Coronary artery diseases
Reading File
Finding Sources
Searching PubMed
"coronary artery disease"[MeSH Terms] AND "management"[MeSH Terms]
Searching the Web
coronary artery disease 2025 guidelines management overview
Reading File
Reading File
Finding Sources
Searching Images
coronary artery disease atherosclerosis plaque diagram
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
I now have rich content from multiple textbooks plus current guideline information. Let me compile a comprehensive response.
Coronary Artery Disease (CAD)
Definition & Epidemiology
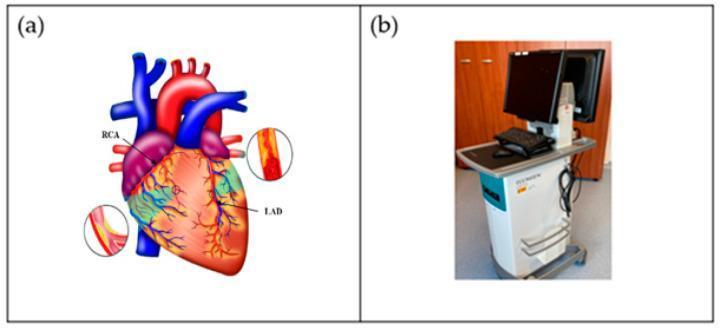
Coronary artery disease is the end-result of atherosclerosis affecting the coronary circulation — the leading cause of morbidity and mortality worldwide. Atherosclerotic cardiovascular disease (ASCVD) currently accounts for >17.3 million deaths per year globally, a figure projected to rise to >23.6 million by 2030 due to aging populations. Nearly one in three deaths in the United States is attributable to ASCVD. — Barash's Clinical Anesthesia, 9e, p. 3382
Pathophysiology
Atherosclerosis: Injury-Response Model
Atherosclerosis is fundamentally a generalized inflammatory disorder of the arterial tree. Its development occurs in two stages:
-
Endothelial Injury: Risk factors (hyperlipidemia, hypertension, smoking, diabetes) damage endothelial cells, causing expression of leukocyte adhesion molecules and release of chemokines (e.g., MCP-1). Increased permeability allows LDL particles and leukocytes to enter the subendothelial space.
-
Response to Injury: Monocyte-derived macrophages engulf oxidized LDL and transform into foam cells — the hallmark of the early "fatty streak" lesion. Cytokines (TNF-α, IL-1, IL-6, IL-8, IL-18) and growth factors recruit further inflammatory and smooth muscle cells. Over time, foam cells + extracellular lipid form a necrotic core surrounded by smooth muscle cells and a collagen-rich fibrous cap.
-
Plaque Progression → Luminal Narrowing: Progressive atheroma narrows the arterial lumen, creating an imbalance between myocardial oxygen supply and demand.
-
Vulnerable Plaque & Rupture: Inflammatory mediators and proteolytic enzymes thin the fibrous cap; rupture triggers platelet aggregation and acute thrombosis — the culprit event in most acute coronary syndromes. — Barash's Clinical Anesthesia, 9e, p. 3383–3384
Risk Factors
| Non-Modifiable | Modifiable |
|---|---|
| Age (♂ >45, ♀ >55) | Hyperlipidemia (↑LDL, ↓HDL) |
| Male sex | Hypertension |
| Family history | Diabetes mellitus |
| Genetic predisposition | Smoking |
| Obesity / Metabolic syndrome | |
| Sedentary lifestyle | |
| Chronic kidney disease |
The same risk factors that drive CAD also produce vascular disease in cerebral, aortic, and peripheral beds — hence ~40–60% of patients with peripheral arterial disease have concurrent CAD. — Barash's Clinical Anesthesia, 9e, p. 3385
Clinical Spectrum
CAD manifests along a continuum from silent disease to sudden death:
Silent CAD → Stable Angina → Unstable Angina → NSTEMI → STEMI → Sudden Cardiac Death
↑_____________Acute Coronary Syndromes (ACS)______________↑
1. Stable Angina Pectoris
- Mechanism: Fixed stenosis causing demand ischemia (supply-demand mismatch with exertion)
- Symptoms: Retrosternal chest tightness/pressure, radiation to left arm/shoulder/jaw; relieved by rest or nitroglycerin; triggered by exertion, cold, stress
- Prognosis: ~2% average annual mortality (only twice age-matched controls)
- ECG: ST depression during episodes; normal at rest
2. Unstable Angina (UA)
Three presentations:
- Angina at rest
- New-onset severe angina
- Crescendo angina (increasing frequency, severity, or duration with less provocation)
