical debridement is critical — removes necrotic tissue required for C. tetani proliferation
- Antibiotics: Penicillin G or metronidazole (preferred — unlike penicillin, metronidazole does not inhibit GABA, thus avoids worsening CNS excitability)
"Tetanus is a totally preventable disease. Universal active immunisation with tetanus toxoid should be mandatory." — Jawetz, Melnick & Adelberg's Medical Microbiology, 28e
— Jawetz, Melnick & Adelberg's Medical Microbiology, 28e; Medical Microbiology 9e; Sherris & Ryan's Medical Microbiology, 8e; Goldman-Cecil Medicine International Edition; Tintinalli's Emergency Medicine, 8e
Summary Table
| Feature | Detail |
|---|---|
| Organism | Gram-positive, anaerobic, spore-forming rod |
| Key morphology | Drumstick/tennis-racket terminal spore |
| Toxin | Tetanospasmin (zinc metalloproteinase) |
| MOA | Cleaves synaptobrevin → blocks glycine/GABA release → spastic paralysis |
| Cardinal sign | Trismus + risus sardonicus + opisthotonos |
| Lab dx | Clinical; culture ~30% positive; mouse neutralisation test (reference lab) |
| Prevention | Tetanus toxoid; DPT in childhood; Td booster every 10 years |
| Post-exposure | Td ± TIG based on immunisation history + wound debridement |
This is a shared conversation. Sign in to Orris to start your own chat.