Excellent — rich material from all sources. Now let me compile the comprehensive educational response.
SENSORY ATAXIA & CANVAS SYNDROME
A Comprehensive Clinical Reference
PART 1: SENSORY ATAXIA
Definition
Sensory ataxia is a form of ataxia (disordered coordination/balance) caused by impairment of the proprioceptive afferent pathways rather than a cerebellar or motor deficit. The patient loses the sense of limb and body position in space, resulting in profound instability — particularly when vision is removed.
It differs fundamentally from:
- Cerebellar ataxia — due to cerebellar/cerebellar pathway disease
- Vestibular ataxia — due to peripheral or central vestibular dysfunction
- Motor/spastic ataxia — due to upper motor neuron lesions
Neuroanatomical Basis
The proprioceptive pathway relies on an intact circuit:
Peripheral large-fiber nerve (Ia afferents)
↓
Dorsal root ganglion (DRG)
↓
Posterior columns (dorsal funiculi) of spinal cord
[Gracile tract — lower limb; Cuneate tract — upper limb]
↓
Nucleus gracilis / cuneatus (medulla)
↓
Medial lemniscus
↓
Thalamus (VPLc nucleus)
↓
Somatosensory cortex (parietal lobe)
Any disruption along this chain — from peripheral nerve to cortex — produces sensory ataxia. The most clinically significant sites are:
- Peripheral large-fiber neuropathy (e.g., diabetes, CANVAS, paraneoplastic)
- Dorsal root ganglion (DRG) neuronopathy/ganglionopathy (e.g., CANVAS, Sjögren's, paraneoplastic anti-Hu)
- Posterior column disease (e.g., Vitamin B12 deficiency/subacute combined degeneration, tabes dorsalis, MS)
- Medial lemniscus lesions (brainstem strokes)
"Dorsal horn and column injury alone may result from tabes dorsalis... Friedreich ataxia represents a genetic etiology, and vitamin B12 deficiency may result in subacute combined degeneration with spastic paretic gait and sensory ataxia." — Bradley & Daroff's Neurology in Clinical Practice
Clinical Features of Sensory Ataxia
Gait
- High-stepping, stamping, or stomping gait — patient lifts feet high and brings them down forcefully to get ground-contact feedback
- Wide-based stance — compensatory base widening for stability
- Worsens dramatically in darkness or with eyes closed — vision is the primary compensatory mechanism
- Patient looks at the ground when walking (visual substitution)
- Tandem walking severely impaired
Romberg Sign (Positive / Sharpened)
The cornerstone bedside sign:
- Eyes open: patient stands adequately (visual compensation active)
- Eyes closed: patient sways markedly and may fall
- Positive = sensory ataxia — the test specifically unmasks proprioceptive failure by removing vision
- Cerebellar ataxia produces instability with eyes OPEN as well; sensory ataxia is markedly worse with eyes CLOSED
Fukuda Stepping Test
- 50 marching steps in place, eyes closed, arms forward
- Rotation > 30° or marked drift toward one side = positive
- In sensory ataxia, there is deviation because proprioceptive feedback cannot correct body position during stepping
Limb/Digit Signs
- Loss of joint position sense (JPS) — patient cannot detect finger/toe movement
- Loss of vibration sense — tuning fork (128 Hz) not felt at toes/ankles
- Pseudo-athetosis — involuntary writhing movements of fingers/toes when eyes closed (loss of position sense allows drift)
- Positive Romberg with pseudo-athetosis of hands is highly specific for posterior column / DRG pathology
- Finger-nose test may be normal (proprioception in upper limbs often relatively preserved early)
- Heel-shin test impaired — foot slides off the shin when eyes closed
Other Features
- Deep tendon reflexes often reduced or absent (large Ia afferent fibers carry the reflex arc)
- No weakness (purely sensory syndrome)
- Sensory examination: loss of vibration, JPS, light touch, 2-point discrimination; pain and temperature usually preserved (small-fiber involvement occurs in some subtypes)