- Troponins are negative (distinguishes from NSTEMI)
3. NSTEMI
- Same presentation as UA but with elevated troponins (myocardial necrosis confirmed)
- No ST elevation on ECG (may show ST depression, T-wave changes)
- 30-day mortality 10–20% despite treatment
4. STEMI
- Complete occlusion of a coronary artery → transmural infarction
- ST elevation in ≥2 contiguous leads (or new LBBB)
- Medical emergency requiring immediate reperfusion (primary PCI preferred)
Prinzmetal (Variant) Angina
- Caused by coronary artery spasm (with or without fixed stenosis)
- Occurs at rest, often in early morning, may awaken patient from sleep
- ST elevation (not depression) during episodes
- More common in women
- Treatment: calcium channel blockers (first-line); nitrates also effective; beta-blockers contraindicated in pure vasospasm — Swanson's Family Medicine Review, p. 119
Diagnosis
| Test | Use |
|---|---|
| ECG | ST/T changes, Q waves, rate/rhythm |
| Cardiac Troponins (I/T) | Myocardial necrosis; serial testing in ACS |
| Stress ECG (Exercise test) | Initial evaluation of stable CAD |
| Stress Echo / Nuclear (Thallium) | Functional ischemia, viability |
| Coronary CT Angiography (CCTA) | Non-invasive anatomy; calcium scoring; plaque characterization |
| Coronary Angiography | Gold standard for anatomy; guides revascularization |
| Intravascular Ultrasound (IVUS) | Plaque characterization; guide PCI — Class I per 2025 ACS Guideline |
Initial labs: CBC, lipid panel, glucose, HbA1c, renal function, thyroid function, CXR
Treatment
Three Pillars
- Medical Therapy
- Percutaneous Coronary Intervention (PCI)
- Coronary Artery Bypass Grafting (CABG)
Medical Therapy
All patients with CAD / stable angina (without contraindications) should receive:
| Drug Class | Agent | Mechanism / Role |
|---|---|---|
| Antiplatelet | Aspirin 75–100 mg | COX-1 inhibition → ↓TXA2 → ↓platelet aggregation |
| Statin | Atorvastatin, Rosuvastatin | LDL ↓; plaque stabilization; reduce MI events |
| Beta-blocker | Metoprolol, Bisoprolol | ↓HR, ↓contractility → ↓O₂ demand; mortality benefit post-MI |
| ACE inhibitor / ARB | Ramipril, Losartan | Neurohormonal blockade; post-MI remodeling prevention |
| Nitrates | Sublingual NTG (acute); ISDN/ISMN (prophylaxis) | Venodilation → ↓preload; coronary vasodilation |
| Calcium channel blocker | Amlodipine (DHP); Diltiazem/Verapamil (non-DHP) | ↓afterload; anti-anginal; vasospasm (diltiazem preferred) |
For ACS (UA/NSTEMI):
- Aspirin + P2Y12 inhibitor (clopidogrel, ticagrelor, or prasugrel) = DAPT
- Anticoagulation: unfractionated heparin or LMWH (enoxaparin)
- Consider GPIIb/IIIa inhibitor for high-risk patients
- Early invasive strategy (cardiac catheterization) — Swanson's Family Medicine Review, p. 119–120
PCI (Percutaneous Coronary Intervention)
- Balloon angioplasty + drug-eluting stent (DES) placement
- Success rate >90% per stenosis; complication rate ~4%
- Does not reduce overall mortality vs. medical therapy in stable CAD
- Does improve symptoms and reduces need for medication
- Drug-eluting stents dramatically reduced restenosis (previously up to 40%)
- 2025 ACS Guideline update: Intracoronary imaging (IVUS/OCT) during complex PCI upgraded from Class IIa → Class I recommendation
CABG (Coronary Artery Bypass Grafting)
CABG is preferred over PCI / medical therapy for:
- Left main stem disease (≥50% stenosis)
- 3-vessel CAD
- 2-vessel disease with proximal LAD involvement
- Diabetic patients with multivessel disease
In these high-risk subsets, CABG provides a survival benefit over medical therapy. For stable, lower-risk patients, CABG improves symptoms when medical therapy fails.