Causes of Sensory Ataxia (Classification)
| Category | Examples |
|---|
| Genetic | CANVAS (RFC1), Friedreich ataxia (FXN), AVED (vitamin E deficiency), abetalipoproteinemia |
| Metabolic/Nutritional | Vitamin B12 deficiency (subacute combined degeneration), copper deficiency, vitamin E deficiency |
| Infectious | Tabes dorsalis (tertiary syphilis — the classic cause) |
| Autoimmune/Inflammatory | Sjögren's syndrome (sensory neuronopathy), paraneoplastic (anti-Hu, anti-CV2), Miller-Fisher, CIDP |
| Toxic | Pyridoxine (B6) toxicity, cisplatin, taxanes |
| Vascular | Posterior spinal artery infarction (posterior cord syndrome) |
| Demyelinating | Multiple sclerosis (posterior column plaques) |
| Compressive | Subacute myelopathy from disc/tumour affecting posterior columns |
Distinguishing Sensory vs Cerebellar Ataxia (Key Table)
| Feature | Sensory Ataxia | Cerebellar Ataxia |
|---|
| Worsens with eyes closed | Yes (markedly) | Mild worsening |
| Romberg sign | Positive | May be positive (different mechanism) |
| Gait | Stamping, high-stepping | Wide-based, lurching, reeling |
| Dysmetria (finger-nose) | Usually normal | Abnormal |
| Nystagmus | Absent | Often present |
| Deep tendon reflexes | Reduced/absent | Normal or pendular |
| JPS / vibration sense | Lost | Normal |
| Speech | Normal | Dysarthric |
| Pseudo-athetosis | Present | Absent |
| MRI | Posterior column T2 signal / DRG atrophy | Cerebellar atrophy |
PART 2: CANVAS SYNDROME
Definition and Historical Overview
CANVAS = Cerebellar Ataxia, Neuropathy, Vestibular AreSia Syndrome
- First described clinically by Szmulewicz et al. in 2011 as a triad of bilateral vestibular areflexia, cerebellar ataxia, and sensory neuropathy
- Genetic basis identified in 2019 by Cortese et al.: biallelic AAGGG pentanucleotide repeat expansions in intron 2 of the RFC1 gene
- Now recognised as the most common cause of autosomal recessive ataxia in adults
- Mean age at onset: 54 ± 9 years (range: onset always after age 35)
Genetics
| Feature | Detail |
|---|
| Gene | RFC1 (Replication Factor C Subunit 1) |
| Chromosome | 4p14 |
| Mutation | Biallelic (AAGGG)n intronic repeat expansion in intron 2 |
| Normal allele | (AAAAG)n repeat |
| Pathogenic allele | (AAGGG)n expansion — biallelic (homozygous or compound heterozygous) |
| Inheritance | Autosomal recessive |
| Asian/Pacific variant | ACAGG repeats may be found instead of AAGGG in Asian populations |
| Detection | Standard exome/genome sequencing CANNOT detect this — requires repeat-primed PCR or long-read sequencing |
The RFC1 protein is a DNA polymerase accessory protein required for coordinated synthesis of both DNA strands during replication and after DNA damage. The (AAGGG)n expansion forms stable secondary DNA structures that disrupt gene function — the precise downstream mechanism causing neurodegeneration is still under active research.
Pathology — What Degenerates?
CANVAS targets three systems preferentially:
1. DORSAL ROOT GANGLIA (DRG) — "Ganglionopathy"
→ Large sensory neurons degenerate
→ Loss of proprioception → sensory ataxia
→ Non-length-dependent pattern (feet AND hands affected early)
→ Sural nerve biopsy: loss of large myelinated axons
2. VESTIBULAR END-ORGAN / NERVE
→ Bilateral degeneration of hair cells and/or vestibular ganglia
→ Absent or severely reduced bilateral VOR
→ No nystagmus (bilateral loss = no asymmetry)
→ Oscillopsia and gait failure in darkness
3. CEREBELLUM — especially posterior lobe/vermis
→ Progressive Purkinje cell degeneration
→ MRI: cerebellar atrophy (posterior lobe, vermis lobules VI–VIIb)
→ Contributes additional kinetic ataxia beyond the sensory component
Autopsy studies confirm degeneration of the dorsal root ganglia and posterior columns.
Clinical Features
The Core Triad
| Component | Clinical Manifestation | Frequency |
|---|
| Sensory neuropathy | Loss of JPS, vibration, proprioception; sensory ataxia; positive Romberg | 100% |
| Bilateral vestibular impairment | Oscillopsia, imbalance worsening in dark/on unstable surfaces, no spontaneous nystagmus | 69–93% of those tested |
| Cerebellar syndrome | Gait ataxia, limb dysmetria, kinetic tremor | 63% |
Additional Features (Expanding Spectrum)
| Feature | Frequency |
|---|
| Chronic dry cough | 64% — often the earliest symptom, preceding ataxia by years; due to hypersensitive cough reflex (vagal neuropathy) |
| Dysautonomia | 32–50% — orthostatic hypotension, bladder dysfunction, constipation |
| Parkinsonism | Rare — reported in some RFC1-positive patients |
| ALS-like features | Rare — upper and lower motor neuron involvement (spasticity, fasciculations) |
| Cognitive/memory impairment | Generally spared in typical CANVAS |
"CANVAS can also present with dysautonomia and features of parkinsonism. NCS reveal low-amplitude or absent sensory responses that are in a non-length-dependent pattern." — Harrison's Principles of Internal Medicine 22E (2025)
RFC1 Spectrum Disorder (Not Just Classic CANVAS)
In a cohort of 100 RFC1-positive individuals:
- 63% had full CANVAS triad
- 16% had complex sensory ataxia + cerebellar OR vestibular (incomplete triad)
- 15% had sensory neuropathy only — no clinically detectable cerebellar or vestibular features
This means RFC1 should be tested even in patients presenting with just sensory ataxia or isolated chronic cough with balance problems.
Neuroimaging Findings (MRI)
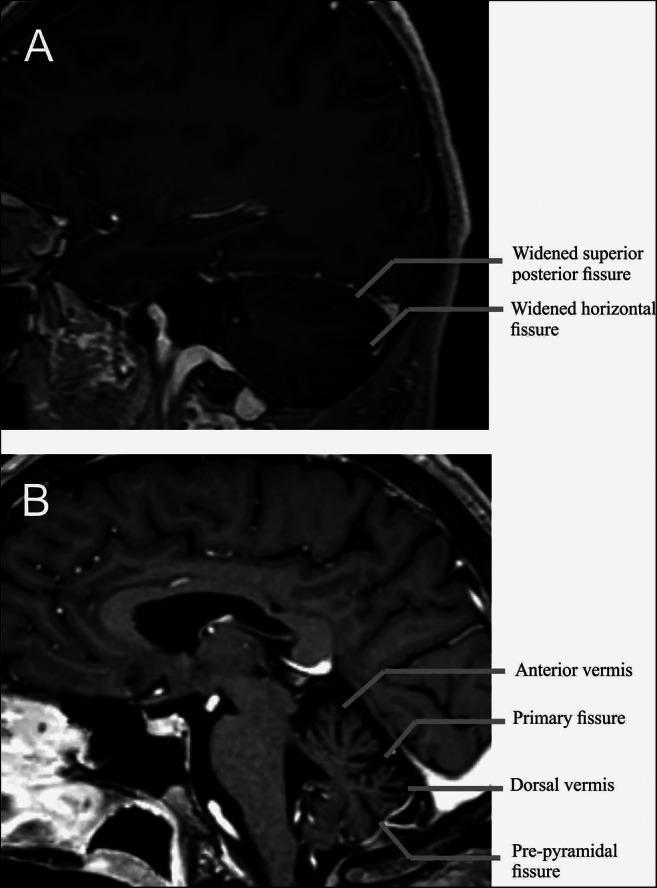
3D T1 MRI in CANVAS: parasagittal (A) showing hemispheric atrophy; midsagittal (B) showing dorsal vermis atrophy at lobules VI–VIIb, with widening of the superior posterior fissure and horizontal fissure.