Revascularization Decision Summary
Single-vessel CAD (non-LAD) → Medical therapy OR PCI
Proximal LAD / 2-vessel → PCI or CABG (anatomy-dependent)
3-vessel CAD / Left main → CABG preferred (especially with ↓EF or diabetes)
STEMI → Primary PCI within 90 minutes (door-to-balloon time)
Secondary Prevention
| Intervention | Target |
|---|---|
| Statin therapy (high-intensity) | LDL-C <70 mg/dL (or <55 mg/dL for very high risk) |
| Blood pressure control | <130/80 mmHg |
| Glycemic control (DM) | HbA1c <7% |
| Antiplatelet therapy | Lifelong aspirin ± P2Y12 post-ACS |
| Smoking cessation | Complete cessation |
| Cardiac rehabilitation | Exercise, education, psychosocial support |
| Diet & weight management | Mediterranean-style diet; BMI <25 |
2025 ACS Guideline — Key Updates
The 2025 ACC/AHA/ACEP/NAEMSP/SCAI ACS Guideline consolidates the 2013 STEMI and 2014 NSTEMI guidelines and introduces several practice-changing recommendations:
- Intracoronary imaging (IVUS/OCT) during PCI for complex lesions: upgraded to Class I
- Ticagrelor monotherapy (≥1 month post-PCI) as a bleeding-reduction strategy
- PPI use recommended for patients at GI bleeding risk on DAPT
- In patients requiring long-term anticoagulation after PCI: aspirin discontinuation at 1–4 weeks with continuation of P2Y12 inhibitor (preferably clopidogrel)
Sources:
- Barash's Clinical Anesthesia, 9e, pp. 3382–3390
- Swanson's Family Medicine Review, pp. 119–120
- Sabiston Textbook of Surgery
- 2025 ACC/AHA/ACEP/NAEMSP/SCAI ACS Guideline — Circulation & JACC
Nstemi vs stemi
Finding Sources
Searching Images
STEMI NSTEMI ECG ST elevation depression comparison
Searching the Web
NSTEMI vs STEMI difference management 2025
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
I now have comprehensive content. Let me compile a thorough, well-structured comparison.
NSTEMI vs STEMI — Complete Comparison
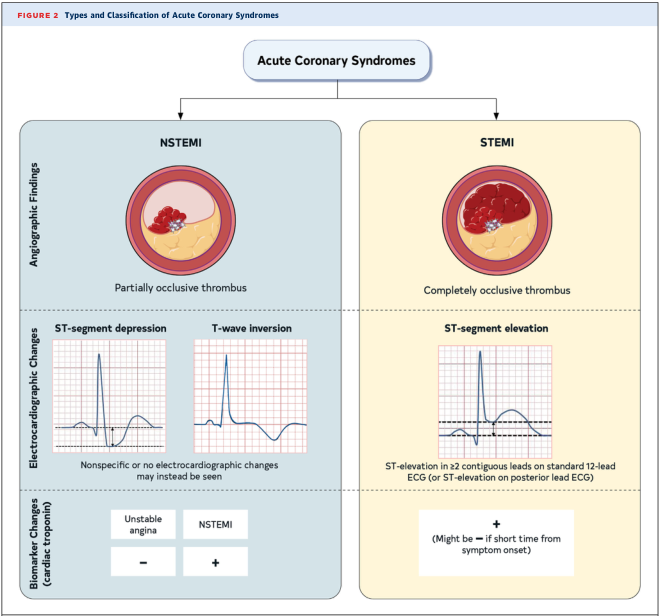
The Core Distinction
Both are myocardial infarctions (confirmed necrosis via troponin rise) within the Acute Coronary Syndrome (ACS) spectrum. The fundamental difference is the degree of coronary occlusion and its ECG signature:
| NSTEMI | STEMI | |
|---|---|---|
| Coronary occlusion | Partial (subtotal) thrombus | Complete (total) occlusion |
| Myocardium at risk | Subendocardium (inner layer) | Transmural (full thickness) |
| Troponin | Positive ↑ | Positive ↑ (may be negative very early) |
| ECG hallmark | ST depression, T-wave inversion, or nonspecific changes | ST elevation ≥1–2 mm in ≥2 contiguous leads |
Pathophysiology
Both arise from atherosclerotic plaque rupture or erosion → platelet aggregation → thrombus formation:
-