- Cerebellar vermal atrophy — most prevalent (~90%), predominantly posterior vermis
- Cerebellar hemispheric atrophy
- Brainstem atrophy — pontine in more severe cases
- Spinal cord thinning — cervical and thoracic, reflecting posterior column degeneration
- Some cases show bilateral pallidal T2 abnormalities (less common)
Electrophysiology
From the largest characterisation study (Lainez et al., Clin Neurophysiol 2025, PMID 39721397, n=31):
| Test | Finding |
|---|
| Sensory NCS | Absent or severely low amplitude SNAPs — 100% of patients |
| Motor NCS | Normal — purely sensory disorder |
| NCS pattern | Non-length-dependent (upper and lower limb SNAPs equally affected — DRG ganglionopathy pattern, not distal neuropathy) |
| Tibial SEPs | Absent or severely attenuated — 100% |
| Median SEPs | Moderately affected |
| H-reflexes | Preserved in 85.7% — distinguishes from lower motor neuron disease |
| Needle EMG | May show active denervation/chronic reinnervation in some |
| Autonomic tests | Predominantly normal, but 74% showed impaired quantitative sensory test (thermoalgesic) |
The non-length-dependent sensory loss on NCS is the electrophysiological fingerprint — it points to DRG degeneration rather than a peripheral dying-back neuropathy.
Vestibular Testing in CANVAS
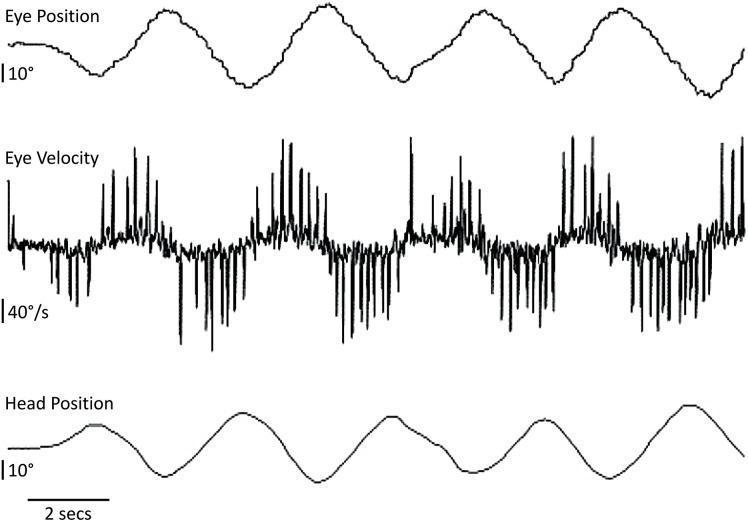
Visually-assisted VOR (VVOR) test in CANVAS: the eye velocity trace shows high-amplitude compensatory saccadic spikes (40°/s) representing a "broken-up" smooth pursuit and severely impaired VOR — characteristic of bilateral vestibular areflexia.
| Test | Expected Finding in CANVAS |
|---|
| vHIT | Bilaterally reduced VOR gain (< 0.6); bilateral corrective saccades |
| Caloric | Bilaterally reduced or absent responses; no canal paresis asymmetry |
| Rotational chair | Reduced gain all frequencies; reduced time constant; no directional preponderance (symmetric) |
| cVEMP | Absent or severely attenuated bilaterally (saccular failure) |
| oVEMP | Absent or severely attenuated bilaterally (utricular failure) |
| Spontaneous nystagmus | Absent — bilateral symmetric loss = no asymmetric drive |
| Positional tests | Normal — no BPPV |
The diagnostic trap: Because loss is bilateral and (often) symmetric, clinicians do not see lateralising signs — no spontaneous nystagmus, no canal paresis on one side. The patient appears "vestibularly normal" on a standard vestibular screen. Only bilateral quantitative testing (vHIT, caloric total, rotational chair) reveals the deficit.