STEMI: Thrombus is completely occlusive → abrupt cessation of flow in an epicardial artery → transmural necrosis progressing in a "wave-front" from endocardium to epicardium. Necrosis begins in as little as 15–20 minutes; partial salvage achievable if reperfused within 3–6 hours.
-
NSTEMI: Thrombus is partially occlusive (or complete occlusion with good collateral supply) → subendocardial ischemia/necrosis. Less immediate threat to full-thickness myocardium but still causes cell death (hence positive troponin). — Goldman-Cecil Medicine, p. 654
ECG Differences in Detail
STEMI ECG Criteria
- ST elevation ≥2 mm (0.2 mV) in V2–V3 in men; ≥1.5 mm in women
- ≥1 mm in ≥2 other contiguous chest or limb leads
- Reciprocal ST depression in opposite leads (e.g., inferior STEMI shows elevation in II, III, aVF with depression in aVL/I)
- Hyperacute T waves may precede ST elevation in earliest phase
- Evolution: ST elevation → T-wave inversion → pathologic Q waves (irreversible necrosis)
- New LBBB = STEMI equivalent
Three ECG phases of STEMI evolution:
- Early Acute (minutes–hours): Hyperacute T waves → convex "tombstone" ST elevation
- Evolved Acute (hours–days): ST elevation regresses, T-wave inversion, Q waves form
- Chronic (weeks–months): Q waves persist, ST returns to baseline — Goldman-Cecil Medicine, p. 655
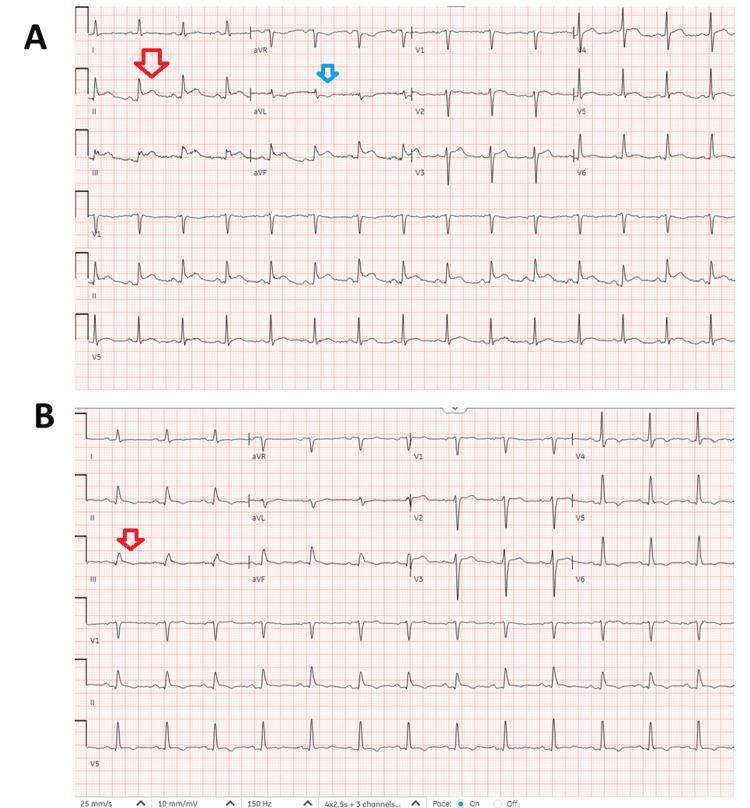
NSTEMI ECG Findings
- ST depression (horizontal or downsloping) in precordial leads
- T-wave inversion (deep symmetrical — "Wellens pattern" with proximal LAD involvement)
- Nonspecific ST-T changes
- Normal ECG (~30% of cases — diagnosis rests on troponin)
- No ST elevation and no pathologic Q waves (by definition)
Biomarkers
| Biomarker | STEMI | NSTEMI |
|---|---|---|
| High-sensitivity Troponin I/T | ↑ (may be negative in first 1–2 hrs) | ↑ (serial testing required if initial negative) |
| CK-MB | ↑ | ↑ (smaller rise) |
| BNP/NT-proBNP | ↑ if LV dysfunction develops | ↑ if LV dysfunction develops |
- Diagnosis of MI requires acute rise and/or fall of troponin with at least one value above the 99th percentile upper reference limit + clinical evidence of ischemia. — Goldman-Cecil Medicine, p. 655
- UA vs NSTEMI distinction: same presentation, but UA has negative troponins (no necrosis).
Clinical Presentation
Both share the same symptoms — but STEMI tends to be more severe and sudden:
- Chest pain: Persistent (>10–20 min), heavy retrosternal pressure, radiation to left arm/jaw/shoulder — not relieved by rest or nitroglycerin
- Associated symptoms: Diaphoresis, nausea, vomiting, dyspnea, weakness
- Silent MI: ~20% of MIs are painless; more common in elderly, women, and diabetics
- Autonomic signs: anterior MI → sympathetic activation (tachycardia, hypertension, diaphoresis); inferior MI → vagal activation (bradycardia, hypotension)
Infarct Territories by Culprit Artery
| Artery | Territory | ECG Leads |
|---|---|---|
| LAD (left anterior descending) | Anterior wall, septum, apex | V1–V6 |
| RCA (right coronary artery) | Inferior wall, RV | II, III, aVF |
| LCx (left circumflex) | Lateral / posterior wall | I, aVL, V5–V6; posterior leads V7–V9 |
Posterior STEMI: ST depression in V1–V4 may indicate posterior injury from LCx occlusion — extend to posterior leads (V7–V9) to confirm. — Goldman-Cecil Medicine, p. 655
Management: The Critical Difference
The key principle: STEMI = emergent reperfusion NOW. NSTEMI = risk-stratified invasive approach within hours to days.
STEMI Management
Time is muscle. Myocardial salvage is inversely proportional to ischemia duration.
Step 1 — Immediate (<10 min after arrival)
- 12-lead ECG
- Aspirin 325 mg (chewed)
- IV access, oxygen (if SpO₂ <90%), cardiac monitoring
- Nitroglycerin (unless contraindicated — hypotension, recent sildenafil use, inferior MI with RV involvement)
- Morphine 2–4 mg IV for refractory pain (use cautiously — may mask symptoms)
- Beta-blocker (oral, within 24 hrs) if no HF, cardiogenic shock, or bradycardia
Step 2 — Reperfusion Strategy
| Strategy | When to Use |
|---|---|
| Primary PCI (preferred) | When available within 90 min of first medical contact |
| Fibrinolysis | When PCI not available within 90–120 min; give within 30 min of door arrival |
| Emergency CABG | Failed PCI, anatomy not suitable for PCI, mechanical complications |
Primary PCI advantages over fibrinolysis: superior vessel patency (TIMI 3 flow), less reinfarction, no intracranial hemorrhage risk, better survival. Always preferred in cardiogenic shock, Killip class III/IV, fibrinolysis contraindications, or prior PCI/CABG. — Washington Manual of Medicine
Signs of successful reperfusion after fibrinolysis:
- Chest pain relief
- ≥50% reduction of ST elevation at 60–90 min
- Reperfusion arrhythmia (accelerated idioventricular rhythm) → Absence = rescue PCI required
Step 3 — Antithrombotic therapy
- DAPT: Aspirin + P2Y12 inhibitor (ticagrelor or prasugrel preferred over clopidogrel)
- Anticoagulation: UFH or bivalirudin (with PCI); LMWH or fondaparinux (with fibrinolysis)
- Complete revascularization: Non-culprit lesions amenable to PCI should be treated (reduces CV death/MI); timing = before discharge or staged outpatient
Step 4 — Post-MI surveillance
- Malignant arrhythmias (VF, VT) — peak risk first 24–48 hrs
- Mechanical complications: free wall rupture, papillary muscle rupture (MR), VSD
- RV infarction (inferior STEMI): treat with IV fluids, avoid nitrates
- LV function assessment (echo) before discharge
NSTEMI Management
Less time-critical than STEMI but still urgent — risk stratification determines timing of invasive strategy.