Diagnostic Criteria / When to Suspect CANVAS
Per GeneReviews/NIH (2025), suspect RFC1 CANVAS/Spectrum Disorder when:
- Onset after age 35
- Any combination of:
- Complex imbalance/coordination problem of peripheral + vestibular + cerebellar origin
- Sensory neuropathy or neuronopathy (non-length-dependent)
- Bilateral vestibular areflexia (or bilateral hyporeflexia)
- Cerebellar dysfunction (progressive)
- Chronic dry cough (highly specific — pursue RFC1 testing in any ataxia patient with unexplained chronic cough)
- Dysautonomia
Investigations supporting the diagnosis:
- NCS showing absent/low SNAPs, non-length-dependent, normal motor
- Bilateral vestibular hypofunction on vHIT / caloric / rotational chair
- MRI brain: cerebellar (posterior lobe/vermis) atrophy
- Absent tibial SEPs
- RFC1 biallelic AAGGG repeat expansion confirmed on repeat-primed PCR
Differential Diagnosis
| Condition | Key Distinguishing Feature |
|---|
| Friedreich ataxia | Younger onset (< 25 yrs), cardiomyopathy, scoliosis, FXN GAA expansion |
| SCA3 (MJD) | Autosomal dominant, pyramidal/extrapyramidal signs, sensorimotor (not pure sensory) neuropathy |
| Sjögren's neuronopathy | Anti-Ro/La antibodies, dry eyes/mouth, keratoconjunctivitis sicca |
| Paraneoplastic ganglionopathy | Rapid onset, anti-Hu/CV2, associated malignancy |
| Vitamin B12 deficiency | Posterior column T2 signal on MRI spine, elevated MMA/homocysteine, treatable |
| Vitamin E deficiency (AVED) | Low serum vitamin E, Friedreich-like, treatable |
| POLG1 mutations (SANDO) | Ophthalmoplegia, epilepsy, myopathy |
| Copper deficiency myelopathy | Posterior column T2 signal, low serum copper, often post-gastric surgery |
Management
There is currently no disease-modifying or curative treatment for CANVAS. Management is entirely supportive:
Symptomatic
- Chronic cough: Gabapentin, pregabalin, speech therapy (laryngeal sensory retraining); avoid ACE inhibitors (worsen cough)
- Neuropathic pain (if thermoalgesic fibers involved): Duloxetine, pregabalin, amitriptyline
- Dysautonomia: Fludrocortisone, compression stockings (orthostatic hypotension); bladder anticholinergics if indicated
- Cerebellar tremor/ataxia: No proven pharmacotherapy; riluzole and acetyl-DL-leucine are under investigation
Rehabilitation
- Vestibular rehabilitation — gaze stabilisation exercises, bilateral VOR substitution training
- Balance physiotherapy — ataxia-specific programs (progressive foam/unstable surface training)
- Occupational therapy — fall prevention, home adaptation
- Walking aids — canes, rollators; particularly important in darkness
Safety
- Avoid aminoglycosides absolutely (will catastrophically worsen bilateral vestibular function)
- Driving assessment — oscillopsia from VOR failure may render driving unsafe
- Adequate lighting at home; remove trip hazards
Genetic Counselling
- Autosomal recessive — siblings have 25% risk
- Offspring are obligate carriers (not affected), but partner carrier status matters
- Family cascade testing if symptomatic relatives
Prognosis
- Slowly progressive over 10–15 years
- Most patients eventually require a walking aid
- Cognitive function generally preserved
- Life expectancy not markedly reduced in typical CANVAS
- Severe cases with ALS-like features or marked dysautonomia have worse prognosis
Summary Diagram
RFC1 gene — biallelic (AAGGG)n repeat expansion
↓
Degeneration of 3 systems:
DRG Ganglionopathy Vestibular Areflexia Cerebellar Atrophy
↓ ↓ ↓
Sensory neuropathy Bilateral loss of Posterior lobe
(non-length-dependent) VOR/otolith function vermis atrophy
Loss of proprioception, No nystagmus; oscillopsia Limb dysmetria
vibration, JPS Imbalance in dark Kinetic tremor
↓ ↓ ↓
Positive Romberg vHIT bilat gain ↓↓ MRI cerebellar atrophy
Fukuda deviation Caloric bilat absent Gait cerebellar component
Stamping gait cVEMP/oVEMP absent
+ Chronic cough (64%) + Dysautonomia (32%)
Sources: Harrison's Principles of Internal Medicine 22E (2025) | Bradley & Daroff's Neurology in Clinical Practice | Lainez et al.,
Clin Neurophysiol 2025 (
PMID 39721397) | Shukla et al.,
Mol Neurobiol 2025 (
PMID 38898197) |
GeneReviews RFC1 CANVAS/Spectrum Disorder (NIH, 2025)