Step 1 — Immediate medical therapy (all patients)
- Aspirin + P2Y12 inhibitor (DAPT)
- Anticoagulation: LMWH (enoxaparin), UFH, fondaparinux, or bivalirudin
- Beta-blocker (oral)
- High-intensity statin
- ACE inhibitor / ARB
- Nitroglycerin for ongoing pain
Step 2 — Risk Stratification
| Risk Score | Use |
|---|---|
| TIMI score (0–7) | ≥3 = benefit from early invasive strategy |
| GRACE score | Better discriminates in-hospital mortality |
High-risk features mandating urgent angiography (<2 hrs):
- Refractory ischemia / ongoing chest pain
- Cardiogenic shock or hemodynamic instability
- Life-threatening arrhythmias
- Acute HF / pulmonary edema
- Mechanical complications
Step 3 — Invasive Strategy Timing
- Very high risk → Immediate angiography (<2 hrs)
- High risk (positive troponin, new ST changes, GRACE >140) → Early invasive (<24 hrs)
- Intermediate risk → Invasive within 24–72 hrs
- Low risk → Consider non-invasive testing first (conservative strategy)
Randomized trials show PCI is superior to intensive medical therapy alone in reducing mortality and MI, with the benefit largely confined to high-risk patients. — Harrison's Principles, 22e
Step 4 — GPIIb/IIIa inhibitors (e.g., eptifibatide, tirofiban): Consider in high-risk patients or as "upstream" therapy before catheterization if not already on potent P2Y12 inhibitor.
Side-by-Side Summary Table
| Feature | NSTEMI | STEMI |
|---|---|---|
| Thrombus | Partial occlusion | Complete occlusion |
| Necrosis depth | Subendocardial | Transmural |
| ECG | ST depression / T-wave inversion / Normal | ST elevation / new LBBB |
| Q waves | Absent | Develop over hours–days |
| Troponin | ↑ (positive) | ↑ (may be early negative) |
| Reperfusion urgency | Hours to days (risk-based) | <90 min (emergent) |
| Primary PCI timing | 2–72 hrs based on risk | Immediately / door-to-balloon <90 min |
| Fibrinolysis | Not indicated | Yes, if PCI unavailable |
| Infarct size | Smaller (subendo) | Larger (transmural) |
| Mortality (30-day) | 10–20% (similar or worse long-term) | 5–10% (higher acute mortality) |
| Long-term prognosis | Can be worse long-term due to diffuse disease | Better long-term if reperfused rapidly |
Key Mnemonic
"No ST elevation? No fibrinolysis. Risk-stratify and go invasive within hours." "ST elevation? Time is muscle — primary PCI within 90 minutes."
Sources:
- Goldman-Cecil Medicine, International Edition, pp. 654–657
- Washington Manual of Medical Therapeutics, pp. 155–158
- Harrison's Principles of Internal Medicine, 22e
- 2025 ACC/AHA/ACEP/NAEMSP/SCAI ACS Guideline
This is a shared conversation. Sign in to Orris to start your own chat.